Chronic Total Occlusion Percutaneous Coronary Intervention: 2026 Update
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/25.00301. Epub February 17, 2026.
Abstract
Percutaneous coronary intervention (PCI) for chronic total occlusion (CTO) is continually evolving through improvements in strategy, imaging, and equipment. This review provides a summary of the published literature in CTO PCI between September 2023 and April 2025, categorized by procedural outcomes, techniques, complications, and ongoing studies. Recent multicenter analyses report technical success rates exceeding 90% in expert centers, accompanied by significant reductions in angina and dyspnea at long-term follow-up. Despite higher complexity, radiation exposure has significantly decreased during the past decade as a result of equipment upgrades and operator awareness. Procedural refinements such as the use of coronary computed tomography for guidance, intravascular ultrasound guided re-entry, hydrodynamic contrast recanalization, novel retrograde techniques, and artificial intelligence-based tools are improving success and safety. Ongoing randomized trials and large-scale registries will continue to shape practice patterns and refine strategy selection in CTO PCI.
Introduction
Chronic total occlusion (CTO) percutaneous coronary intervention (PCI) is a dynamic field with continuous developments in equipment and techniques. This review summarizes contemporary evidence on CTO PCI published between September 2023 and April 2025, encompassing data from large multicenter registries, randomized and observational studies, and ongoing clinical investigations. The included studies address several key domains, including procedural and clinical outcomes, outcome prediction and risk stratification, imaging and technique optimization, outcomes in complex clinical and anatomical scenarios, complications, educational and cultural aspects of CTO PCI, and emerging areas of investigation. The following sections synthesize these findings to provide an integrated overview of recent advances and remaining challenges in contemporary CTO PCI practice.
Outcomes
Contemporary outcomes of CTO PCI have been primarily defined by large registries and multicenter studies, providing insight into procedural success, safety, and clinical effectiveness across a broad range of practice settings.
Observational studies
Impact of COVID-19 pandemic on CTO PCI
In the Prospective Global Registry for the Study of Chronic Total Occlusion Intervention (PROGRESS-CTO) registry, there was a 37.1% decline in CTO PCI volume during the pandemic.1 Technical (87.1%) and procedural success remained high, and major adverse cardiac event (MACE) rates were low across all periods. However, the incidence of in-hospital repeat PCI (0.4%) and acute myocardial infarction (MI) (0.7%) was slightly higher during the pandemic. The clinical significance of those findings is unknown.
Geographic diversity
Of 11 466 CTO PCIs in the PROGRESS-CTO registry, 8479 (74%) were performed in North America (NA).2 Compared with non-NA cases, NA patients had higher body mass index, were older, were more likely to have undergone prior coronary artery bypass graft surgery (CABG), and had more complex lesions. Use of the retrograde approach (31.0% vs 22.1%; P < .001), antegrade dissection and re-entry (ADR) (21.2% vs 9.2%; P < .001), and intravascular ultrasound (IVUS) (69.0% vs 10.1%; P < .001) was significantly more common in NA, as was use of plaque modification techniques such as intravascular lithotripsy (IVL), atherectomy, and scoring balloons. Technical success (86.7% vs 86.8%; P > .90), procedural success (85.4% vs 85.8%; P = .70), and in-hospital MACE (1.9% vs 1.7%; P = .40) were similar between regions.
Racial impact on outcome
In a study of 11 800 CTO PCIs from the PROGRESS-CTO registry, most patients were White (84.5%), followed by Black (5.7%), Hispanic (2.9%), Asian (2.4%), Native American (0.7%), and Other (3.9%). There was no significant difference between races in technical success or MACE.3 Whether the higher proportion of White patients represents some referral bias or other factors is not fully understood. The relatively lower representation of African-American patients in contemporary CTO PCI cohorts may, in part, reflect selection bias related to a higher prevalence of comorbid conditions such as chronic kidney disease, left ventricular dysfunction, and advanced vascular disease, which may influence referral patterns and procedural risk assessment. Although data on patients not selected for CTO PCI are limited, these observations highlight the need for further studies examining access to care, referral practices, and outcomes across racial and ethnic groups.
Registry outcomes
Mohebbi et al analyzed 790 CTO PCIs from the RAIAN (RAjaie - Iran) registry, reporting an overall success rate of 70% for antegrade and 80% for retrograde cases despite low availability of dedicated CTO devices.4 At 1-year follow-up, only the successful group had significant improvement in angina symptoms and quality of life (Table 1).
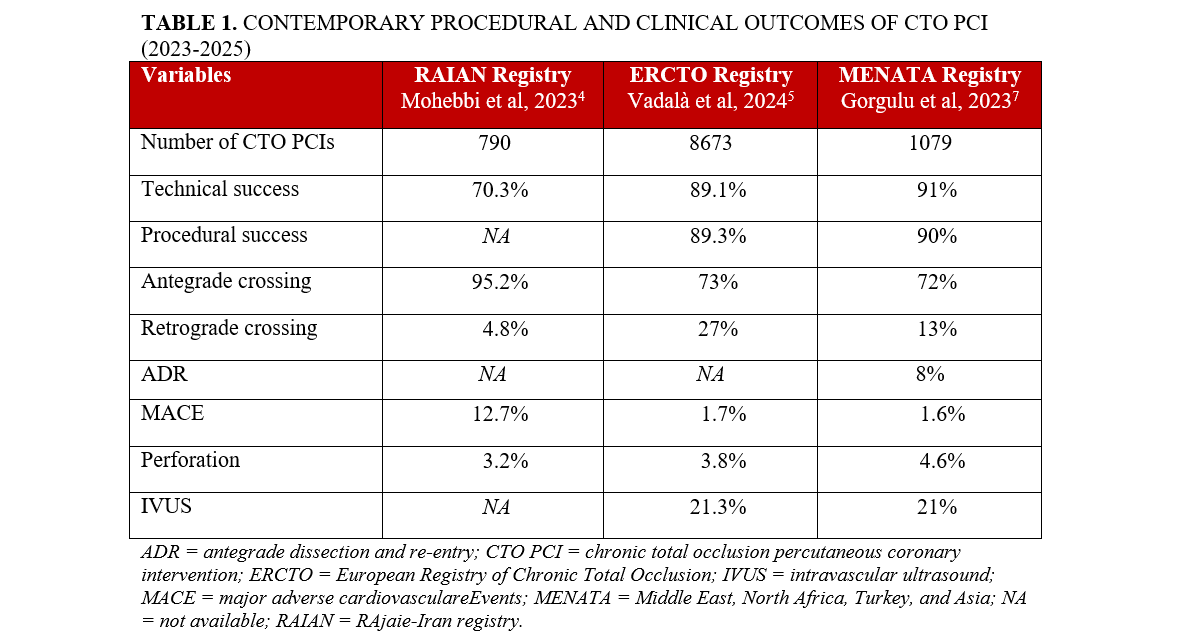
Vadalà et al analyzed the data from 8673 CTO PCI procedures performed between January 2021 and October 2022 in the European CTO registry5 comparing retrograde to antegrade PCI. Retrograde cases were more complex—as evidenced by a higher mean Japanese CTO (J-CTO) score (3.0 ± 1.0)—compared with antegrade cases (1.9 ± 1.2; P < .001), and had higher MACE (3.1% vs 1.2%; P = .018), higher coronary perforation rates (8.3% vs 2.1%; P < .001), and higher incidence of tamponade (1.5% vs 0.4%; P < .001).
Verrault-Julien et al looked at outcomes following retrograde or antegrade procedures from the Canadian CTO PCI (CCTOP) registry between March 2014 and October 2019.6 Out of 1033 patients, 48% used retrograde crossing. The retrograde approach was associated with higher lesions complexity (J-CTO score: 2.7 ± 1.1 vs 2.3 ± 1.1; P < .001) and lower technical success (81.2% vs 91.5%; P < .001). The risk of in-hospital MACE was higher with the retrograde approach (10.2% vs 4.7%, P < .001), and all deaths occurred in the retrograde group (0.8% vs 0%, P = .038).
Gorgulu et al described the in-hospital outcomes of CTO PCI across 10 high-volume centers in the MENATA region (Middle East, North Africa, Turkey, and Asia).7 A total of 1079 CTO PCIs were performed in 1063 patients between 2018 and 2022. The mean J-CTO score was 2.1 ± 1.2. Technical and procedural success rates were high (91% and 90%, respectively), even though the population included complex lesions with frequent proximal cap ambiguity, calcification, tortuosity, and long occlusion length. The incidence of MACE was low (1.6%) but was higher in failed procedures (7.5% vs 1.0%, P < .001). During follow-up, 81% of patients reported improvement in angina, 64% in dyspnea, and 60% in fatigue.
Contemporary procedural and clinical outcomes of CTO PCI from major registries are summarized in Table 1.
CTO PCI vs medical therapy
Van Veelen et al conducted an extended 10-year follow-up of the EXPLORE (Evaluating Xience and Left Ventricular Function in Percutaneous Coronary Intervention on Occlusions After ST-Elevation Myocardial Infarction) trial, which compared CTO PCI vs medical therapy in patients with ST-segment elevation MI (STEMI) who also had a concurrent CTO.8 The study included 302 patients who were randomized either to early CTO PCI within about 1 week after STEMI or to medical therapy without CTO PCI. The primary endpoint of the substudy, a composite of cardiovascular death, CABG, or MI, occurred in 25% of the CTO PCI group compared with 24% of the no-CTO PCI group (hazard ratio [HR], 1.11; 95% CI, 0.70-1.76). Cardiovascular mortality was higher in the CTO PCI group (16% vs 10%; HR, 2.09; 95% CI, 1.10-2.50), while all-cause mortality did not differ significantly between groups (25% vs 17%; HR, 1.53; 95% CI, 0.93-2.50). Although angina relief was similar between groups, the CTO PCI group had significantly greater improvement in dyspnea at 10 years (83% vs 65% in the medical therapy group, P = .005).
CTO PCI vs CABG
The Fractional Flow Reserve versus Angiography for Multivessel Evaluation (FAME) 3 trial randomized 1444 patients to fractional flow reserve (FFR)-guided PCI vs CABG in patients with 3-vessel coronary artery disease and showed that FFR-guided PCI did not meet the pre-specified noninferiority criterion compared with CABG for the 1-year composite outcome of death, MI, stroke, or repeat revascularization.9 Additional analyses demonstrated that revascularization of CTO lesions did not significantly improve outcomes compared with medical therapy alone in either treatment arm.
Time-dependent changes in coronary circulation after CTO PCI
Lombardi et al conducted a comprehensive meta-analysis of 17 studies evaluating intracoronary physiological changes triggered by CTO revascularization.10 The analysis revealed a consistent and significant improvement in myocardial fractional flow reserve (FFRmyo) in both the donor and CTO vessels. Specifically, FFRmyo in donor arteries increased shortly after revascularization (mean difference [MD], 0.04; 95% CI, 0.02-0.06; P < .01) as did the FFRmyo in the CTO vessel (MD, 0.45; 95% CI, 0.27-0.64; P < .01). After a mean follow-up of 4 months, collateral vessel FFR decreased (FFRcoll: MD -0.16; 95% CI, -0.18 to -0.15; P < .01), reflecting regression of collateral circulation as native flow resumed. During follow-up, absolute coronary blood flow increased (MD, 53.2 mL/min; 95% CI, 40.3-66.1; P < .01) and microvascular resistance decreased (fixed effects model: MD, -94.3 Wood units; 95% CI, -137.6 to -50.9; P < .01), suggesting favorable remodeling of the microcirculation.
Checklist-based nursing care
Ge et al conducted a quasi-randomized study evaluating the impact of a checklist-based nursing care process on 120 patients undergoing CTO PCI.11 The checklist-based process led to a significant reduction in preoperative nursing time (37.61 ± 5.15 vs 50.98 ± 5.61 min, P < .05) and was associated with fewer total adverse events (12% vs 50%, P < .05), including fewer puncture-site bleedings and pressure sores. In the intervention group, postoperative patient anxiety was lower, and doctor and patient satisfaction was higher.
Procedural outcomes
Vascular access
Radial access has been associated with a lower risk of vascular access complications compared with femoral access in CTO PCI.12 Distal radial access was shown to have similar success and complication rates with standard radial access in the PROGRESS-CTO registry.
Triple access is sometimes required in prior CABG patients who have multiple sources of filling of the distal true lumen. An alternative strategy is to use the sequential dual guide catheter technique in which only 2 guide catheters are used at any given time, changing guide catheters as CTO crossing progresses.13
Same-day discharge
In a series from the Netherlands that used large-bore access (≥ 7F sheaths in at least 1 access site in 99% of patients), same-day discharge (SDD) was done in 62% of 948 patients undergoing CTO PCI.14 Technical success (odds ratio [OR], 0.44; 95% CI, 0.25-0.78) and female sex (OR, 0.64; 95% CI, 0.44-0.95) were significantly associated with overnight stay. At 30-day follow-up, hospital readmission and MACE rates were low and comparable between the SDD and non-SDD patients.
Activated clotting time
Simsek et al investigated the relationship between activated clotting time (ACT) and clinical outcomes in CTO PCI in 4377 patients.15 ACT values were categorized as less than 250 seconds (19%), 250 to 349 seconds (50%), and greater than or equal to 350 seconds (31%). A U-shaped relationship emerged between mean ACT and net adverse cardiovascular events (NACE): both low (< 200 s) and high (> 400 s) ACTs were associated with higher NACE compared with the 200- to 400-second range (adjusted OR, 2.06; 95% CI, 1.18-3.62; P = .012).
