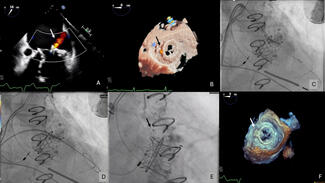
Cardiopulmonary Bypass is Safe and Effective Support for Concomitant High-Risk Percutaneous Coronary Intervention and Transcatheter Aortic Valve Replacement
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/25.00352. Epub February 23, 2026.
Abstract
Objectives. Patients with coexisting severe aortic stenosis (AS) and critical coronary artery disease (CAD) face high procedural risk when treated percutaneously. Although mechanical circulatory support (MCS) can mitigate hemodynamic instability, evidence guiding the use of cardiopulmonary bypass (CPB) or extracorporeal membrane oxygenation (ECMO) during concomitant transcatheter aortic valve replacement (TAVR) and percutaneous coronary intervention (PCI) is lacking.
Methods. The authors retrospectively reviewed 6 high-risk patients who underwent concomitant TAVR and PCI with planned MCS using CPB or venoarterial (VA)-ECMO between 2015 and 2023. Outcomes were classified according to VARC-3 criteria.
Results. Six male patients (mean age 77.3 ± 8.3 years) underwent concomitant TAVR and PCI with planned MCS using CPB (n = 4, 2015-2020) or VA-ECMO (n = 2, 2022-2023). Complete coronary revascularization was achieved in 4 cases (66.7%). Implanted valves included 2 self-expanding and 4 balloon-expandable prostheses. The mean catheter-measured transaortic gradient decreased from 37.2 ± 17.6 mm Hg to 3.9 ± 2.1 mm Hg (P = .006). Trace (1+) paravalvular leak occurred in 4 patients (66.7%), with none 2+ or higher. There were no intraprocedural complications, neurologic events, major bleeding, vascular complications, or 30-day deaths. Three patients (50%) developed new conduction abnormalities, all without pacemaker requirement. Acute kidney injury occurred in 3 (Stage I, 50%). The mean intensive care and total hospital stays were 5.5 ± 3.5 and 8.3 ± 6.0 days, respectively.
Conclusions. Planned CPB or VA-ECMO support enabled safe performance of concomitant TAVR and PCI in non-surgical, high-risk patients, representing a feasible approach for selected complex cases.
Introduction
Transcatheter aortic valve replacement (TAVR) is now an established therapy for severe aortic stenosis (AS) across all surgical risk categories.1,2 Coronary artery disease (CAD) is present in up to 60% of patients undergoing TAVR evaluation3 and is associated with an increased risk of adverse cardiovascular events and mortality.4 For patients with concomitant severe AS and obstructive CAD who are not candidates for surgical aortic valve replacement (SAVR) and/or coronary artery bypass grafting (CABG), combined transcatheter approaches using TAVR and percutaneous coronary intervention (PCI) offer a viable alternative.
However, both TAVR and PCI can precipitate profound hemodynamic instability in patients with advanced AS or left ventricular (LV) dysfunction.1,5,6 Among high-risk nonsurgical patients undergoing isolated TAVR, mechanical circulatory support (MCS) is required in up to 10% of cases,7 with associated in-hospital mortality rates of 25% to 50%.7-9 Notably, elective MCS deployment has been associated with approximately a 50% reduction in mortality compared with emergent use, underscoring the potential benefit of planned hemodynamic support strategies.8,9
A variety of MCS modalities have been utilized to mitigate procedural risk. Devices such as the intra-aortic balloon pump (IABP) and Impella (Abiomed) are commonly employed in isolated or staged high-risk PCI and TAVR procedures.10,11 The use of cardiopulmonary bypass (CPB) and extracorporeal membrane oxygenation (ECMO) has also been reported, primarily in isolated TAVR cases or staged PCI-TAVR interventions.12-14 However, reports of their planned use during truly concomitant TAVR and PCI are exceedingly limited. To date, only isolated case reports involving 1 or 2 patients have been published.15-17 Consequently, the optimal strategy for MCS during combined TAVR and PCI remains undefined, and evidence to guide clinical decision-making is virtually absent.
In this study, we describe the procedural characteristics and short- and mid-term outcomes of 6 high-risk patients who underwent concomitant TAVR and PCI under planned CPB or ECMO support at a single Veterans Affairs Healthcare system.
