Treating a Patient With First-Line Low-Risk Polycythemia Vera
Raajit K. Rampal, MD, PhD, Memorial Sloan Kettering Cancer Center, New York, New York, explains his frontline treatment choices for the case of a 48-year-old patient with low-risk polycythemia vera (PV). In this case, the patient expressed concern with regard to both his long term prognosis and current symptoms with polycythemia vera, and wished to pursue a treatment strategy which could reduce his risk of cardiovascular and thrombotic events and reduce his risk of disease progression.
Transcript:
Hi, I'm Raajit Rampal. I'm the director of the myeloproliferative neoplasm program at Memorial Sloan Kettering Cancer Center in New York.
Today I'm going to discuss a case of a patient with polycythemia vera and some of the implications for treatment. We'll start with the presentation. We have a 48-year-old male who presents to his primary care physician with a 3-month history of increasingly frequent headaches and a persistent sense of lightheadedness that he thought was initially due to work stress. But he also complains of pruritus, particularly with warm showers, which he's tried to manage with topical solutions without any real relief. The patient also notes that he's very fatigued and often has to nap after work and on the weekends, which is new. He's had some shortness of breath with exertion. And in terms of his comorbidities, he has hypertension and hyperlipidemia for which he's on medications and which are well-managed.
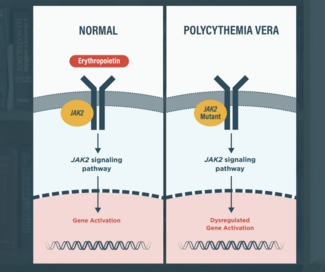
On exam, the patient has a ruddy complexion. His other cardio and pulmonary findings are within normal limits. The abdomen is notable for the palpable edge of the spleen, but otherwise the rest of the exam is unremarkable. The white count at presentation was 16.6 with a hemoglobin of 18.9 and a hematocrit of 58%, with a platelet count of 520,000. So the patient's referred to a hematologist for workup. And notably on his workup, a serum arthropoitin is subnormal and a JAK2 mutation, the canonical V617F mutation is detectable by peripheral blood next generation sequencing (NGS). And the variant allele fraction of this is about 30%.
The diagnosis is rendered here initially of polycythemia vera, and it's confirmed with a bone marrow examination. And the question is now, how do we approach this patient? To briefly summarize, we have a newly diagnosed patient with polycythemia vera who has no history of thrombotic events, but has medical history of hypertension and hyperlipidemia.
The patient is symptomatic from disease and therefore, of course, needs treatment. His hematocrit presentation is 58%, so it exceeds where we would like to see the hematocrit in a patient with polycythemia vera. Now, thinking about what the patient wants—the patient is worried about long-term prognosis as he's only 48 years old. He's worried about a symptom burden, and he really wants to think about treatment strategies that are going to reduce the immediate risk of the disease, which are cardiovascular and thrombotic events, but also the risk of disease progression.
As we think about polycythemia, we know that there is a risk of progression to myelofibrosis as well as to AML. We have a number of different things to deal with. We need to deal with symptoms. We need to attenuate the risk of thrombosis, and we need to try to reduce our risk of disease progression.
So what do we know in this instance? Well, certainly phlebotomy and aspirin have been considered standard of care, and we know that phlebotomizing patients and keeping their hematocrit under 45% is associated with better cardiovascular and overall outcomes than letting the hematocrit be higher than 45%. We know that aspirin certainly has an impact in terms of overall outcomes with regard to thrombosis in these patients. Those are 2 things for which there is randomized phase 3 data that shows that these are both beneficial things to think about. But as it pertains to symptoms and disease progression, these are a little bit more elusive. We do know that patients with PV do have a fairly large symptom profile, and that things like phlebotomy don't necessarily help with many of the symptoms. They can relieve things like feelings of shortness of breath, for example, but do they help with itching? Not necessarily. Do they help with fatigue? Not necessarily. What are other options?
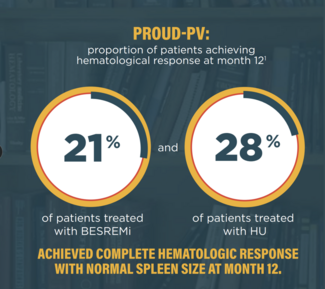
In terms of first-line therapies, we have ropeginterferon, which was approved based on a phase 3 trial versus hydroxyurea, and it was shown to be better than hydroxyurea in terms of count control. And now there is data further along from that trial that shows that there's actually an event-free survival benefit, and the events in that study were thrombotic events, progression to myelofibrosis, and progression to leukemia, all of the things that we care about. So that becomes an option for us, in this case.
What about JAK inhibitors? Ruxolitinib is FDA-approved for the treatment of polycythemia vera, but only in patients who are resistant or intolerant to hydroxyurea. So if that's the case, why not just use hydroxyurea at the get- go? Well, certainly there is retrospective data to show that hydrea, as compared to phlebotomy alone, may have a benefit in terms of reduction in thrombotic risk, but there really isn't any data to show that it helps with long-term outcomes. And again, you have data versus ropeginterferon showing that ropeginterferon is actually superior with regard to count control, but potentially also has an event-free survival benefit. We have a situation where starting this patient on aspirin and hydroxyurea is a possible consideration, but may not address symptoms or long-term disease control. Aspirin and phlebotomy certainly could be considered, but likely has no benefit with regards to long-term disease control.
We have the option here of ropeginterferon as a first-line approved therapy. That is a very reasonable thing. Ruxolitinib could be considered, but again, only if the patient is started on hydroxyurea potentially as a bridge to going on to ruxolitinib. There's also emerging event-free survival data supporting the use of ruxolitinib, but ropeginterferin can be used here in the frontline.
There are some other data to keep in mind. There have been upfront trials in younger, low-risk patients like this patient, randomized trials versus phlebotomy, ropeginterferon versus phlebotomy that have initially shown benefit with ropeginterferon. One randomized study that has shown this. There is a study, a retrospective study looking at outcomes of patients treated with interferons, hydroxyurea, or phlebotomy in terms of risk of their disease progression.
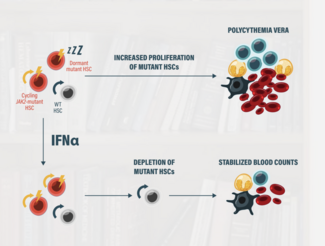
What it turns out from that analysis is that there is a long-term benefit for patients who are on interferons. This is historic data, so we always have to be careful with historic data, but nonetheless, the bulk of the data would suggest that interferons do potentially have disease-modifying activity. And in a patient like this who is really worried about long-term outcomes, that is a very reasonable choice to make.