Radiation dose
In the PROGRESS-CTO registry, the median air kerma (AK) radiation dose remained steady at 2.1 Gy (IQR: 1.1-3.6) with no significant reduction since 2020, despite prior improvements.16 Lesion complexity remained unchanged, suggesting plateaued gains in radiation safety. Werner et al reported a 52% reduction in AK radiation dose between 2012 and 2023 in the European CTO registry, from a median of 2.50 Gy (Q1-Q3: 1.54-4.04 Gy) to 1.20 Gy (Q1-Q3: 0.66-2.12 Gy) (P < .001), with the proportion of procedures exceeding 5 Gy dropping from 15.8% to 3.7%.17 Determinants of high AK radiation dose were higher BMI, female gender, prior CABG, and use of the retrograde approach.
Impact of CTO on TAVR outcomes
Of 1487 consecutive patients who underwent transcatheter aortic valve replacement (TAVR) at a single center, 11.2% had a concomitant CTO.18 CTO patients had significantly lower left ventricular ejection fraction (LVEF) and higher EuroSCORE values compared with those without CTO. However, the presence of a CTO was not associated with an increase in in-hospital or 30-day mortality, and no differences in long-term survival were observed. Subgroup analysis revealed that patients with CTOs supplying non-viable myocardium had significantly higher 30-day and follow-up mortality (P = .008 and P = .024, respectively), indicating that myocardial viability may be an important prognostic factor.
IVUS guidance
In the PROGRESS-CTO registry, which included 8983 CTO PCI cases, IVUS was used in 44.5% of procedures, most commonly for stent optimization (33.1%).19 Despite being applied in more complex lesions, IVUS use for stent optimization was associated with lower radiation exposure and contrast volume. In the PROGRESS-CTO registry, among cases in which the guidewire had already successfully crossed the CTO segment, IVUS-guided stent optimization was associated with a higher likelihood of achieving the registry-defined procedural endpoints compared with angiography guidance alone, as well as lower radiation exposure and contrast use (99% vs 96%, P < .001 and 96% vs 95%, P = .002).
Similar findings were observed in a retrospective analysis from a single high-volume US center including 5547 PCI procedures, of which 2040 (36.8%) were CTO PCI.20 After propensity score matching, systematic intravascular imaging (IVI) significantly reduced radiation dose, contrast volume, and the rate of cardiac tamponade (0.8% vs 1.6%, P = .015). Similarly, a retrospective study from a high-volume Japanese center (2884 CTO PCIs, with 69.5% performed under IVI guidance) showed that IVI-guided CTO PCI was associated with a significantly lower risk of coronary perforation (OR, 0.59; 95% CI, 0.43-0.80; P < .001).21
In the recent drug-coated balloon analysis of the European Registry of Chronic Total Occlusion (ERCTO), including 40 449 CTO-PCI, IVUS use was not associated with a lower risk of cardiac tamponade in multivariable regression analysis, but was independently associated with a higher rate of procedural completion and angiographic success.
In the prespecified CTO substudy of the RENOVATE-COMPLEX-PCI (Randomized Controlled Trial of Intravascular Imaging Guidance versus Angiography-Guidance on Clinical Outcomes after Complex Percutaneous Coronary Intervention), IVI guidance significantly reduced the risk of target vessel failure (TVF) (HR, 0.30; 95% CI, 0.13-0.71; P = .006) compared with angiographic guidance. When these optimization targets were achieved, the benefit was even greater, with a 10-fold reduction of TVF compared with angiography (1.3% vs 13.5%; HR, 0.14; 95% CI, 0.02-0.56; P = 0.003).22
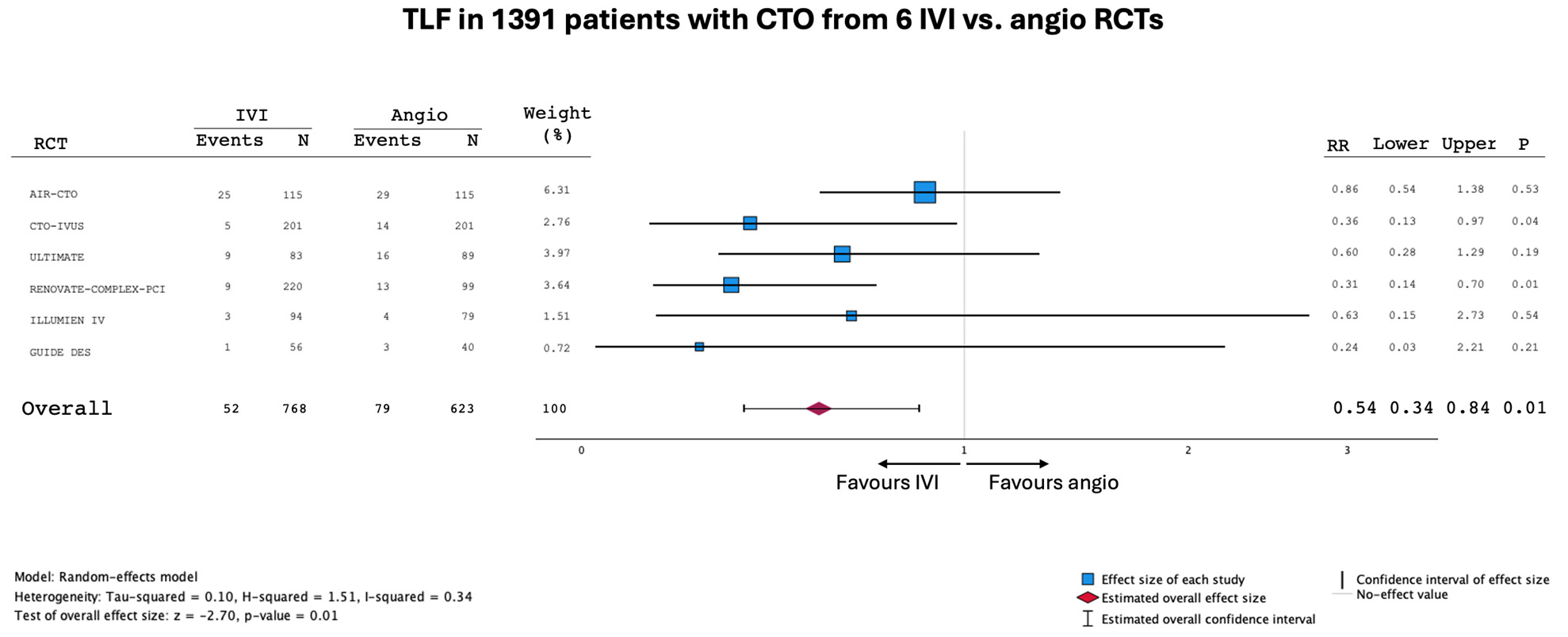
A recent meta-analysis including 5 randomized controlled trials (RCT) (1296 patients, 55% IVI-guided PCI) demonstrated that MACE was significantly reduced with IVI guidance (7.2% vs 13%; risk ratio [RR], 0.55; 95% CI, 0.35-0.88; P = .012). Target vessel revascularization was also lower in the IVI-guided group (3.1% vs 6.7%; RR, 0.52; 95% CI, 0.29-0.97; P = .038).23 Adding data from a sixth RCT (GUIDE-DES), the updated meta-analysis shown in Figure 1 confirmed these findings and demonstrated an even greater reduction in target lesion failure (TLF) among CTO patients compared with all-comers,24 further strengthening the recommendation for IVUS guidance in CTO recanalization.
Outcome prediction
Beyond overall outcomes, multiple contemporary studies have focused on predicting procedural complexity and success in CTO PCI, aiming to improve case selection and procedural planning.
Success and complications scores
The J-CTO and the PROGRESS-CTO Complications scores remain the most commonly used scores for determining the difficulty of CTO crossing and risk of complications, respectively.25 A vessel-specific J-CTO score was recently created26 but did not improve prediction on external validation.27 New scores were recently developed using machine learning with superior predictive accuracy (area under the curve [AUC] = 0.760 vs J-CTO: 0.697; Clinical and Lesion-related: 0.662; EuroCTO CASTLE: 0.659; P < .005 for all) but their utilization remains limited at present.20
Computed tomography-based scores
Coronary computed tomography angiography (CCTA) has been increasingly used in CTO planning, leading to the development of CT-based scoring systems that complement angiographic scores.28 The Computed Tomography Registry of CTO Revascularization (CT-RECTOR) and the Korean Multicenter CTO CT Registry (KCCT) are the 2 most widely used CCTA-based frameworks for procedural planning and outcome prediction.
The CT-RECTOR score incorporates lesion characteristics such as occlusion length, proximal cap morphology, degree of calcification, presence of side branches at the distal cap, and vessel course, aiming to predict guidewire crossing difficulty and procedural complexity. Similarly, the KCCT registry-derived score emphasizes CT-derived anatomical features including severe calcification, tortuosity, blunt proximal cap, occlusion length, and distal vessel quality.
Compared with angiography-based scores such as J-CTO, CT-based scores provide additional information on plaque composition, 3-dimensional vessel course, and distal vessel visualization, which may facilitate selection of antegrade vs retrograde strategies and procedural planning. However, CT-based scoring systems require high-quality imaging and are not universally available, limiting their routine use in all centers.29
Le et al found that pericoronary adipose tissue attenuation (PCATA) was independently associated with CTO PCI procedural success.30 They created the PCATA-CTO score that had an AUC of 0.72; this score includes severe calcification (1 point), lesion length greater than or equal to 15 mm (1 point), and perivascular fat attenuation index (FAI) less than or equal to -69.5 HU (1 point),
Xing et al studied 565 CTO lesions and developed a model based on 6 radiomic features, 3 shape-based and 3 texture-based, with an AUC of 0.86, accuracy of 74.6%, sensitivity of 81.6%, and specificity of 61.9%, significantly outperforming traditional scores such as J-CTO, PROGRESS-CTO, and CTA J-CTO.31
Zhou et al analyzed 534 patients with 565 CTO lesions and developed a deep learning (DL) model using CCTA data to predict guidewire crossing within 30 minutes and PCI success.32 In the external test set of 186 patients with 189 CTOs, the DL model demonstrated superior predictive performance compared with conventional CCTA-based manual scoring systems, with guidewire crossing success rates of 91.0% and PCI success rates of 93.7%. The DL model achieved a high AUC of 0.97 (95% CI, 0.89-0.99) in the training set and 0.96 (95% CI, 0.90-0.98) in an external test set. The DL model required less time for image reconstruction and analysis vs manual analysis (73.7 vs 418.2-466.9 s, P < .001).
Contemporary CTO PCI outcome prediction tools and scoring systems are summarized in Table 2.
Antegrade approach
Several parameters, such as proximal cap ambiguity, blunt or absent proximal stump, moderate/severe calcification and tortuosity, bifurcation at the distal cap, and in-stent occlusions can affect antegrade wiring (AW) success. Rempakos et al developed a machine learning score for determining the likelihood of antegrade wiring success with an AUC of 0.780 in the testing set.33 Occlusion length was the most impactful predictor of AW success, followed by blunt/no stump, interventional collaterals, vessel diameter, and proximal cap ambiguity. At the same time, aorto-ostial lesion location contributed the least.
Taniguchi et al examined 118 lesions approached with the parallel wire technique, with a success rate of 55% (65/118).34 In multivariable analysis, 2 parameters were associated with parallel wire success: lower J-CTO score (OR, 0.66; 95% CI, 0.44-0.99; P = .04), and sufficient clarity of the CTO exit site (OR, 5.16; 95% CI, 1.75-15.20; P < .01).
Retrograde approach
Several scores have been developed specifically for retrograde CTO PCI. Tanaka et al developed the Japanese Retrograde CTO (JR-CTO) score to predict guidewire success during primary retrograde CTO PCI using 4 variables: collateral connection (CC) grade less than or equal to 1, non-septal collateral channels, moderate to severe lesion calcification, and tortuosity.35 When compared with the J-CTO score, the JR-CTO score demonstrated superior discrimination (C-statistic 0.71 vs 0.60), better calibration, and greater net clinical benefit in decision curve analysis. Ochiumi et al showed that hemodialysis, distal runoff of less than 1 mm, the existence of calcification, and CTO lesion tortuosity (all P < .05) were associated with antegrade crossing failure of CTOs with short occlusion length (< 20 mm), favoring a primary retrograde approach.
The J-CTO channel score assesses the likelihood of successful collateral channel crossing based on 4 angiographic features: small channel size, presence of a reverse bend, presence of continuous bends, and a corkscrew-like course.36 Each feature adds complexity and is associated with decreased crossing success. The J-CTO channel score provided good prediction in 1 external study but modest in another study for both wire (AUC = 0.653) and microcatheter (AUC = 0.654) crossing.37 In a study of 257 retrograde CTO PCIs from the Netherlands, a successful collateral channel was achieved in 81% with an AUC of 0.743 for the J-channel score vs 0.699 for the Rentrop classification.38 In a study of 329 attempted collateral channels from Taiwan, the overall collateral crossing success rate was 67.8% and the final technical success rate was 92.2%.39 The simpler CC score (that only includes collateral size and tortuosity) had an AUC of 0.87 (95% CI, 0.83-0.90) compared with 0.61 (95% CI, 0.55-0.67) for the J-channel score (P < .001).
Across the EuroCTO and PROGRESS-MENATA registries, the retrograde approach was reserved for complex CTOs and associated with a higher risk.5,7 In EuroCTO, it was used in 20.8% of 8673 cases and had a higher J-CTO score (3.0 ± 1.0 vs 1.9 ± 1.2; P < .001), lower technical success (79.3% vs 92.8%; P < .001), and higher incidence of major adverse cardiac and cerebrovascular events (MACCE) (3.1% vs 1.2%), perforation (8.3% vs 2.1%), and tamponade (1.5% vs 0.4%). In PROGRESS-MENATA, retrograde access was more common in failed cases and was primarily through septal (70%) and epicardial (25%) collaterals. Perforations (4.6%) were more frequent in retrograde cases, most often due to wire exit, with 28% requiring pericardiocentesis. Despite higher risk, retrograde CTO PCI contributed to overall technical and procedural success rates above 90%.