Methods
Study design and population
We conducted a retrospective review of medical records for patients who underwent concomitant TAVR and PCI under planned MCS between 2015 and 2023 at the San Francisco Veterans Affairs Healthcare System (SFVAHCS). After screening 666 patients evaluated for TAVR in a prospectively maintained institutional database, 6 patients undergoing concomitant TAVR and PCI with planned MCS were identified and included.
Eligible patients had severe AS and critical CAD and were deemed prohibitive surgical candidates for open aortic valve replacement and/or coronary artery bypass grafting (CABG) because of high or unacceptable operative risk. Candidacy was determined by a multidisciplinary cardiovascular team composed of interventional cardiologists, cardiac surgeons, and cardiac anesthesiologists.
Only patients undergoing concomitant TAVR and PCI with planned, preemptive MCS using CPB or venoarterial (VA)-ECMO were included. Planned MCS was selected based on anticipated hemodynamic risk related to the severity of AS, complexity of coronary disease, ventricular function, and procedural considerations. Patients requiring unplanned or emergent MCS during isolated TAVR procedures (eg, bailout support for intraprocedural complications) were excluded. No patients undergoing concomitant TAVR and PCI required unplanned or emergent MCS.
This study was approved by the institutional review board of the SFVAHCS, with a waiver of informed consent granted due to the retrospective nature of the study and minimal risk to participants.
Data collection
Clinical, echocardiographic, and procedural data were collected from the Veterans Affairs electronic health record. Procedural details were abstracted from operative notes and catheterization laboratory reports. Postprocedural and follow-up outcomes were recorded according to the Valve Academic Research Consortium-3 (VARC-3) definitions, including mortality, bleeding, vascular and access-related complications, conduction disturbances, and renal outcomes.18 Multivessel CAD was defined as significant stenosis in at least 2 major epicardial coronary arteries.
Procedural approach
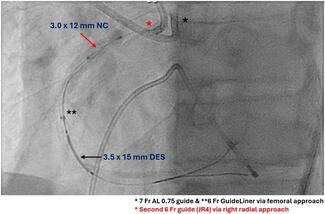
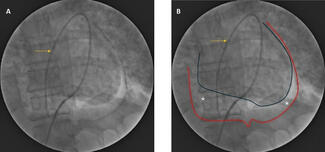
Before each concomitant TAVR and PCI procedure, the multidisciplinary cardiovascular team—including cardiac surgery, interventional cardiology, anesthesiology, and perfusion—performed a comprehensive evaluation to determine perioperative risk and procedural strategy. MCS was provided using CPB in early cases, whereas beginning in 2022 the institution transitioned to VA-ECMO as the preferred modality.
For both CPB and VA-ECMO, arterial cannulation was performed through the common femoral artery, with the cannula advanced into the abdominal aorta under fluoroscopic guidance. Venous drainage was established via the common femoral vein, with the cannula advanced into the right atrium. Bilateral femoral arterial access was routinely obtained to allow arterial pressure monitoring, angiographic imaging, and procedural control. MCS was initiated prophylactically at the start of the intervention or promptly upon signs of hemodynamic instability or myocardial ischemia.
The coronary revascularization strategy was determined by a multidisciplinary heart team. Complete revascularization was defined as percutaneous treatment of all intervenable coronary lesions at the time of the index procedure, whereas targeted revascularization was defined as treatment of high-risk culprit lesions with intentional deferral of non-culprit disease for staged intervention. Lesions selected for treatment were those judged to confer the greatest risk of periprocedural ischemia or hemodynamic instability during TAVR, including left main (LM), high grade proximal disease, or supplying large myocardial territories.
The procedure began with percutaneous coronary revascularization of critical coronary lesions, followed by TAVR during the same index procedure. MCS was planned and used throughout the intervention; when feasible, patients were weaned from MCS prior to TAVR, and all patients were successfully weaned by the end of the concomitant PCI-TAVR procedure. Weaning from cardiopulmonary bypass or extracorporeal membrane oxygenation was performed using a standardized stepwise approach similar to that used in open cardiac surgery, with gradual reduction in pump flow and progressive ventricular loading under transesophageal echocardiographic and hemodynamic monitoring. Vasopressors and inotropes were administered as needed to support vasoplegia or impaired myocardial contractility. Once weaned, the cannulas were clamped and left in place for a brief period of observation to confirm hemodynamic stability prior to decannulation.
