Boston Scientific Presents Two Late-Breaking Trials at ACC 2026 Highlighting New Evidence in Stroke Prevention for AFib and Treatment of Intermediate-Risk Pulmonary Embolism
At the American College of Cardiology (ACC) 2026 Scientific Session, Boston Scientific presented results from two late-breaking randomized controlled trials evaluating device-based approaches in cardiovascular care: CHAMPION-AF, studying the WATCHMAN FLX™ Left Atrial Appendage Closure Device as a first-line strategy for stroke risk reduction in patients with non-valvular atrial fibrillation (AFib), and HI-PEITHO, assessing the EKOS™ Endovascular System for patients with intermediate-risk pulmonary embolism (PE). Results from both trials were published in The New England Journal of Medicine.
CHAMPION-AF: WATCHMAN FLX™ vs Oral Anticoagulants in Non-Valvular AFib
CHAMPION-AF is described as the largest global randomized controlled trial to evaluate the WATCHMAN FLX device as a first-line therapy versus blood thinners in patients with non-valvular AFib, enrolling 3,000 patients worldwide. Three-year data showed the study met all endpoints, with WATCHMAN FLX demonstrating statistically superior protection from bleeding (P<0.0001) and similar protection against stroke compared with oral anticoagulants.
WATCHMAN FLX is a minimally invasive, one-time implant designed to seal the left atrial appendage, which is cited as the source of more than 90% of stroke-causing blood clots in patients with AFib. Boston Scientific noted that non-adherence may leave up to 40% of patients taking blood thinners unprotected, and said these findings may inform first-line stroke risk reduction strategies for patients concerned about bleeding risk or long-term medication adherence.
Investigators and authors:
- Saibal Kar, MD (HCA Healthcare), principal investigator, ACC presenter, and NEJM co-author
- Shephal Doshi, MD (Pacific Heart Institute), principal investigator and lead author of the NEJM manuscript
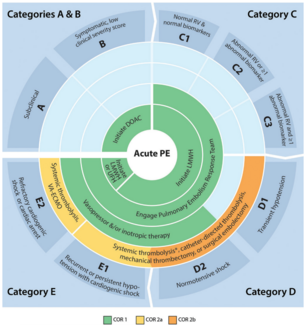
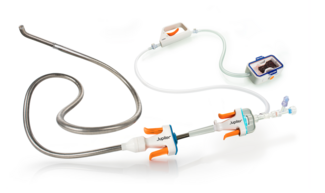
HI-PEITHO: EKOS™ Endovascular System Plus Anticoagulation vs Anticoagulation Alone in Intermediate-Risk PE
Also published in The New England Journal of Medicine, HI-PEITHO (n=544 across the United States and Europe) is described as the largest published global randomized controlled trial comparing device intervention to standard of care for the treatment of acute intermediate-risk pulmonary embolism using clinically meaningful endpoints.
The trial met its combined primary endpoint, showing that treatment with the EKOS Endovascular System plus anticoagulation was superior to anticoagulation alone (4.0% vs 10.3%, P=0.005), representing 61% fewer patients experiencing primary endpoint events, defined as PE-related mortality, non-fatal symptomatic recurrence of PE, and non-fatal hemodynamic cardiorespiratory decompensation or collapse within seven days. Results were achieved without an increased risk of major bleeding compared with anticoagulation alone. There were no episodes of intracranial bleeding at 30 days in either trial arm. Patients treated with EKOS also had a shorter hospital stay and shorter duration in critical care/ICU compared with those treated with anticoagulation alone.
Investigators and authors:
- Michael Jaff, DO, Chief Medical Officer, Vascular Therapies, Boston Scientific (NEJM author)
- Stavros Konstantinides, MD, principal investigator of HI-PEITHO and Medical Director, Center for Thrombosis and Hemostasis, University Medical Center Mainz, Germany (NEJM author)
About the ACC Scientific Session 2026
The American College of Cardiology Scientific Session is a leading cardiovascular meeting featuring late-breaking science, clinical research, and educational programming.
Find More:
Renal Denervation Topic Center
Cardiovascular Ambulatory Surgery Centers (ASCs) Topic Center
Grand Rounds With Morton Kern, MD
Peripheral Artery Disease Topic Center