Urgent mechanical circulatory support (MCS)
Among 2784 CTO PCIs in the PROGRESS-CTO registry, urgent mechanical circulatory support (MCS) was needed in 62 (2.2%). Patients requiring urgent MCS were older, with lower LVEF, greater lesion complexity, and more frequent use of retrograde techniques.40 A model was developed to predict urgent MCS use using the following parameters: (1) retrograde crossing strategy, (2) LVEF, and (3) lesion length. The model demonstrated good discriminative ability with an AUC of 0.79 (95% CI, 0.73-0.86), specificity of 86%, and sensitivity of 52%.
One-year outcomes
Hirai et al investigated the independent predictors of one-year outcomes following successful CTO PCI using new-generation drug-eluting stents, analyzing data in 3666 patients enrolled in the Japanese CTO-PCI Expert Registry.41 In-stent occlusion (ISO) was the strongest lesion-related determinant for both target lesion revascularization (TLR) (OR, 2.604) and MACCE (OR, 1.499). Hemodialysis was the most powerful patient-related predictor, being associated with significantly higher risk for both TLR (OR, 1.784) and MACCE (OR, 2.214). Other significant predictors for TLR included insulin-treated diabetes mellitus, moderate-to-severe calcification, and the right coronary artery (RCA) as the target vessel, while arteriosclerosis obliterans and multivessel disease were independently associated with MACCE.
Complex subgroups
Patient
Prior CABG. Prior CABG patients constituted 29% of 12 164 patients in the PROGRESS-CTO registry and were older, with more comorbidities, and more complex lesion characteristics.42 They more often required use of the retrograde approach and had lower technical (82.1% vs 88.2%) and procedural (80.8% vs 86.8%) success, longer procedure times, and higher radiation doses. Periprocedural complications such as MI (0.9% vs 0.5%), in-hospital mortality (0.8% vs 0.3%), and perforation (7.0% vs 4.2%) were higher, although pericardial tamponade was less frequent (0.1% vs 1.3%). On multivariable analysis, prior CABG was not independently associated with technical success (OR, 0.95; 95% CI, 0.82-1.11, P = .5), in-hospital mortality (OR, 1.53; 95% CI, 0.70-3.30; P = .3), or acute MI (OR, 1.50; 95% CI, 0.85-2.62; P = .2). This suggests that although prior CABG patients present greater procedural challenges and have higher unadjusted complication rates, the adverse outcomes are likely driven more by anatomical and procedural complexity rather than the CABG history itself. At 2-year follow-up, prior CABG patients showed higher rates of MACE, repeat PCI, and acute coronary syndromes, but all-cause mortality was similar to that of non-prior CABG patients.
In some prior CABG patients, retrograde CTO PCI is performed via a patent saphenous vein graft (SVG). Whether the SVG should be occluded following recanalization of the corresponding native coronary CTO remains controversial. Poletti et al showed that at 1-year follow-up, CTO vessel longer-term failure was significantly more frequent in cases with a final bypass graft Thrombolysis in Myocardial Infarction (TIMI) flow of 3 compared with a TIMI flow of less than 3 (20.6% vs 4.7%; HR, 5.6; 95% CI, 1.2-27.5; P = .033).43 Hence, intentional occlusion with coils of the SVG is preferable if it has TIMI 3 following native CTO recanalization.
Surgical and post-surgical CTO PCI subgroups are summarized in Table 3.

Left ventricular dysfunction. CTO PCI in patients with low LVEF has been associated with higher in-hospital and follow-up mortality. Sammour et al showed that mean LVEF improved from 48.2% ± 15.4% at baseline to 51.8% ± 14.2% after 8.4 months of follow-up (ΔLVEF: +3.6%; P < .001).44 Among patients with reduced baseline LVEF (< 50%, n = 50), the mean LVEF improved significantly from 32.6% ± 9.7% to 40.0% ± 12.9% (ΔLVEF: +7.6%; P < .001), and 48% of these patients experienced an increase of 10% or more in LVEF.
Gallo et al reported outcomes of 27 patients with severe left ventricular dysfunction (mean baseline LVEF: 23 ± 7%), CTOs, and complex multivessel coronary artery disease (SYNTAX score: 35 ± 11) undergoing PCI with the Impella device (Abiomed).45 At 90-day follow-up, LVEF significantly improved (mean ΔLVEF: +10%; 95% CI, 6-15), as did the patient symptoms, with 83% achieving Canadian Cardiovascular Society class 1 angina status and 71% achieving New York Heart Association class 1 dyspnea status. The incidence of MACE was 11%.
Obstructive sleep apnea syndrome (OSAS). In an analysis of 7403 patients who underwent CTO PCI, 942 (13%) had OSAS.46 Compared with patients without OSAS, those with OSAS were older, more often male, had a higher prevalence of comorbidities, had more complex CTOs, and had similar success and in-hospital MACE but higher follow-up MACE (HR, 2.32; 95% CI, 1.22-3.26; P = .006).
Lesion location
Left main coronary artery (LMCA) CTOs. In an analysis of the PROGRESS-CTO registry, 85 of 15 254 CTO PCIs (0.6%) performed in 14 969 patients were left main (LM) CTO PCIs.47 LM CTOs were more complex and more often required elective use of ventricular assist devices (VADs) (8.2% vs 2.0%, P = .002) but had similar procedural success and MACE compared with non-LM CTOs.
Ostial CTOs. In an analysis of 10 814 CTOs from the Japanese CTO-PCI expert pegistry between January 2014 and December 2022, 3.9% were ostial, of which 52% were aorto-ostial CTOs.48 Aorto-ostial CTOs were more complex with higher J-CTO scores and a higher need for retrograde crossing, but technical success was 88% (191/218).
In an analysis of 8788 CTO PCIs from the PROGRESS-CTO registry, 1058 (12%) were ostial CTOs: 68.1% aorto-ostial and 31.9% side-branch ostial. Ostial CTOs had higher complexity, required retrograde crossing more often (32.4% vs 16.2%, P < .001), and had lower technical (82.3% vs 86.2%, P = .001) and procedural (80.6% vs 84.8%, P = .001) success. On multivariable analysis, ostial location was not independently associated with technical success (OR, 1.03; 95% CI, 0.83-1.29; P = .73) or in-hospital MACE (OR, 1.15; 95% CI, 0.71-1.86; P = .56). However, side-branch ostial CTOs were independently associated with higher in-hospital MACE (OR, 2.23; 95% CI, 1.1-4.1; P = .01).
Bifurcation lesions. In an analysis from the EuroCTO registry, approximately one-third of all CTOs involved a bifurcation involving the proximal (38%) or distal (37%) cap or both (9%), and 16% the occlusion body (intralesional).49 CTOs with intralesional bifurcations had significantly lower technical success (83.7%) compared with bifurcations located at the proximal (93.3%), distal (93.4%), or both caps (89.0%) (P < .001). PCI of CTOs involving bifurcations was associated with higher side branch occlusion (2.5% vs 0.9%, P < .001), urgent target vessel revascularization (0.3% vs 0.1%, P = .025), and hemodynamic instability (4.9% vs 1.7%, P < .001), particularly in the intralesional and distal bifurcation subgroups.
Key lesion-related features associated with increased procedural complexity in CTO PCI are summarized in Table 4.

Lesion morphology
Proximal vessel tortuosity. Proximal vessel tortuosity is part of the PROGRESS-CTO and CASTLE (Coronary artery bypass graft history, Age (≥ 70 years), Stump anatomy, Tortuosity degree, Length of occlusion, and Extent of calcification) scores. In a study of 14 141 patients, 3974 (28.1%) had moderate or severe proximal vessel tortuosity. These patients had more comorbidities and complex angiographic features, including longer lesion length and a higher prevalence of a side branch at the proximal cap.50 Tortuous lesions were also more often balloon uncrossable and undilatable. Technical and procedural success rates were significantly lower in patients with moderate or severe proximal vessel tortuosity compared with those with mild or no tortuosity (82.6% vs 88.8% and 81.1% vs 87.6%, respectively; P < .001 for both). In-hospital MACE was also more frequent in these patients (2.34% vs 1.73%, P = .021), who also required longer procedure times (median: 128.00 vs 106.00 minutes, P < .001), higher radiation doses, and more stents (2.17 ± 1.02 vs 2.36 ± 1.11, P < .001). On multivariable analysis, proximal tortuosity remained independently associated with lower technical success but not with a significant increase in MACE.
Calcium. In an analysis of 13 079 CTO PCIs, moderate or severe angiographic calcification was present in 46.6% of lesions.51 Calcified CTOs were more complex, often requiring retrograde crossing both as the initial (16.5% vs 10.1%) and final successful crossing strategy (22.5% vs 15.1%). Rotational atherectomy was used in 7.3% of cases, laser atherectomy in 3.4%, IVL in 3.4%, scoring balloons in 3.3%, cutting balloons in 1.8%, and orbital atherectomy in 1.5%. Calcified lesions had lower technical (83.0% vs 89.9%) and procedural (81.0% vs 89.1%) success (P < .001 for both), and higher in-hospital MACE (3.0% vs 1.2%; P < .001), tamponade (1.7% vs 0.5%) and coronary perforation (6.5% vs 3.4%).
Full moon calcification (involving the entire vessel area on CCTA) was present in 20% of 140 CTO PCIs in 1 series and was associated with lower procedural success (71.4% vs 87.5%) and higher incidence of coronary perforation (14.2% vs 3.5%).52
Myocardial bridging (MB). In a cohort of 5959 left anterior descending artery (LAD) lesions, MB was significantly more common in CTO than in non-CTO lesions (40.4% vs 25.8%, P < .0001).53 Nearly half (48.7%) of 113 LAD CTOs with an MB had a stent that extended into the MB, which was associated with higher TLF at 2 years: 26.3% vs 0% in LAD CTOs without MB stenting (P = .0004), and 9.6% in LAD CTOs without MB (P = .02). Hence, MB stenting should be avoided.
Antegrade
Several new antegrade crossing techniques have been introduced with variable implementation across different geographies.
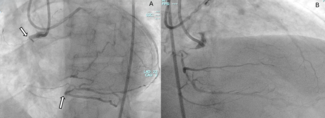
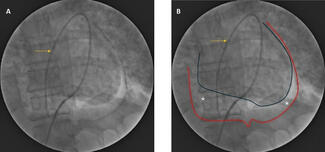
Parallel wiring (PW). PW remains a key technique for antegrade crossing if the initially used guidewire enters the extraplaque space (Figure 2). In an analysis from the EuroCTO registry, PW was used in 10.8% of primary antegrade cases, with its use increasing from 6.7% in 2015 to 10.7% in 2022.54 Use of a dual-lumen microcatheter also increased from 8.3% to 17% during the study period. Compared with ADR, PW had similar success but lower complication rates and required less radiation and contrast. Over time, Gaia (ASAHI INTECC), Pilot (Abbott), or Gladius (ASAHI INTECC) wires were more often used as the final successful wire in PW cases.
Brilakis ES. Manual of Coronary Chronic Total Occlusion Interventions. A Step-by-Step Approach. 3rd ed. Elsevier; 2023.
ADR. In an analysis from PROGRESS-CTO, ADR use declined from 37.9% in 2012 to 14.5% in 2022 (P < .001).55 Lesions treated with ADR were more complex. ADR was associated with lower technical success and higher in-hospital MACE. Among re-entry techniques, the STINGRAY device (Boston Scientific) was initially the most common (used in 55% of ADR cases), but its use declined in recent years, with Subinimal Tracking and Re-entry (STAR) becoming the most frequently used re-entry technique in 2022 (44% STAR vs 38% STINGRAY; P for trend < .001). Garbo described a modification of the STAR technique (called IVUS-guided STAR 2.0) by emphasizing precise inflow and outflow management, confirmed and guided via IVUS at each procedural step and also incorporating the use of drug-coated balloons in cases with long dissection planes.56
Hirai et al performed a 6-center randomized trial to determine the optimal timing of staged PCI following STAR in CTO-PCI.57 The study enrolled 150 patients and showed a trend for higher technical success with early (5-7 weeks) vs late (12-14 weeks) staged PCI (83.6% vs 71.4%, P = .08).58 TIMI flow grade 2 to 3 in the target vessel at the start of staged procedure was higher in the early group (64.4% vs 44.2%, P = .012; P = .048 after adjustment).