All patients received periprocedural antiplatelet loading prior to PCI, and procedural anticoagulation with heparin was reversed with protamine at the conclusion of the procedure. Valve type and size were selected at the discretion of the treating heart team based on patient-specific factors, anatomic considerations, and procedural feasibility.
Statistical analysis
Continuous variables were summarized as mean ± standard deviation (SD) and categorical variables as counts and percentages. Changes in pre- and post-procedure left ventricular ejection fraction (LVEF) and mean transaortic gradient were evaluated using the paired Student’s t-test. All analyses were performed using R software (version 2024.04.2; R Foundation for Statistical Computing). A 2-tailed P-value of less than 0.05 was considered statistically significant.
Results
Patient characteristics
From 2015 to 2023, 666 patients underwent TAVR at SFVAHCS. Among these, 6 consecutive patients with severe symptomatic AS and critical CAD underwent concomitant TAVR and PCI with MCS using either CPB or ECMO.
All patients were male, with a mean age of 77.3 ± 8.3 years. The cohort carried a substantial comorbidity burden, most commonly hypertension (5 patients, 83.3%), dyslipidemia (5, 83.3%), and a history of smoking (3, 50%). A bicuspid aortic valve was identified in 1 patient (16.7%). All patients were symptomatic, presenting with New York Heart Association (NYHA) functional class III or IV heart failure (100%). Three patients (50%) had a history of prior PCI, and 1 (16.7%) had undergone previous CABG. Two patients (33.3%) had decompensated heart failure preprocedurally, including 1 patient (16.7%) in cardiogenic shock requiring low-dose single-agent vasopressor support.