Extraplaque hematoma can hinder re-entry. Aspiration through the STINGRAY balloon itself or through a microcatheter using a syringe can decompress the hematoma, re-expand the distal true lumen, and facilitate re-entry.59 A Penumbra pump (Penumbra, Inc.) can also be used to decompress an extraplaque hematoma by applying stronger and continuous suction.60
The balloon-assisted subintimal entry (BASE technique) is often used to gain access into the extraplaque space.61 A novel “double-side BASE” can be used when the proximal cap is a trifurcation to allow extraplaque crossing of the CTO while maintaining access to all side branches.62
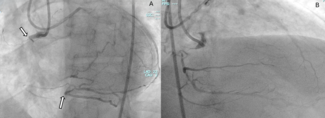
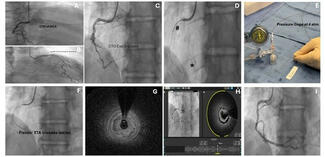
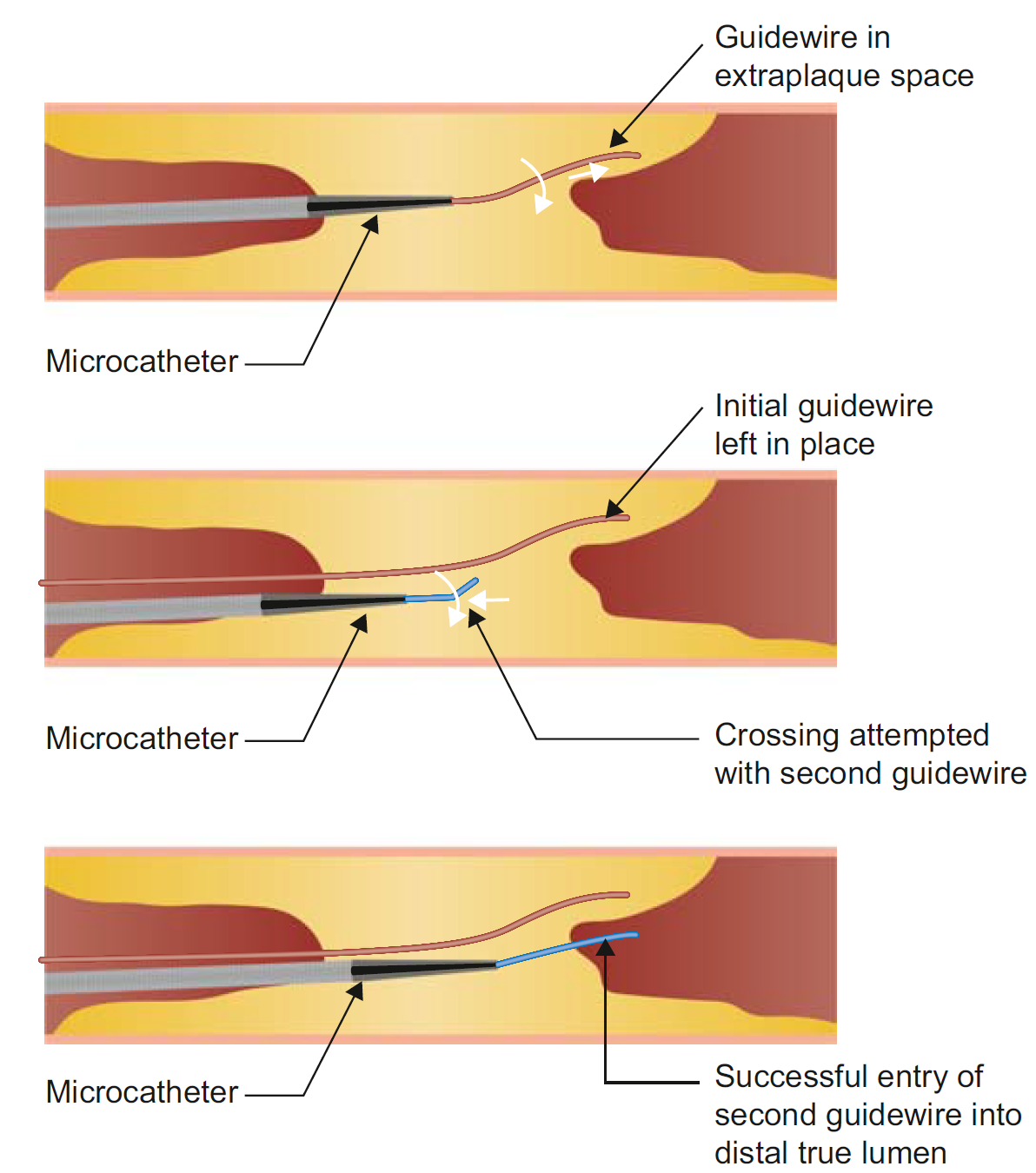
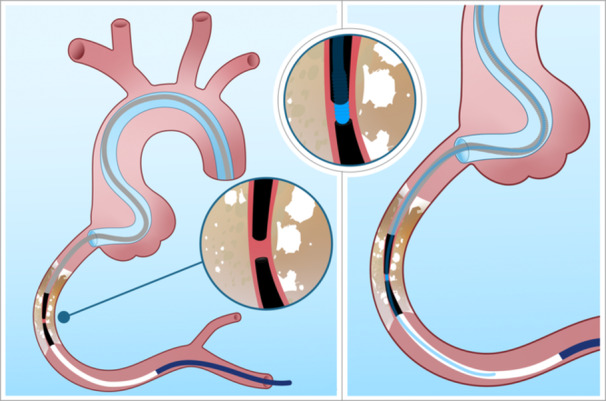
Tip detection ADR (TD-ADR). TD-ADR uses intravascular ultrasound to locate the wire tip and guide puncture from the extraplaque space into the true lumen (Figure 3). It can be facilitated by using a Conquest Pro 12 Sharpened Tip wire (ASAHI INTECC).63 It may also be assisted by the subintimal transcatheter withdrawal (STRAW) technique in case of large extraplaque hematoma.64 In a study of 54 CTO PCIs, TD-ADR had 100% success compared with 67% for STINGRAY-facilitated ADR (P = .002).65 TD-ADR was associated with a shorter procedural time and lower radiation dose, contrast volume, and subintimal passage length. However, TD-ADR requires 2 experienced operators, is technically demanding, and is still used infrequently outside Japan.
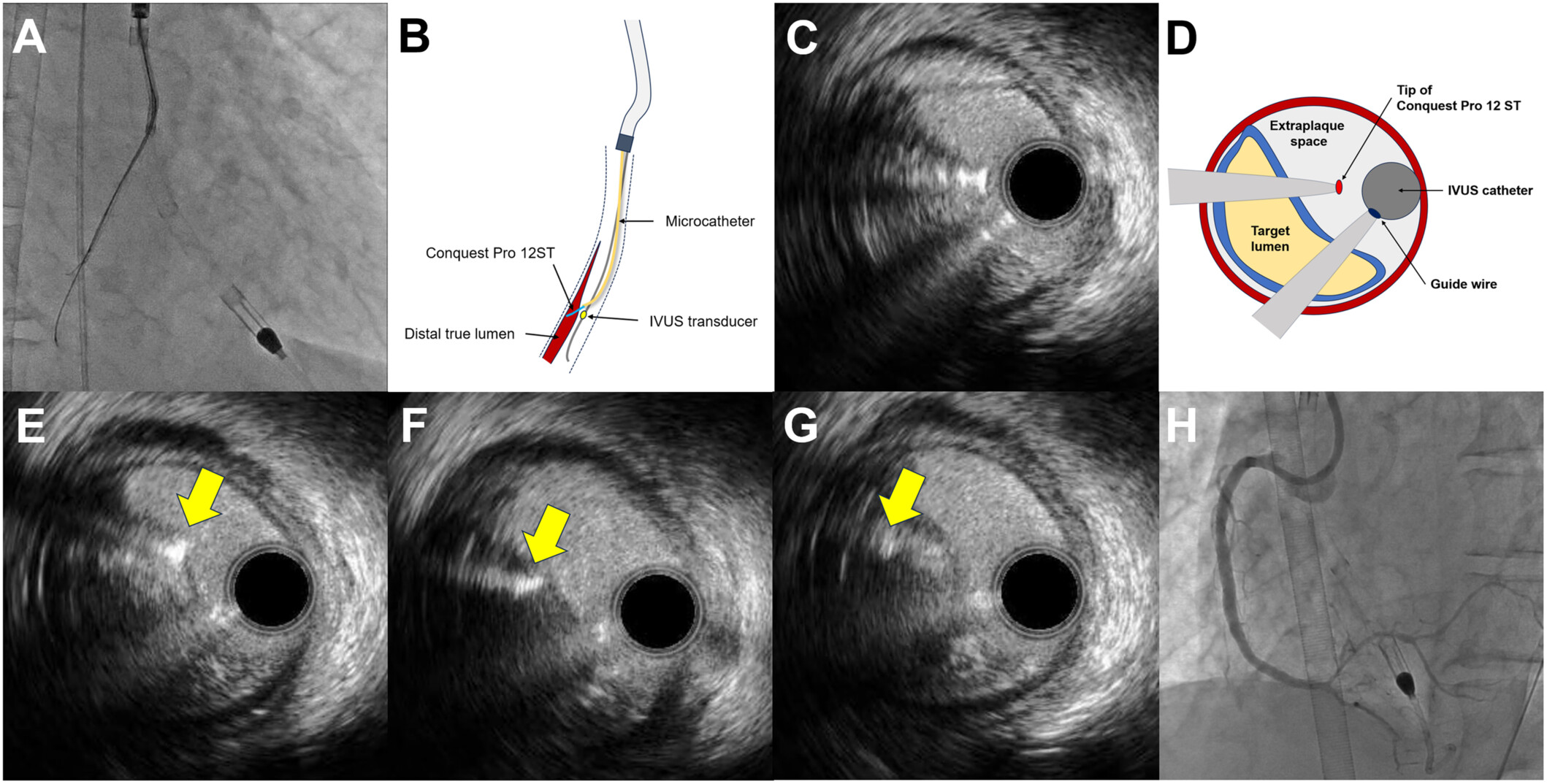
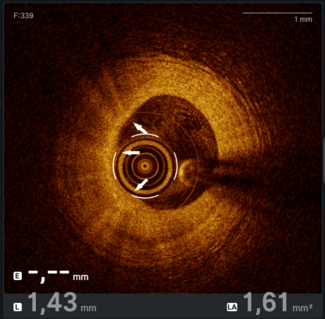
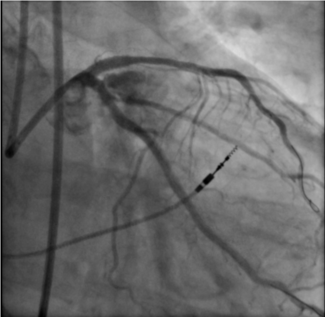
Hydrodynamic contrast recanalization (HDR). The Carlino technique (contrast injection into the CTO through a microcatheter) has traditionally been used as a bailout technique for wire-uncrossable CTOs and to clarify wire position.66 Recently, Carlino et al introduced hydrodynamic contrast recanalization (HDR), which integrates contrast microinjection with the controlled use of polymer-jacketed wires to create an intraplaque path for CTO crossing.67 Unlike traditional contrast-based strategies often reserved for bailout situations, HDR is applied early, particularly in anatomically favorable cases where both the proximal cap and distal target are well visualized.
HDR uses a medium-to-high penetration force wire for proximal cap puncture. The wire is advanced only a few millimeters into the CTO body to minimize extraplaque tracking. A torqueable microcatheter is then positioned just beyond the proximal cap, and a small volume of contrast (≤ 0.5 mL) is injected gently under cineangiography. This creates a visual roadmap of the intraplaque space, revealing characteristic contrast patterns (Figure 4). A polymer-jacketed wire (eg, Fielder or Gladius Mongo [ASAHI INTECC]) is then steered along the contrast-stained channel. If progression is halted, another microinjection can be performed slightly further along the CTO body to redefine the plaque path. If needed, the operator can repuncture the cap at a new location or switch to dissection/reentry or retrograde crossing.
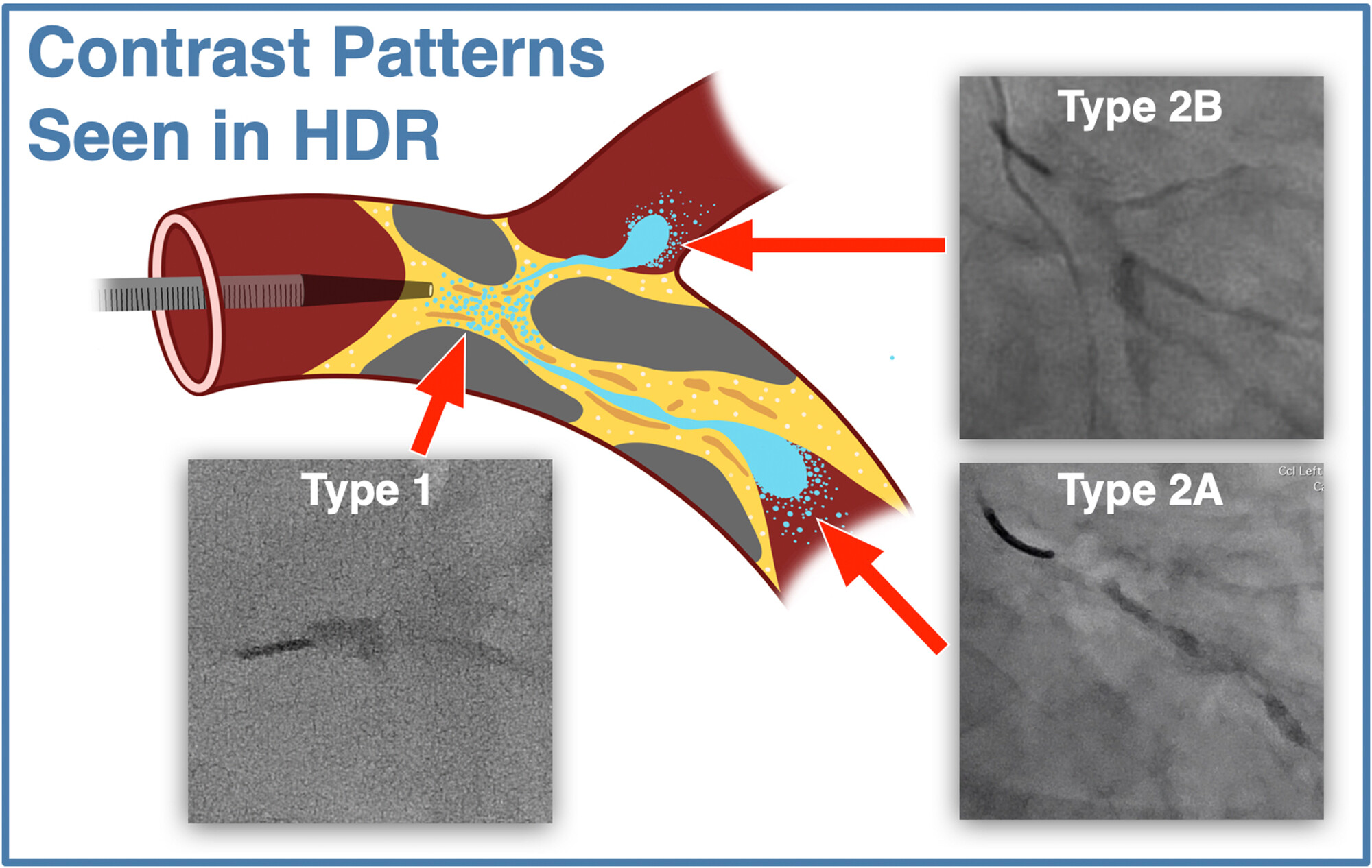
In a first-in-man series, HDR was employed in 45 CTO lesions, achieving 100% technical success with no reported complications. Median crossing time was 12 minutes, and a median of 3 wires was needed for lesion crossing. Additional studies are required to better understand the outcomes of HDR.
Three-dimensional wiring. Three-dimensional (3D) wiring can be done using fluoroscopic guidance.68 AW advancement should stop 10-mm proximal to the distal cap, preserving adequate longitudinal distance for controlled redirection in untouched vessel territory. Next, the microcatheter is advanced close to the wire tip to maximize support, and high-penetration wires like the Gaia Next 3 (ASAHI INTECC), Conquest 12g (ASAHI INTECC), or Hornet 14 (Boston Scientific) are recommended (bent at 45° at 1 mm and an additional 15° at 7 mm). A torque device is essential for precise angular control of the wire. Recognizing distal cap morphology is critical, as tapering, calcified, blunt, or side-branch-adjacent caps represent “tough” anatomies requiring an upgraded wire strategy. Dual-plane retrograde angiography (eg, left anterior oblique [LAO] 45/right anterior oblique [RAO] 45 for mid-RCA, LAO cranial 30/RAO cranial 30 for mid-distal LAD) enables an accurate 3D understanding of the cap, allowing primary rotation in the far view and secondary fine tuning in the near view. The technique highlights that far views provide better spatial accuracy, whereas near views carry parallax distortion, warranting cautious wire rotation of less than 40°. In cases of a near-side miss, the wire should be pulled back 15 mm to gain directional runway. For far-side overshoots, a 180° rotation after centering the wire just proximal to the distal cap is advised before reattempting puncture.
Retrograde
Retrograde outcomes. While the Retrograde approach section focuses on angiographic and collateral-based tools used to predict the feasibility of a retrograde approach, the following section summarizes contemporary procedural outcomes and complications associated with retrograde CTO PCI in large registries. Retrograde crossing remains a key technique for achieving high success rates, especially in complex CTOs. In an analysis of 11 808 CTO PCIs from the PROGRESS-CTO registry, retrograde crossing was attempted in 4058 cases (34.3%).69 The retrograde lesions were highly complex. Retrograde collateral crossing pathways included septal collaterals (62.0%), SVGs (17.4%), and epicardial collaterals (19.1%). Success rates varied by collateral size: 72.0% for Werner CC2, 59.8% for CC1, and 46.0% for CC0 connections. The Sion, Sion Black, and Suoh 03 (all ASAHI INTECC) were the most commonly used guidewires for collateral crossing. At the same time, Corsair/Corsair Pro (ASAHI INTECC), Turnpike LP (Teleflex), and Caravel (ASAHI INTECC) microcatheters were the preferred microcatheters. Pericardiocentesis was needed in 1.3% of cases, while any coronary perforation occurred in 9.5% of all retrograde attempts.
Retrograde crossing. In the PROGRESS CTO registry, among successful crossing strategies, retrograde true lumen crossing (31.8%) and conventional reverse controlled antegrade and retrograde tracking (CART) (34.4%) were the most common crossing techniques, followed by guide extension-assisted reverse CART (19.4%), AW with the retrograde wire as a marker (5.4%), contemporary reverse CART (4.4%), extended reverse CART (2.0%), and CART (2.1%).70 Use of guide extension-assisted reverse CART increased over time, while conventional reverse CART declined (P < .001).69 The Ringer perfusion balloon (Teleflex) has also been successfully used to facilitate reverse CART.70
The CART technique (retrograde balloon inflation followed by AW) is increasingly being used: Moscardelli et al reported use of CART in 45 of 603 retrograde cases (7.5%) often using SVGs for retrograde access (62.2%).71 Despite high anatomic complexity, the overall success rate of CART was 73.3%, with 82.2% technical and procedural success. Even when CART failed (26.7%), it often facilitated subsequent reverse CART. Procedural complications included coronary perforations (8.9%), without any occurrence of in-hospital deaths or other MACE.
The electrocautery-assisted re-entry (E-CART) technique can also be used for challenging re-entry, such as in cases of aorto-ostial CTOs caused by leaflet obstruction following TAVR.72
Sometimes retrograde crossing fails as the antegrade and retrograde wires are in different planes (extraplaque–intraplaque). Moscardelli et al successfully employed intracoronary snaring of the retrograde externalization wire to overcome a dissection flap barrier.73 After wiring the retrograde system into a side branch, an antegrade snare was used to capture the retrograde guidewire. The flap was severed pon retraction, separating the 2 planes and establishing continuity between the systems and enabling completion of the PCI.
Portal techniques. Portal techniques are used when externalization of the retrograde wire is not possible or carries high risk, such as in cases of epicardial collaterals, extreme angulation, or severe calcification.74 A key portal technique is “tip-in”, where the retrograde wire is directly intubated into an antegrade microcatheter positioned in the guide catheter or guide extension, allowing the microcatheter to advance through the CTO over the retrograde wire (Figure 5).75 Variants include the facilitated tip-in technique, in which the back end of a guidewire is inserted into the antegrade microcatheter for added support, and the “balloon tip-in”, which substitutes the microcatheter with a low-profile balloon for cases where immediate lesion preparation and PCI are feasible without externalization. There is also the “bite-in” technique, wherein the antegrade microcatheter is advanced and rotated through a guide catheter extension to “capture” the retrograde wire passively.76
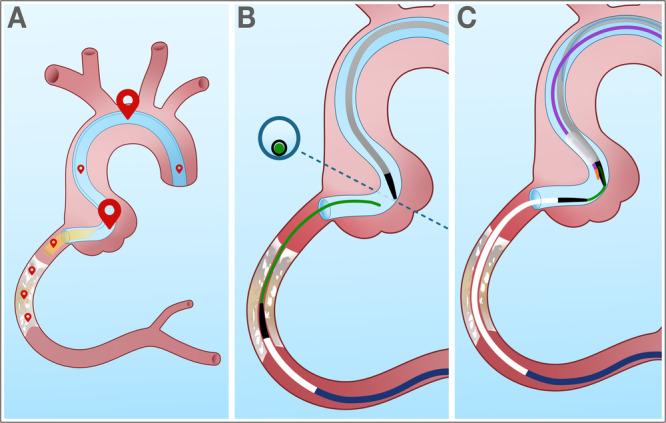
Another portal technique is the rendezvous technique, in which an antegrade wire is advanced into the retrograde MC within the CTO body (Figure 6).
“Catch-it” leverages the aortic arch, subclavian artery, or coronary cusp to guide the retrograde wire into the antegrade guide catheter without snaring. Antegrade microcatheter probing allows rapid conversion to antegrade work after retrograde microcatheter crossing by introducing an antegrade wire into the retrograde microcatheter inside the guide.
Ensuring coaxial alignment of catheters, selecting microcatheters with radiopaque tips for visibility, using supportive wires like Gaia or polymer-jacketed wires depending on lesion characteristics, and applying techniques like balloon-assisted tip-in or reverse trapping for added stability can facilitate the portal techniques. Troubleshooting includes adjusting catheter position, reshaping wires for optimal angles, utilizing biplane imaging for precise alignment, and coordinating push-pull maneuvers between operators. The portal techniques reduce manipulation of collateral channels and donor artery ostia, potentially lowering complication rates, and should be used when traditional retrograde connection strategies fail or are not desirable.
Retrograde microcatheter uncrossable CTOs. The retrograde microcatheter sometimes fails to advance through the CTO after successful crossing with a wire. To overcome this challenge, Zhu et al created the active pulling technique (APT), which has 5 steps: (1) anchoring the retrograde wire with a balloon or snare inside the antegrade guide catheter, (2) pulling back the retrograde guide catheter slightly into the aorta to avoid ostial injury, (3) securing a torque device at the entry port of the retrograde microcatheter to firmly couple it to the retrograde wire, (4) pulling the antegrade guide catheter outward to draw the retrograde microcatheter across the CTO segment, and (5) re-engaging the antegrade guide catheter and repeating the pulling maneuver until the retrograde microcatheter enters the antegrade guide.77 Among 80 patients, the APT group achieved higher device success rates, significantly shorter crossing times (5.3 ± 3.8 vs 18.6 ± 5.9 min, P < .001), and lower radiation dose.
Risk of retrograde CTO PCI. Retrograde CTO PCI has been associated with a higher complication rate than antegrade CTO PCI in multiple studies, which is at least in part related to the higher complexity of CTOs requiring retrograde crossing.6,78 The risk appears to be higher when using epicardial collaterals, especially ipsilateral collaterals.79
Some of the complications attributed to retrograde crossing may be due to antegrade crossing attempts. In an analysis from the EuroCTO registry, antegrade rescue techniques, which had historically been classified as retrograde, accounted for 6.7% of the cohort but carried disproportionately high complication rates and poor outcomes.78
Complications
Compared with non-CTO PCI, CTO PCI carries a higher risk of complications, especially perforation. A global consensus document on CTO PCI complications was recently published.80
Perforation
Large vessel perforations are usually treated with covered stents, whereas small vessel perforations are treated with coil or fat embolization. The Ringer balloon was recently approved and allows sealing of the coronary perforation while maintaining antegrade flow. Creating antegrade or retrograde dissection flaps can also be used to seal a perforation.81
Perforation in prior CABG patients may lead to loculated effusions and dry tamponade. Unlike classic tamponade, dry tamponade presents with hemodynamic instability without free-flowing pericardial effusion, instead resulting from compressive epicardial, intramyocardial, subepicardial, or dissecting intramyocardial hematomas.82 Diagnosis hinges on detecting hemodynamic derangement in the presence of a focal hematoma, often identified by transthoracic or transesophageal echocardiography, computed tomography, cardiac magnetic resonance imaging (MRI), or right heart catheterization. Epicardial hematomas appear echolucent, while intramyocardial hematomas are echodense. Delayed onset is common with hemodynamic compromise, often occurring hours or even days post-procedure.
Management differs based on stability. For unstable patients, treatment may involve fluids, vasopressors, covered stents or coils, MCS, and surgical or interventional radiology evacuation. For stable patients, immediate protamine administration and cessation of the procedure may allow for spontaneous sealing. Close intensive care unit monitoring is recommended for at least 24 hours, with delayed reattempt of PCI (up to 3 months) in cases of residual hematoma. In 1 study, overall mortality was 24% but was 100% for dissecting intramyocardial hematomas.
Donor vessel injury
In the PROGRESS-CTO registry, donor vessel injury occurred in 0.35% of cases (n = 43), most commonly as a result of guide catheter-induced dissection (53.5%), followed by thrombosis (20.9%) and forceful contrast injection (11.6%).83 Most injuries were treated with stenting alone (83.7%), but 16.3% of cases required additional urgent MCS, primarily for cardiac arrest or shock. Compared with uncomplicated cases, donor vessel injuries had significantly lower procedural success (69.8% vs 85.2%, P = .004) and higher MACE (23.3% vs 2.0%, P < .001), including 9.3% mortality and 11.6% periprocedural MI.
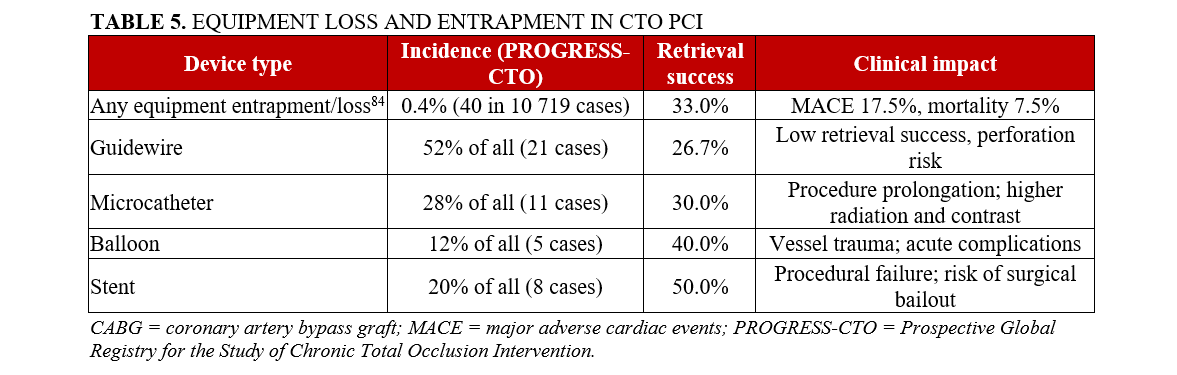
Equipment loss/entrapment
In the PROGRESS-CTO registry, the incidence of equipment loss or entrapment was 0.4%. Guidewire loss was the most common (n = 21): 4 simple entrapments, 7 fractures, and 10 combined entrapment-fractures.84 The causes included over-rotation, aggressive pulling, calcified lesions, or deformation during device manipulation. Wire retrieval was attempted in 71.4% of cases, but only 26.7% were successfully retrieved, and 3 cases resulted in death, including 2 requiring emergency CABG.