All patients presented with severe AS, with a mean pre-procedure transaortic gradient of 37.2 ± 17.6 mm Hg and aortic valve orifice area of 0.89 ± 0.31 cm². The mean number of diseased coronary vessels was 3.33 ± 1.21. Preprocedural patient characteristics are summarized in Table 1.
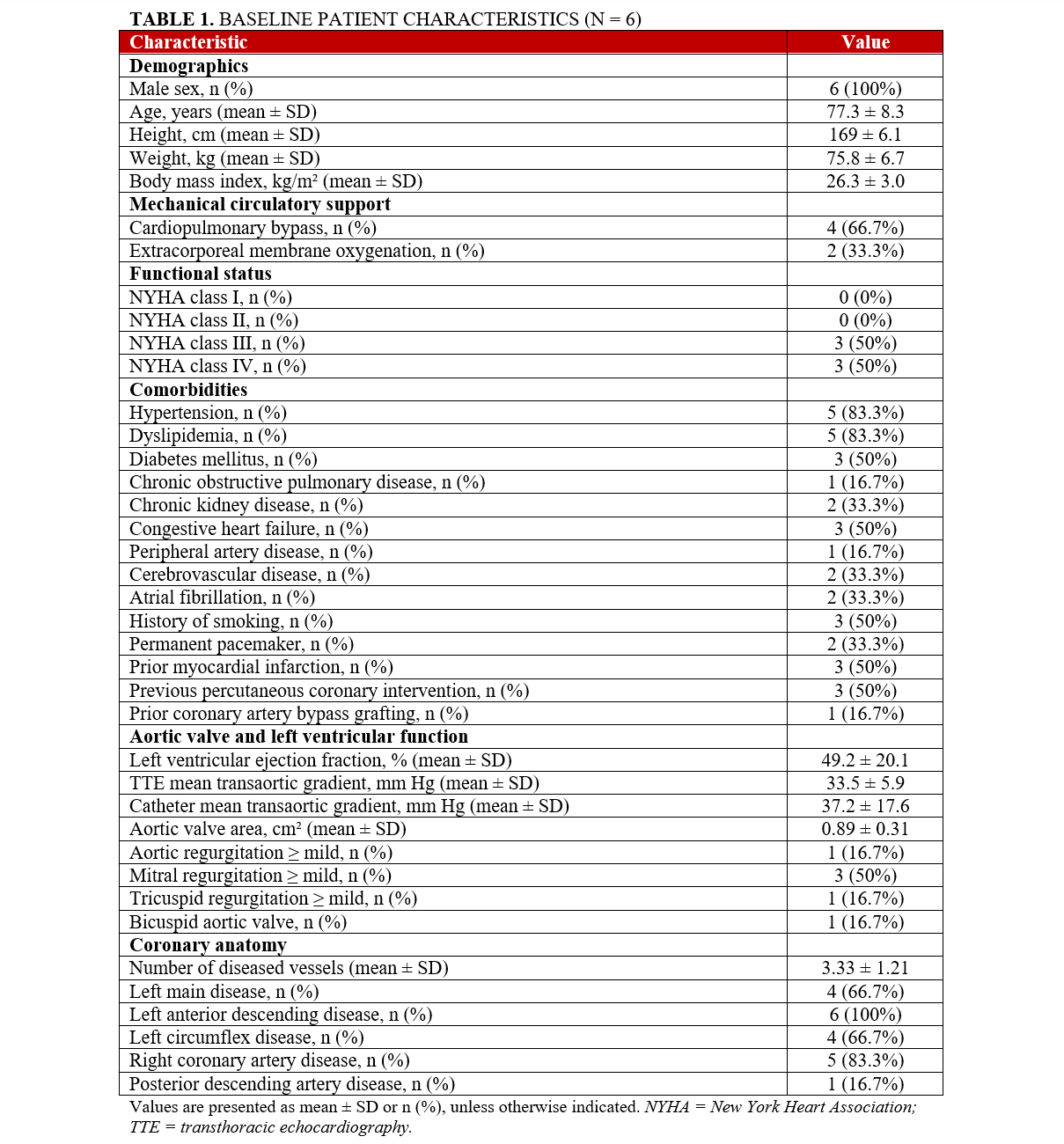
Procedural characteristics
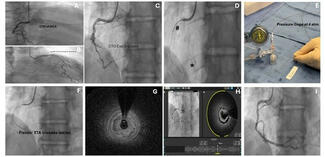
All patients underwent TAVR and PCI during a single procedural session. CPB was employed in 4 patients (66.7%) between 2015 and 2020, while VA-ECMO was used in 2 patients (33.3%) in more recent cases (2022-2023). MCS was planned preprocedurally in all cases. Valve deployment was successful in all 6 patients, with no instances of device malposition, migration, or conversion to open surgery. Implanted valves included 2 tall-frame self-expanding prostheses (26-mm CoreValve and 34-mm CoreValve Evolut R [Medtronic]) and 4 short-frame balloon-expandable valves (29-mm SAPIEN 3, 23-mm SAPIEN 3, 29-mm SAPIEN 3, and 26-mm SAPIEN 3 Ultra [Edwards Lifesciences]).
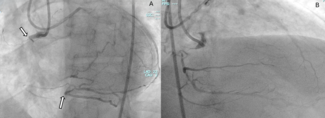
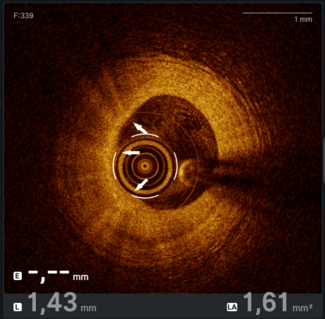
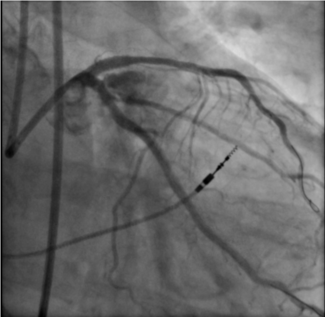
Complete coronary revascularization was achieved in 4 (66.7%) patients, while successful technical revascularization of all targeted culprit lesions was achieved in all patients. Detailed coronary lesion characteristics and PCI procedural details are summarized in Table 2. A mean of 1.33 ± 1.03 stents was implanted per patient, with more than 1 target lesion treated in 3 patients (50%). PCI most frequently involved the LM, left anterior descending (LAD), and left circumflex (LCx) arteries, each treated in 4 patients (66.7%), while the right coronary artery (RCA) was treated in 1 patient (16.7%). Overall, the cohort demonstrated multiple features of complex coronary anatomy, including frequent multivessel disease, LM involvement, long lesions, and severe calcification.
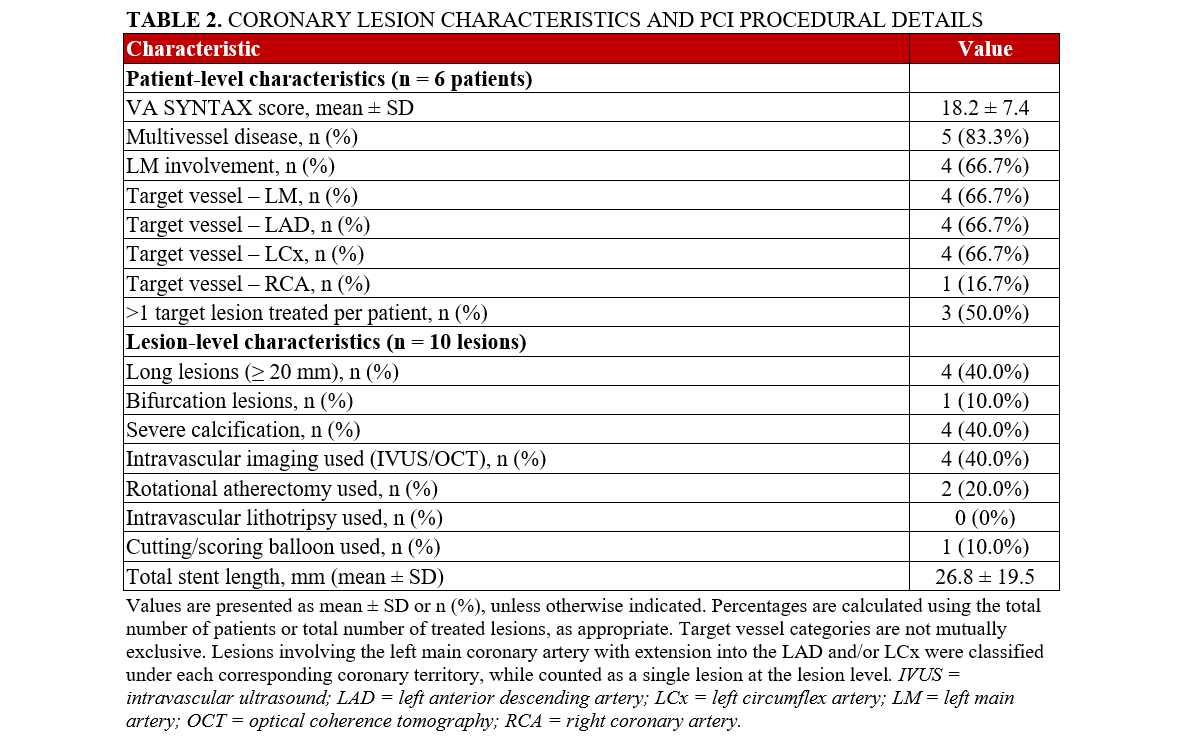
The mean total procedure time was 217 ± 33 minutes, and the mean duration of mechanical circulatory support was 63.7 ± 46.8 minutes. There were no intraprocedural deaths. Two patients (33.3%) required intraoperative transfusion of packed red blood cells.
Hemodynamic and echocardiographic outcomes
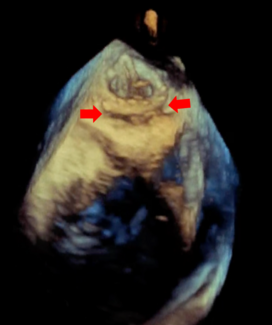
Postprocedural invasive hemodynamic assessment demonstrated a mean catheter-measured transaortic gradient of 3.85 ± 2.05 mm Hg, significantly reduced from a pre-procedure gradient of 37.2 ± 17.6 mm Hg (P = .006). Transthoracic echocardiography (TTE) similarly showed a marked decrease in mean transaortic gradient from 33.5 ± 15.9 mm Hg pre-procedure to 12.1 ± 6.03 mm Hg following valve implantation (P = .01).
LV systolic function remained stable following intervention, with a mean postprocedural LVEF of 52.5 ± 19.7%, compared with 49.2 ± 20.1% before the procedure (P = .33).
Paravalvular regurgitation was minimal in all cases. Postprocedural echocardiography demonstrated trace paravalvular leak (PVL) (1+) in 4 patients (66.7%), while the remaining 2 patients (33.3%) had no detectable regurgitation. No moderate or severe (≥ 2+) PVL was observed.
Clinical outcomes
Detailed clinical outcomes according to the VARC-3 criteria are summarized in Table 3.