Microcatheter entrapment or fracture occurred in 11 cases, often because of over-rotation or advancement in heavily calcified vessels. Despite retrieval attempts in 90.9% of these cases, only 30% were successfully retrieved. Complications included pericardiocentesis, MI, bleeding, and coronary perforation.
Entrapment and loss of an atherectomy device is a rare but serious complications. Percutaneous retrieval is usually successful but surgical removal may be needed in some cases.85
There were 8 cases of stent loss, typically due to inadequate vessel preparation, tortuosity, aggressive device withdrawal, or jailing during retrograde attempts. Retrieval was attempted in 100%, yet only 50% were retrieved successfully. Half of these cases were also complicated by perforation, with 2 resulting in MI, and 1 by access site injury and bleeding.
Balloon-related complications (n = 5) included shaft fracture, rupture, or lithotripsy balloon entrapment. Retrieval was attempted in all but was successful in only 40%. There was a high rate of acute MI, emergency surgery, perforation, and vascular injury.
Equipment loss or entrapment was strongly associated with higher lesion complexity, including severe calcification, use of the retrograde approach, and worse procedural outcomes: lower technical and procedural success and higher MACE, including perforation, MI, and death. The incidence, mechanisms, clinical impact, and management strategies of equipment entrapment and loss during contemporary CTO PCI are summarized in Table 5.
Aortocoronary dissection
Aortocoronary dissections are more common in CTO PCI, especially in RCA CTOs. In a single-center study, all 6 patients with aortocoronary dissection remained stable and were managed conservatively with beta-blockers and antihypertensives, avoiding urgent surgery.86 Heavy calcification, retrograde injection, and forceful catheter manipulation were associated with higher dissection risk.
Emergency CABG
In PROGRESS-CTO, emergency CABG was needed in 0.12% (n = 17) of 14 512 CTO PCI procedures.87 The leading cause of emergent CABG was coronary perforation (71%), and in-hospital mortality was 35.3%.
Access site complications
Alexandrou et al reported 0.9% incidence of vascular access complications among 16 810 CTO PCIs.88 These complications were large hematomas (31.7%), small hematomas (24.1%), pseudoaneurysms (7.6%), and acute arterial closures (8.2%), with the remainder unspecified. Vascular access complications were more frequent in older patients, women, femoral access, and larger sheath sizes (≥ 7F), especially when urgent MCS was required. Increasing use of radial access and meticulous femoral access technique can help reduce the risk of access site complications.
Reported complications, predictors, and mitigation strategies in contemporary CTO PCI are summarized in Table 4.
CTO education, culture, and practice
Live-stream proctoring
Ungureanu et al evaluated the feasibility of remote proctoring during CTO PCI using a dedicated audio-video (AV) communication system (Sonar, Incathlab), enabling full procedural guidance by an off-site expert.89 The system consisted of 2 high-definition robotic cameras and a high-frequency audio interface, allowing the proctor to receive and transmit real-time imaging, audio, and procedural data. In this pilot study, all procedural steps were performed under the guidance of the remote proctor, who was able to follow precise angiographic details, such as collateral channels and wire tip movements, and intervene when needed, despite not being physically present. Technical success was achieved in 100% of the cases (9), including complex CTO lesions with high J-CTO scores, and no acute complications occurred during hospitalization. The AV setup did not interfere with the catheterization laboratory workflow or intrateam communication. This immersive remote‑support method could help new CTO operators and may boost safety in complex procedures, leading to wider adoption and larger studies.
Survey on CTO PCI operators
Simsek et al conducted an international survey involving 545 CTO PCI operators and 190 interventional cardiology fellows to evaluate global practices and attitudes regarding CTO interventions.90 Nearly half of the participants were based in NA, with a median CTO PCI volume of 20 cases annually, although many were high-volume PCI operators. Use of dual access varied widely, but only 40% used it in over 90% of cases. IVI after successful wiring was used frequently (> 90% of cases) by 38%, while 31% used it in less than 25% of cases. Most operators employed scoring tools such as J-CTO (81%) and PROGRESS-CTO (35%) scores, while a minority used other scores, such as CASTLE and RECHARGE. Perforation management skills varied, with higher-volume operators expressing greater self-reported comfort with advanced techniques such as coiling (median comfort score 7.3 vs 5.0 on a 0-10 scale) and fat embolization (median comfort score 5.0 vs 2.1). Despite high confidence ratings for physical and mental well-being, 31% of operators reported burnout and 14% reported depression, often related to administrative burden or perceived lack of institutional support.
Mindset, culture, and continuous improvement
Azzalini and Lombardi explored a frequently overlooked dimension of CTO PCI: the psychological mindset and institutional culture that shape operator performance and program success.91 While technical skills are essential, they argue that traits like emotional regulation, resilience, and “confident humility” are equally critical for safe, adaptive practice. They described the cognitive arc of CTO operators, from early overconfidence to mature awareness. They advocated for reflection, mindfulness, and psychological flexibility to avoid tunnel vision and impulsive risk-taking during complex cases.
The article also emphasizes the necessity of creating a supportive culture among interventional colleagues, catheterization laboratory staff, cardiothoracic surgeons, and hospital administrators. It emphasizes that sustainability of a CTO PCI program depends on strong relationships, transparent complication reporting, and a shared growth mindset. Establishing trust within the team, involving staff in case planning, and humanizing work relationships are essential strategies to mitigate burnout and elevate morale.
Ongoing studies
PCI vs optimal medical therapy (OMT) for arrhythmias
The CTO-ARRHYTHMIA is a substudy of the NOBLE-CTO (Nordic-Baltic Randomized Registry Study for Evaluation of PCI in CTO) trial (NCT03392415) that is assessing the impact of CTO PCI vs optimal medical therapy (OMT) on arrhythmia burden.92 Patients with CTO lesions suitable for PCI but without implantable cardioverter defibrillator indications (ie, LVEF ≥ 35%) are randomized 1:1 to OMT alone or OMT plus PCI. All participants receive implantable loop recorders to detect arrhythmias, with the primary endpoint being the incidence of atrial fibrillation or 8 runs or more of ventricular premature beats over 6 months. The study combines continuous rhythm monitoring with cardiac MRI and echocardiography to investigate whether CTO PCI reduces arrhythmias linked to chronic ischemia.
ISCHEMIA-CTO trial
The ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) - CTO trial (NCT03563417) is a randomized, multicenter study evaluating whether CTO PCI improves outcomes beyond OMT in patients with ischemia and a CTO lesion greater than or equal to 2.5 mm.93 After 3 months of medical therapy and treatment of non-CTO lesions, patients are stratified by symptoms and ischemia burden. Low-symptom patients (Cohort A) are randomized to assess major clinical events, while symptomatic patients (Cohort B) are randomized to evaluate quality of life at 6 months.
ORBITA-CTO Trial
The Comparison of the Impact of CTO PCI Versus Placebo on Angina in Patients With Background Optimal Medical Therapy - a Pilot Study (ORBITA-CTO pilot study, (NCT05142215) is a double-blind, placebo-controlled study of CTO PCI vs no CTO PCI.94 The co-primary outcomes are feasibility (blinding) in this cohort and angina symptom score using an ordinal clinical outcome scale for angina. Secondary outcomes include changes in quality-of-life measures, Seattle Angina Questionnaire (SAQ), peak VO2, and anaerobic threshold on the cardiopulmonary exercise test.
Conclusions
Our comprehensive review of recent CTO PCI publications provides a contemporary overview of outcomes, techniques, prediction tools, and complications associated with CTO PCI. Large registries and multicenter analyses demonstrate that high technical and procedural success are achievable even in complex CTOs at experienced centers, albeit with a persistent risk of complications that underscores the importance of careful patient selection and procedural planning.
Advances in outcome prediction, including angiographic, CTA-based, and retrograde-specific scoring systems, have improved case stratification and strategy selection, while growing data support the role of IVI in procedural optimization and long-term vessel patency. Contemporary analyses of complex clinical subgroups highlight both the feasibility and heterogeneity of outcomes in higher-risk populations, reinforcing the need for individualized decision making and shared patient-physician discussions.
Although improvements in hard clinical endpoints such as mortality and myocardial infarction remain modest in some contemporary analyses, successful CTO PCI has consistently been associated with symptomatic relief, improved quality of life, and reduction in ischemic burden, which remain the primary patient-centered goals of the procedure. Ongoing studies continue to refine technique selection, imaging integration, radiation management, and patient-centered outcomes, emphasizing that incorporation of these recent findings into daily clinical practice has the potential to further improve the safety, effectiveness, and consistency of CTO PCI.
Affiliations and Disclosures
Semih Ceylan1; Deniz Mutlu, MD2,3; Eleni Kladou, MD3; Nick Williford, MD3; Sandeep Jalli, MD4; Ahmed Al-Ogaili, MD5; Masahisa Yamane, MD6; Khaldoon Alaswad, MD7; Allison Hall, MD8; Rhian Davies, DO9; James W. Choi, MD10; Andrea Gagnor, MD11; Roberto Garbo, MD11; Omer Goktekin, MD12; Sevket Gorgulu, MD13; Jaikirshan J. Khatri, MD14; William Nicholson, MD15; Stephane Rinfret, MD15; Wissam Jaber, MD15; Mohaned Egred, MD16; Anastasios Milkas, MD, PhD17; Niccolò Ciardetti, MD18; Carlo Di Mario, MD, PhD18,19; Kambis Mashayekhi, MD, PhD20; Alexandre Avran, MD21; Gregor Leibundgut, MD22; Yiannis S. Chatzizisis, MD, PhD23; Gerald S. Werner, MD, PhD24; Claudiu Ungureanu, MD25; Yader Sandoval, MD3; Emmanouil S. Brilakis, MD, PhD3
From the 1Bahcesehir University School of Medicine, Istanbul, Turkey; 2SUNY Downstate Health Sciences University, Department of Cardiology, Brooklyn, New York; 3Minneapolis Heart Institute and Minneapolis Heart Institute Foundation, Abbott Northwestern Hospital, Minneapolis, Minnesota; 4Prisma Health, Columbia, South Carolina; 5Division of Cardiovascular Medicine, Penn Presbyterian Medical Center, Perelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania; 6Cardiovascular Division, Saitama-Sekishinkai Hospital, Saitama, Japan; 7Department of Cardiology, Henry Ford Hospital, Detroit, Michigan; 8Eastern Health/Memorial University of Newfoundland, St.John’s Newfoundland, Canada; 9Wellspan York Hospital, York, Pennsylvania; 10Texas Health Presbyterian Hospital, Dallas, Texas; 11Maria Vittoria Hospital, Torino, Italy; 12Memorial Bahcelievler Hospital, Istanbul, Turkey; 13Biruni University Medical School, Istanbul, Turkey; 14Weill Cornell Medical Center, New York, New York; 15Emory University Hospital Midtown, Atlanta, Georgia; 16Department of Cardiology, Freeman Hospital, Newcastle upon Tyne, United Kingdom; 17Athens Naval and Veterans Hospital, Athens, Greece; 18Department of Clinical & Experimental Medicine, University of Florence, Careggi Hospital, Florence, Italy; 19Royal Brompton Hospital, London, United Kingdom; 20University Heart Center Freiburg, Bad Krozingen, Germany; 21Valenciennes Hospital, Valenciennes, France; 22Department of Cardiology, University Hospital Basel, Basel, Switzerland; 23Division of Cardiovascular Medicine, Department of Medicine, University of Miami, Florida; 24University Heart Center, Goethe University, Frankfurt/Main, Germany; 25Department of Cardiology, CHU Helora Jolimont Hospital, La Louviere, Belgium.