Mortality and neurologic events
Thirty-day all-cause mortality and cardiovascular mortality were 0 (0%). No patient experienced a neurological event.
Hospitalization and cardiovascular events
Hospital readmission within 30 days occurred in 1 patient (16.7%). This patient was first readmitted on postoperative day (POD) 18 for worsening chronic kidney disease due to hypovolemia. Kidney function improved with volume resuscitation without need for dialysis. No patient experienced myocardial infarction during the index hospitalization or follow-up period.
Bleeding, transfusion, and vascular complications
Life-threatening or major bleeding occurred in 0 patients (0%), while 4 (66.7%) required transfusion of packed red blood cells postoperatively. No major vascular or access-site complications were observed in any patient (0%).
Cardiac structural and valve-related complications
No cardiac structural complications such as annular rupture, coronary obstruction, or ventricular injury were identified. Bioprosthetic valve dysfunction was not observed, and there were no cases of leaflet thickening, reduced leaflet motion, or clinically significant valve thrombosis on follow-up imaging.
Conduction disturbances and arrhythmias
New conduction abnormalities were detected in 3 patients (50%). Two (33.3%) developed new right bundle branch block, and 1 (16.7%) developed first-degree atrioventricular block. No patient required new permanent pacemaker implantation, and none experienced new-onset atrial fibrillation or other clinically significant arrhythmias.
Renal outcomes
Acute kidney injury (AKI) occurred in 3 patients (50%), classified as Stage I in all cases. No patient required new renal replacement therapy.
Pulmonary and other postoperative complications
One patient (16.7%) developed an incidental, asymptomatic pulmonary embolism detected on postoperative imaging and was treated medically. Another patient (16.7%) developed a pleural effusion requiring tube thoracostomy.
Length of stay
The mean intensive care unit (ICU) length of stay was 5.5 ± 3.5 days, and the mean total hospital length of stay was 8.3 ± 6.0 days.
Patient-reported outcomes
At 30 days, 6 surviving patients (100%) reported improvement in functional status.
Discussion
Our experience demonstrates that the elective application of CPB or VA-ECMO can safely facilitate concomitant TAVR and PCI in select high-risk patients. In this case series of 6 men with severe AS and critical CAD deemed unsuitable for surgery, both complete revascularization and successful valve implantation were achieved without procedural or 30-day deaths. The absence of major bleeding, vascular injury, or neurologic events underscores the safety and feasibility of elective MCS for these complex, non-surgical cases.
Minor complications were limited and clinically manageable. Three patients (50%) developed mild, Stage I AKI that resolved without dialysis, and 3 (50%) experienced new conduction abnormalities—2 right bundle branch blocks and 1 first-degree atrioventricular block—none requiring permanent pacing. Four patients (66.7%) required postprocedural blood transfusions, but no major bleeding or vascular complications occurred. These findings are comparable with complication rates reported in concomitant TAVR/PCI populations, where AKI occurs in approximately 20% to 40% of patients and transfusion requirements range from 15% to 40%.8,19-21
Importantly, the need for MCS is reported in fewer than 10% of TAVR cases,7 reflecting the greater procedural complexity and comorbidity burden of our high-risk, non-surgical cohort. In this context, the use of planned CPB or VA-ECMO added only minor morbidity and may enhance hemodynamic safety during complex combined interventions.
Elective or prophylactic use of VA-ECMO or CPB in high-risk patients undergoing PCI or TAVR remains uncommon.8 Traditionally, these modalities have been reserved for intraprocedural complications such as LV or aortic annular rupture, hemodynamic deterioration, cardiac arrest, or LM obstruction.11,13,22,23 Emergent VA-ECMO or CPB use, however, has been associated with mortality rates of 25% to 50%.7-9 While elective deployment of ECMO or CPB is an evolving concept, it has already been associated with improved outcomes and an approximately 50% reduction in mortality compared with emergent use.8,9 In our case series of 6 patients undergoing elective VA-ECMO or CPB, no deaths occurred through 90 days, further demonstrating the safety of this strategy.
Elective use offers several advantages. Planned deployment allows optimization of cannulation strategy, procedural sequencing, and minimization of time from hemodynamic instability to initiation of support. At our institution, elective MCS is considered during concomitant PCI/TAVR in patients with severe AS and multivessel CAD, particularly involving the LM, proximal LAD, or RCA. Patients with biventricular dysfunction or acute heart failure are also considered for elective support.24 Decisions for elective ECMO or CPB use are made in a multidisciplinary cardiovascular meeting that includes interventional cardiology, cardiac surgery, and cardiac anesthesiology. While the Impella device is commonly used for MCS,10,11 our center has introduced the elective use of VA-ECMO and CPB in these high-risk patients because they allow unloading of both ventricles and full hemodynamic control, whereas Impella is limited to LV support with lower flow capacity.12,25 Furthermore, we have transitioned from CPB to VA-ECMO, which allows for more rapid deployment, less heparinization, and efficient weaning while providing comparable circulatory support.
Published experience with truly concomitant TAVR and PCI under planned ECMO or CPB support remains extremely limited. Prior reports have been confined to isolated case descriptions or small 2-patient series.15-17 Kmiec et al reported successful use of planned VA-ECMO during concomitant PCI and TAVR in an 88-year-old man with low-flow, low-gradient AS, LM and LAD disease, reduced LVEF (35%), and severe pulmonary hypertension, without intra- or postoperative complications.16 Marchese et al similarly described prophylactic VA-ECMO with IABP in a 79-year-old man with severe AS, multivessel CAD involving the LM and LAD, occluded RCA, moderate chronic obstructive pulmonary disease, and an LVEF of 26% who underwent PCI with intravascular lithotripsy and stenting followed by TAVR, also without complications.17 Bulnes et al presented a 10-patient series of planned VA-ECMO for TAVR and/or PCI, of whom only 2 underwent truly concomitant procedures.15 Reported complications included 1 (10%) vascular injury requiring surgical repair and 1 (10%) perioperative MI from LM dissection, with no mortality. These isolated experiences demonstrated procedural feasibility but offered limited follow-up and incomplete outcome reporting.
Our study expands upon prior reports by presenting, to our knowledge, the largest dedicated experience of concomitant TAVR and PCI performed with pre-planned CPB or VA-ECMO support. The absence of procedural mortality, stroke, or major vascular complications highlights the safety and reproducibility of this approach in carefully selected nonsurgical patients. These findings suggest that proactive hemodynamic support can facilitate complete revascularization and successful valve implantation in patients who would otherwise face prohibitive procedural risk.
Limitations
This study is limited by its retrospective design, small sample size, and single-center experience, which restrict generalizability. The cohort was composed entirely of male veterans, further limiting applicability to broader patient populations. Given the absence of a control group, causal inference regarding the benefit of planned CPB or VA-ECMO cannot be established. Additionally, procedural selection reflects institutional experience and multidisciplinary judgment, which may not be uniform across centers. By focusing exclusively on patients undergoing planned, preemptive MCS, our findings do not reflect outcomes of unplanned emergent or bailout support and may underestimate complication rates seen in higher-risk patient populations. Despite these limitations, this case series provides important evidence that elective hemodynamic support can be safely and effectively implemented during concomitant TAVR and PCI in high-risk, nonsurgical patients and warrants further evaluation in larger studies.
Conclusions
Elective use of CPB or VA-ECMO provided hemodynamic stability and allowed complete coronary and valve interventions in patients who would otherwise be considered prohibitive surgical candidates. These findings support the feasibility and safety of proactive circulatory support as a bridge to complex transcatheter therapies.
Affiliations and Disclosures
Axel Gomez, MD1; Madison E. Matthews1; Aaron Grober, MD2; Ryan Ferreira, BS1; Kendrick A. Shunk, MD, PhD2; Jeffrey Zimmet, MD, PhD2; Elaine E. Tseng, MD1; Marko T. Boskovski, MD1
From the 1Division of Adult Cardiac Surgery and Lung Transplantation, Department of Surgery, University of California San Francisco and San Francisco VA Healthcare System, San Francisco, California; 2Division of Cardiology, Department of Medicine, University of California San Francisco and San Francisco VA HealthCare System, San Francisco, California.
Acknowledgments: ChatGPT (GPT-5; OpenAI) was used to assist with language editing across all sections of the manuscript. The authors reviewed and revised all content and take full responsibility for the final manuscript.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Funding: This work was supported by the VA Office of Research and Development [I01CX002365-01A1]; and the National Center for Advancing Translational Sciences [TL1-TR001871].
Data availability statement: The data that support the findings of this study are available from the corresponding author upon reasonable request.