Disclosures: Dr Alaswad receives consulting fees from Boston Scientific. Dr Hall receives speaker fees and/or honoraria from Ates Medical, Shockwave Medical, Sis Medical, and Teleflex. Dr Davies receives speaking fees, honoraria, and/or serves on advisory boards for Asahi, Boston Scientific, Medtronic, Johnson & Johnson MedTech, Rampart, Terumo, and Teleflex. Dr Goktekin reports proctorship and consultancy fees from Boston Scientific, Asahi, Terumo, Medtronic, and Abbott. Dr Egred is a proctor for CTO, rotablation, ELCA, IVI, and CHIP PCI, and has receieved honoraria and consultation and speaker fees from Philips, Abbott Vascular, Boston Scientific, Vascular Perspectives, Biosensors, Biotronik, and AstraZeneca. Dr Di Mario receives research or educational grants from Abbott, Amgen, ASAHİ INTECC, AstraZeneca, Boston Scientific, Cardinal Health, Behring, Chiesi, Daiichi Sankyo, Edwards, Medtronic, Menarini, Pfizer, Sanofi, Shockwave, Teleflex, and Philips/Volcano. Dr Mashayekhi reports consulting/speaker/proctoring honoraria from Abbott Vascular, Abiomed, ASAHİ INTECC, AstraZeneca, Biotronik, Boston Scientific, Cardinal Health, Daiichi Sankyo, Medtronic, Orbus Neich, Shockwave Medical, Teleflex, and Terumo. Dr Sandoval serves as a consultant, advisory board member, and/or speaker for Abbott, CathWorks, Cleerly, GE Healthcare, HeartFlow, Medtronic, Philips, Roche Diagnostics, and Zoll; receives a research grant from Cleerly; owns Systole LLC; serves as an associate editor for JACC Advances; and holds patent 20210401347. Dr Brilakis receives consulting/speaker honoraria from Abbott Vascular, the American Heart Association (associate editor, Circulation), Boston Scientific, Cardiovascular Innovations Foundation (Board of Directors), Cordis, Elsevier, GE Healthcare, Heartflow, IMDS, Medtronic, Recor Medical, SHockwave, SIS Medical, Teleflex, and Terumo; receives research support from AngioWave, Boston Scientific, GE Healthcare; is owner of Hippocrates LLC and Systole LLC; and a shareholder of Cleerly Health, LifeLens Technologies, Inc., MHI Ventures, Stallion Medical, and TrueVue, Inc.The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Emmanouil S. Brilakis, MD, PhD, Minneapolis Heart Institute, 920 E 28th Street #300, Minneapolis, MN 55407, USA. Email: esbrilakis@gmail.com; X: @esbrilakis
References
1. Strepkos D, Alexandrou M, Mutlu D, et al; PROGRESS-CTO Investigators. Impact of the COVID-19 pandemic on CTO PCI: analysis from the PROGRESS-CTO registry. Hellenic J Cardiol. 2025;86:142-144. doi:10.1016/j.hjc.2024.11.005
2. Alexandrou M, Rempakos A, Mutlu D, et al. Geographic diversity in chronic total occlusion percutaneous coronary intervention: insights from the PROGRESS-CTO registry. J Invasive Cardiol. 2024;36(9). doi:10.25270/jic/24.00056
3. Allana SS, Rempakos A, Alexandrou M, et al. Racial disparities in chronic total occlusion percutaneous coronary interventions: insights from the PROGRESS-CTO registry. J Invasive Cardiol. 2024;36(2). doi:10.25270/jic/23.00274
4. Mohebbi B, Sadeghipour P, Zolfaghari R, et al. Outcomes of chronic total occlusion percutaneous coronary intervention from the RAIAN (RAjaie - Iran) registry. Indian Heart J. 2023;75(6):429-435. doi:10.1016/j.ihj.2023.10.002
5. Vadalà G, Galassi AR, Werner GS, et al; Euro Cto Investigators OBOT. Contemporary outcomes of chronic total occlusion percutaneous coronary intervention in Europe: the ERCTO registry. EuroIntervention. 2024;20(3):e185-e197. doi:10.4244/EIJ-D-23-00490
6. Verreault-Julien L, Jahan I, Dendukuri N, et al. Outcomes following antegrade-only versus retrograde chronic total occlusion percutaneous coronary intervention: insights from the CCTOP registry. J Invasive Cardiol. 2025;37(1). doi:10.25270/jic/24.00130
7. Gorgulu S, Kostantinis S, ElGuindy AM, et al Contemporary ın-hospital outcomes of chronic total occlusion percutaneous coronary ınterventions: insights from the MENATA (Middle East, North Africa, Turkey, and Asia) chapter of the PROGRESS-CTO registry. Am J Cardiol. 2023;206:221-229. doi:10.1016/j.amjcard.2023.08.103
8. van Veelen A, Coerkamp CF, Somsen YBO, et al; EXPLORE investigators. Ten-year outcome of recanalization or medical therapy for concomitant chronic total occlusion after myocardial ınfarction. J Am Heart Assoc. 2024;13(10):e033556. doi:10.1161/JAHA.123.033556
9. Otsuki H, Takahashi K, Zimmermann FM, et al; FAME 3 Trial Investigators. Impact of a chronic total occlusion on outcomes after ffr-guided pcı or coronary bypass surgery: a FAME 3 substudy. Circ Cardiovasc Interv. 2024;17(11):e014300. doi:10.1161/CIRCINTERVENTIONS.124.014300
10. Lombardi M, Holck EN, Jerónimo A, et al. Time-dependent changes in the coronary circulation triggered by CTO revascularization: ınsights from ıntracoronary physiology. J Soc Cardiovasc Angiogr Interv. 2025;4(1):102452. doi:10.1016/j.jscai.2024.102452
11. Ge X, Wu H, Zang Z, Xie J. Application of checklist-based nursing care process in patients undergoing intervention for coronary chronic total occlusions: a quasi-randomized study. BMC Cardiovasc Disord. 2023;23(1):591. doi:10.1186/s12872-023-03627-
12. Meijers TA, Aminian A, Valgimigli M, et al. Vascular access in percutaneous coronary ıntervention of chronic total occlusions: a state-of-the-art review. Circ Cardiovasc Interv. 2023;16(8):e013009. doi:10.1161/CIRCINTERVENTIONS.123.013009
13. Al-Ogaili A, Mutlu D, Alexandrou M, et al. Guide catheter extension use in chronic total occlusion percutaneous coronary intervention: insights from the PROGRESS CTO registry. J Invasive Cardiol. 2024;36(12). doi:10.25270/jic/24.00117
14. Somsen YBO, Wilgenhof A, Hoek R, et al. Same-day discharge after large-bore access in percutaneous coronary intervention of chronic total coronary occlusions. EuroIntervention. 2024;20(10):e643-e655. doi:10.4244/EIJ-D-23-00838
15. Simsek B, Rempakos A, Kostantinis S, et al. Activated clotting time and outcomes of chronic total occlusion percutaneous coronary intervention: insights from the PROGRESS-CTO registry. J Invasive Cardiol. 2023;35(12). doi:10.25270/jic/23.00170
16. Mutlu D, Rempakos A, Alexandrou M, et al. Radiation dose during contemporary percutaneous coronary interventions for chronic total occlusion: insights from the PROGRESS-CTO registry. J Invasive Cardiol. 2024;36(4). doi:10.25270/jic/23.00281
17. Werner GS, Avran A, Boudou N, et al. Improvement of radiation management in percutaneous ınterventions of chronic total occlusions in a multicenter registry. JACC Cardiovasc Interv. 2025;18(4):425-435. doi:10.1016/j.jcin.2024.11.009
18. Will M, Schwarz K, Weiss T, et al. The impact of concomitant chronic total occlusion on clinical outcomes in patients undergoing transcatheter aortic valve replacement: a large single-center analysis. Front Cardiovasc Med. 2024;11:1338253. doi:10.3389/fcvm.2024.1338253
19. Karacsonyi J, Kostantinis S, Simsek B, et al. Intravascular ımaging use in percutaneous coronary ınterventions of chronic total occlusions. J Invasive Cardiol. 2023;35(5):E265-E268. doi:10.25270/jic/23.00002
20. Poletti E, Kearney KE, Chung CJ, et al. Impact of systematic intravascular imaging on the outcomes of complex and higher-risk percutaneous coronary intervention. Cardiovasc Revasc Med. 2025;81:70-77. doi:10.1016/j.carrev.2025.04.007
21. Sawayama Y, Sasaki K, Taninobu N, et al. The effect of ıntravascular ımaging-guided percutaneous coronary ıntervention on coronary artery perforation. JACC Asia. 2024;5(1):46-55. doi:10.1016/j.jacasi.2024.09.004
22. Hong D, Kim SM, Lee SY, et al; RENOVATE-COMPLEX-PCI Investigators. Prognostic ımpact of ıntravascular ımaging-guided percutaneous coronary ıntervention in chronic total occlusion. Circulation. 2023;148(11):903-905. doi:10.1161/CIRCULATIONAHA.123.065876
23. Gomes WF, Zerlotto DS, Viana P, et al. Intravascular ımaging ımproves clinical outcomes of percutaneous coronary ıntervention for chronic total occlusions: a meta-analysis of randomized controlled trials. Am J Cardiol. 2025;245:62-70. doi:10.1016/j.amjcard.2025.03.011
24. Stone GW, Christiansen EH, Ali ZA, et al. Intravascular imaging-guided coronary drug-eluting stent implantation: an updated network meta-analysis. Lancet. 2024;403(10429):824-837. doi:10.1016/S0140-6736(23)02454-6
25. Christopoulos G, Wyman RM, Alaswad K, et al. Clinical utility of the japan-chronic total occlusion score in coronary chronic total occlusion ınterventions: results from a multicenter registry. Circ Cardiovasc Interv. 2015;8(7):e002171. doi:10.1161/CIRCINTERVENTIONS.114.002171
26. Niizeki T, Tsuchikane E, Konta T, et al. New angiographic difficulty score for first-attempt chronic total occlusion in the 3 major coronary arteries. JACC Cardiovasc Interv. 2023;16(20):2542-2551. doi:10.1016/j.jcin.2023.08.046
27. Mutlu D, Simsek B, Rempakos A, et al. Validation of the coronary artery specific chronic total occlusion percutaneous coronary intervention angiographic difficulty scores in the PROGRESS-CTO registry. Cardiovasc Revasc Med. 2025;76:66-72. doi:10.1016/j.carrev.2024.10.007
28. Kumar S, Opolski MP, Ahn JM, et al. The role of coronary computed tomography angiography in chronic total occlusion percutaneous coronary ıntervention. JACC Cardiovasc Interv. 2026;19(1):1-14. doi:10.1016/j.jcin.2025.10.055
29. Li J, Wang R, Tesche C, et al. CT angiography-derived RECHARGE score predicts successful percutaneous coronary ıntervention in patients with chronic total occlusion. Korean J Radiol. 2021;22(5):697-705. doi:10.3348/kjr.2020.0732
30. Le Y, Wang R, Xing H, Chen H, Song X, He Y. Pericoronary adipose tissue attenuation predicts outcome of percutaneous intervention for chronic total occlusion. Clin Radiol. 2024;79(2):e232-e238. doi:10.1016/j.crad.2023.10.012
31. Xing H, Tang Z, Zhang L, et al. Radiomics analysis of CTO plaques for predicting successful guidewire crossing within 30 minutes of PCI. Circ Cardiovasc Imaging. 2024;17(8):e016117. doi:10.1161/CIRCIMAGING.123.016117
32. Zhou Z, Gao Y, Zhang W, et al. Deep learning-based prediction of percutaneous recanalization in chronic total occlusion using coronary CT angiography. Radiology. 2023;309(2):e231149. doi:10.1148/radiol.231149
33. Rempakos A, Alexandrou M, Mutlu D, et al. Predicting successful chronic total occlusion crossing with primary antegrade wiring using machine learning. JACC Cardiovasc Interv. 2024;17(14):1707-1716. doi:10.1016/j.jcin.2024.04.043
34. Taniguchi Y, Sakakura K, Jinnouchi H, et al. Determinants of successful parallel wire technique in percutaneous coronary intervention to coronary chronic total occlusion. Cardiovasc Revasc Med. 2024;59:48-52. doi:10.1016/j.carrev.2023.08.010
35. Tanaka H, Tsuchikane E, Kishi K, Okada H, Oikawa Y, Ito Y, Muramatsu T, Yoshikawa R, Kawasaki T, Okamura A, Sumitsuji S, Muto M, Katoh O. Retrograde coronary chronic total occlusion ıntervention (jr-cto) score: from the Japanese CTO-PCI Expert registry. JACC Cardiovasc Interv. 2024;17(11):1374-1384. doi:10.1016/j.jcin.2024.03.023
36. Nagamatsu W, Tsuchikane E, Oikawa Y, et al. Successful guidewire crossing via collateral channel at retrograde percutaneous coronary intervention for chronic total occlusion: the J-Channel score. EuroIntervention. 2020;15(18):e1624-e1632. doi:10.4244/EIJ-D-18-00993
37. Mutlu D, Rempakos A, Alexandrou M, et al. Validation of the J-Channel Score for retrograde channel crossing in the PROGRESS-CTO registry. J Invasive Cardiol. 2024;36(5). doi:10.25270/jic/24.00006
38. Somsen YBO, de Winter RW, Giunta R, et al. Collateral grading systems in retrograde percutaneous coronary intervention of chronic total occlusions. Catheter Cardiovasc Interv. 2023;102(5):844-856. doi:10.1002/ccd.30812
39. Chang HY, Huang CC, Hung CS, et al. Accurate prediction of retrograde collateral channel crossing in coronary artery chronic total occlusion ıntervention. Am J Cardiol. 2024;210:93-99. doi:10.1016/j.amjcard.2023.10.027
40. Karacsonyi J, Stanberry L, Simsek B, et al. Development of a novel score to predict urgent mechanical circulatory support in chronic total occlusion percutaneous coronary ıntervention. Am J Cardiol. 2023;202:111-118. doi:10.1016/j.amjcard.2023.06.051
41. Hirai K, Kawasaki T, Kishi K, et al. Determinants of one-year outcome after successful percutaneous coronary ıntervention for chronic total occlusion; ınsight from Japanese CTO-PCI Expert registry. Am J Cardiol. 2024;225:108-117. doi:10.1016/j.amjcard.2024.06.016
42. Alexandrou M, Kostantinis S, Rempakos A, et al. Outcomes of chronic total occlusion percutaneous coronary ınterventions in patients with previous coronary artery bypass graft surgery. Am J Cardiol. 2023;205:40-49. doi:10.1016/j.amjcard.2023.07.112
43. Poletti E, Dens J, Egred M, et al. Impact of postprocedural graft flow on outcomes following chronic total occlusion ıntervention in postcoronary artery bypass graft patients: a detailed angiographic analysis. Am J Cardiol. 2024;226:24-33. doi:10.1016/j.amjcard.2024.06.015
44. Sammour YM, Bou Chaaya RG, Kharsa C, et al. Assessment of left ventricular function after percutaneous coronary ıntervention for chronic total occlusion. J Soc Cardiovasc Angiogr Interv. 2025;4(1):102460. doi:10.1016/j.jscai.2024.102460
45. Gallo I, Azzalini L, González-Manzanares R, et al. Mechanical circulatory support with ımpella in high-risk patients with chronic total occlusion and complex multivessel disease. Catheter Cardiovasc Interv. 2025;105(4):883-890. doi:10.1002/ccd.31392
46. Ser OS, Mutlu D, Alexandrou M, et al. Obstructive sleep apnea syndrome in chronic total occlusion percutaneous coronary intervention; insights from the PROGRESS-CTO registry. Hellenic J Cardiol. 2025;85:38-47. doi:10.1016/j.hjc.2025.03.010
47. Strepkos D, Alexandrou M, Mutlu D, et al. Outcomes of left main chronic total occlusion percutaneous coronary ınterventions. Catheter Cardiovasc Interv. 2025;105(1):23-31. doi:10.1002/ccd.31289
48. Allana SS, Kostantinis S, Simsek B, et al. Lesion complexity and procedural outcomes associated with ostial chronic total occlusions: insights from the PROGRESS-CTO registry. J Invasive Cardiol. 2023;35(12). doi:10.25270/jic/23.00034
49. Moroni A, Ayoub M, Gorgulu S, et al. Impact of bifurcation ınvolvement and location in chronic total occlusion percutaneous coronary ıntervention: ınsights from the EuroCTO registry. Am J Cardiol. 2024;223:132-146. doi:10.1016/j.amjcard.2024.05.029
50. Strepkos D, Rempakos A, Alexandrou M, et al. Association of proximal vessel tortuosity with technical success and clinical outcomes: analysis from the progress-CTO registry. Catheter Cardiovasc Interv. 2025;105(1):1-10. doi:10.1002/ccd.31338
51. Kostantinis S, Rempakos A, Simsek B, et al. Impact of calcium on the procedural techniques and outcomes of chronic total occlusion percutaneous coronary intervention. Int J Cardiol. 2023;390:131254. doi:10.1016/j.ijcard.2023.131254
52. Panuccio G, Werner GS, De Rosa S, et al. Full-moon coronary calcification as detected with computed tomography angiography in chronic total occlusion percutaneous coronary ıntervention. Am J Cardiol. 2024;222:149-156. doi:10.1016/j.amjcard.2024.05.008
53. Yamamoto K, Sugizaki Y, Karmpaliotis D, et al. Presence and relevance of myocardial vridge in LAD-PCI of CTO and non-CTO lesions. JACC Cardiovasc Interv. 2024;17(4):491-501. doi:10.1016/j.jcin.2023.12.017
54. Werner GS, Rathore S, Avran A, et al; ERCTO contributors. Parallel wire approach for recanalisation of chronic total coronary occlusions in a large contemporary multi-center registry. Catheter Cardiovasc Interv. 2025;105(1):32-42. doi:10.1002/ccd.31315
55. Rempakos A, Alexandrou M, Simsek B, et al. Trends and outcomes of antegrade dissection and re-entry in chronic total occlusion percutaneous coronary ıntervention. JACC Cardiovasc Interv. 2023;16(22):2736-2747. doi:10.1016/j.jcin.2023.09.021
56. Garbo R, Iannaccone M, Bruno F, Arioti M. Intravascular ultrasound-guided STAR 2.0: A new technique for chronic total occlusion recanalization. Catheter Cardiovasc Interv. 2024;103(1):80-88. doi:10.1002/ccd.30920
57. Hirai T, Kearney K, Azzalini L, et al; STAR Study Group. Optimal timing of staged percutaneous coronary intervention after subintimal tracking and re-entry: rationale and design of the subintimal tracking and re-entry with deferred stenting study. Catheter Cardiovasc Interv. 2024;104(3):444-450. doi:10.1002/ccd.31161
58. Azzalini L, Kearney K, Salisbury AC, et al; STAR Study Group. Early vs late staged PCI after subintimal tracking and re-entry for chronic total occlusions: a randomized trial. J Am Coll Cardiol. 2026;87(3):286-293. doi:10.1016/j.jacc.2025.09.1598
59. Chi KW, Wu EB. Extraplaque blood withdrawal stenting: a miraculous cure for subintimal hematoma. J Invasive Cardiol. 2024;36(2). doi:10.25270/jic/23.00175
60. Gilbert P, Patel T, Gupta A. Continuous mechanical suction use during chronic total occlusion revascularization. J Soc Cardiovasc Angiogr Interv. 2024;3(12):102392. doi:10.1016/j.jscai.2024.102392
61. Alexandrou M, Rempakos A, Al Ogaili A, et al. Balloon-assisted subintimal entry (BASE) in chronic total occlusion percutaneous coronary interventions. Catheter Cardiovasc Interv. 2023;102(5):834-843. doi:10.1002/ccd.30830
62. Allana SS, Chidester J, Koshy T, Al-Azizi K, Brilakis ES. Double-side balloon-assisted subintimal entry: solving proximal cap ambiguity in CTO PCI. JACC Cardiovasc Interv. 2025;18(7):936-938. doi:10.1016/j.jcin.2024.12.019
63. Rejeki VG, Tadano Y, Fujita T, Kuramitsu S. Balloon-assisted subintimal entry-facilitated tip detection-antegrade dissection reentry technique: a novel antegrade approach for chronic total occlusion. Catheter Cardiovasc Interv. 2025;105(7):1664-1667. doi:10.1002/ccd.31506.
64. Kitani S, Tsuchikane E, Yamaki M, Igarashi Y. First successful implementation of subintimal transcatheter withdrawal technique in intravascular ultrasound-guided tip detection antegrade dissection and reentry: a case report. Eur Heart J Case Rep. 2023;7(12):ytad580. doi:10.1093/ehjcr/ytad580
65. Tanaka K, Okamura A, Yoshikawa R, et al. Tip detection-antegrade dissection and re-entry with new puncture wire in CTO intervention: revolution through 3D-wiring. JACC Asia. 2024;4(5):359-372. doi:10.1016/j.jacasi.2023.11.017
66. Alexandrou M, Rempakos A, Al Ogaili A, et al. Use of the Carlino technique in chronic total occlusion percutaneous coronary ıntervention. Am J Cardiol. 2023;207:305-313. doi:10.1016/j.amjcard.2023.08.155
67. Carlino M, Nascimbene A, Brilakis ES, et al. HydroDynamic contrast Recanalization (HDR): description of a new crossing technique for coronary chronic total occlusions. Catheter Cardiovasc Interv. 2024;104(5):918-927. doi:10.1002/ccd.31243
68. Wu EB, Nagamatsu W, Okamura A. Ten tips and tricks for successful distal true lumen wiring from three-dimensional wiring experts. J Invasive Cardiol. 2024;36(10). doi:10.25270/jic/24.00096
69. Allana SS, Kostantinis S, Rempakos A, et al. The retrograde approach to chronic total occlusion percutaneous coronary ınterventions: technical analysis and procedural outcomes. JACC Cardiovasc Interv. 2023;16(22):2748-2762. doi:10.1016/j.jcin.2023.08.031
70. Ng P, Mercado N, Lombardi WL, Azzalini L. Reverse CART facilitated by a novel perfusion balloon in chronic total occlusion percutaneous coronary intervention. EuroIntervention. 2025;21(3):e193-e194. doi:10.4244/EIJ-D-24-00895
71. Moscardelli S, Kearney KE, Lombardi WL, Azzalini L. Controlled antegrade and retrograde subintimal tracking (CART) for recanalisation of chronic total occlusions. EuroIntervention. 2024;20(9):571-578. doi:10.4244/EIJ-D-23-01082
72. Kane J, Kearney KE, Lombardi WL, Azzalini L. Electrocautery-assisted re-entry to resolve bilateral aorto-ostial chronic total occlusions due to leaflet obstruction following transcatheter aortic valve replacement. Catheter Cardiovasc Interv. 2023;102(3):489-494. doi:10.1002/ccd.30781
73. Moscardelli S, Condos G, Lombardi WL, Azzalini L. Intracoronary snaring of the retrograde guidewire: A novel method to solve compartment mismatch in complex retrograde CTO PCI. Catheter Cardiovasc Interv. 2024;103(3):435-442. doi:10.1002/ccd.30962
74. Ungureanu C, Avran A, Brilakis ES, et al. Comprehensive overview of retrograde-antegrade connection techniques without externalization in chronic total occlusion PCI: the portal techniques. Catheter Cardiovasc Interv. 2025;105(1):11-22. doi:10.1002/ccd.31346
75. Ungureanu C, Jossart A, Cocoi M. A new technique of "tip-ın" for retrograde chronic total occlusion revascularization: the modified tip-ın technique. JACC Cardiovasc Interv. 2024;17(1):91-93. doi:10.1016/j.jcin.2023.10.037
76. Tan Y, Zhang T, Zhao L. A novel technique of “bite ın” for retrograde chronic total occlusion revascularization. J Am Coll Cardiol Intv. 2024 Oct;17(19):2301-2303.
77. Zhu H, Cai X, Zhan Y, Hong L. The active pulling technique to solve microcatheter-uncrossable lesions in retrograde chronic total occlusion percutaneous coronary intervention. Int J Cardiovasc Imaging. 2024;40(5):1019-1027. doi:10.1007/s10554-024-03068-0
78. Vadalà G, Galassi AR, Werner GS, et al. Euro Cto Investigators. Contemporary outcomes of chronic total occlusion percutaneous coronary intervention in Europe: the ERCTO registry. EuroIntervention. 2024;20(3):e185-e197. doi:10.4244/EIJ-D-23-00490
79. Al-Ogaili A, Alexandrou M, Rempakos A, et al. Retrograde chronic total occlusion percutaneous coronary intervention via ipsilateral collaterals. Catheter Cardiovasc Interv. 2024;103(6):863-872. doi:10.1002/ccd.31019
80. Wu EB, Kalyanasundaram A, Brilakis ES, Mashayekhi K, Tsuchikane E; CTO Global Consensus Group. Global consensus recommendations on ımproving the safety of chronic total occlusion ınterventions. Heart Lung Circ. 2024;33(7):915-931. doi:10.1016/j.hlc.2023.11.030
81. Xenogiannis I, Tajti P, Nicholas Burke M, Brilakis ES. An alternative treatment strategy for large vessel coronary perforations. Catheter Cardiovasc Interv. 2019;93(4):635-638. doi:10.1002/ccd.28034
82. Tiwana J, Kearney KE, Lombardi WL, Azzalini L. Challenges in the diagnosis and management of dry tamponade. Catheter Cardiovasc Interv. 2024;104(6):1228-1240. doi:10.1002/ccd.31241
83. Kostantinis S, Rempakos A, Simsek B, et al. Incidence, mechanisms, treatment, and outcomes of donor vessel injury during percutaneous coronary interventions for chronic total occlusion. Catheter Cardiovasc Interv. 2023;102(4):585-593. doi:10.1002/ccd.30798
84. Alexandrou M, Rempakos A, Mutlu D, et al. Equipment entrapment/loss during chronic total occlusion percutaneous coronary intervention. J Invasive Cardiol. 2024;36(4). doi:10.25270/jic/23.00266
85. Leibundgut G, Achim A, Weilenmann D, et al. Management of lost atherectomy devices in the coronary arteries. Catheter Cardiovasc Interv. 2025;106(3):1766-1780. doi:10.1002/ccd.31734
86. Safdar A, Young L, Khatri J. Management of iatrogenic ascending aortic dissection after percutaneous intervention of chronic total occlusion. Catheter Cardiovasc Interv. 2024;104(7):1442-1446. doi:10.1002/ccd.31262
87. Mutlu D, Rempakos A, Alexandrou M, et al. Emergency coronary artery bypass surgery after chronic total occlusion percutaneous coronary intervention: insights from the PROGRESS-CTO registry. Int J Cardiol. 2024;405:131931. doi:10.1016/j.ijcard.2024.131931
88. Alexandrou M, Strepkos D, Carvalho PEP, et al. Vascular access-site complications in chronic total occlusion percutaneous coronary ıntervention. Catheter Cardiovasc Interv. 2025;105(7):1578-1587. doi:10.1002/ccd.31501
89. Ungureanu C, Yamane M, Kayaert P, et al. The safety and feasibility of live-stream proctoring for CTO procedures. J Invasive Cardiol. 2023;35(10). doi:10.25270/jic/23.00076
90. Simsek B, Rempakos A, Kostantinis S, et al. International survey of chronic total occlusion percutaneous coronary intervention operators. Catheter Cardiovasc Interv. 2024;103(1):12-19. doi:10.1002/ccd.30914
91. Azzalini L, Lombardi WL. Chronic total occlusion percutaneous coronary ıntervention: mindset, culture, and continuous ımprovement. Am J Cardiol. 2024;222:141-148. doi:10.1016/j.amjcard.2024.04.048
92. Christensen MK, Eftekhari A, Raungaard B, et al. Impact of percutaneous ıntervention compared to pharmaceutical therapy on complex arrhythmias in patients with chronic total coronary occlusion. rationale and design of the CTO-ARRHYTHMIA study. Cardiovasc Revasc Med. 2023;54:69-72. doi:10.1016/j.carrev.2023.04.001
93. Råmunddal T, Holck EN, Karim S, et al. International randomized trial on the effect of revascularization or optimal medical therapy of chronic total coronary occlusions with myocardial ischemia - ISCHEMIA-CTO trial - rationale and design. Am Heart J. 2023;257:41-50. doi:10.1016/j.ahj.2022.11.016
94. Khan S, Fawaz S, Simpson R, et al. The challenges of a randomised placebo-controlled trial of CTO PCI vs. placebo with optimal medical therapy: the ORBITA-CTO pilot study design and protocol. Front Cardiovasc Med. 2023;10:1172763. doi:10.3389/fcvm.2023.1172763