Address for correspondence: Marko T. Boskovski, MD, MHS, MPH, Division of Adult Cardiac Surgery and Lung Transplantation, UCSF Medical Center, 500 Parnassus Ave, Suite 405W, Box 0118, San Francisco, CA 94143-0118, USA. Email: Marko.boskovski@ucsf.edu
References
1. Leon MB, Smith CR, Mack M, et al; PARTNER Trial Investigators. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363(17):1597-1607. doi:10.1056/NEJMoa1008232
2. Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143(5):e35-e71. doi:10.1161/CIR.0000000000000932
3. Abdel-Wahab M, Zahn R, Horack M, et al. Transcatheter aortic valve implantation in patients with and without concomitant coronary artery disease: comparison of characteristics and early outcome in the German multicenter TAVI registry. Clin Res Cardiol. 2012;101(12):973-981. doi:10.1007/s00392-012-0486-5
4. Cao D, Chiarito M, Pagnotta P, Reimers B, Stefanini GG. Coronary revascularisation in transcatheter aortic valve implantation candidates: why, who, when? Interv Cardiol. 2018;13(2):69-76. doi:10.15420/icr.2018:2:2
5. Kodali SK, Williams MR, Smith CR, et al; PARTNER Trial Investigators. Two-year outcomes after transcatheter or surgical aortic-valve replacement. N Engl J Med. May 03 2012;366(18):1686-1695. doi:10.1056/NEJMoa1200384
6. Weipert KF, Bauer T, Nef HM, et al. Incidence and outcome of peri-procedural cardiogenic shock: results from the international Euro Heart Survey PCI registry. Eur Heart J Acute Cardiovasc Care. 2020;9(2):120-127. doi:10.1177/2048872618822460
7. Singh V, Damluji AA, Mendirichaga R, et al. Elective or emergency use of mechanical circulatory support devices during transcatheter aortic valve replacement. J Interv Cardiol. 2016;29(5):513-522. doi:10.1111/joic.12323
8. Vallabhajosyula S, Patlolla SH, Sandhyavenu H, et al. Periprocedural cardiopulmonary bypass or venoarterial extracorporeal membrane oxygenation during transcatheter aortic valve replacement: a systematic review. J Am Heart Assoc. 2018;7(14):e009608. doi:10.1161/JAHA.118.009608
9. Shreenivas SS, Lilly SM, Szeto WY, et al. Cardiopulmonary bypass and intra-aortic balloon pump use is associated with higher short and long term mortality after transcatheter aortic valve replacement: a PARTNER trial substudy. Catheter Cardiovasc Interv. 2015;86(2):316-322. doi:10.1002/ccd.25776
10. Singh V, Yarkoni A, O'Neill WW. Emergent use of Impella CP™ during transcatheter aortic valve replacement: transaortic access. Catheter Cardiovasc Interv. 2015;86(1):160-163. doi:10.1002/ccd.25784
11. Banjac I, Petrovic M, Akay MH, et al. Extracorporeal membrane oxygenation as a procedural rescue strategy for transcatheter aortic valve replacement cardiac complications. ASAIO J. 2016;62(1):e1-e4. doi:10.1097/MAT.0000000000000275
12. Di Muro FM, Bellino M, Esposito L, et al. Role of mechanical circulatory support in complex high-risk and indicated percutaneous coronary intervention: current indications, device options, and potential complications. J Clin Med. 2024;13(16):4931. doi:10.3390/jcm13164931
13. Avula V, Karacsonyi J, Hammadah M, Brilakis ES. Venoarterial extracorporeal membrane oxygenation for life-threatening complications of percutaneous coronary and structural heart interventions. Cardiovasc Revasc Med. 2022;40S:308-311. doi:10.1016/j.carrev.2021.11.011
14. Lesbekov T, Mussayev A, Alimbayev S, et al. Prophylactic awake peripheral V-A ECMO during TAVR. J Clin Med. 2023;12(3):859. doi:10.3390/jcm12030859
15. Bulnes JF, Martínez A, Sepúlveda P, et al. Outcomes of a modified, low-cost, veno-arterial extracorporeal membrane oxygenation (V-A ECMO) for elective, periprocedural support of high-risk percutaneous cardiac interventions: an experience from a latinamerican center. Perfusion. 2024;39(5):998-1005. doi:10.1177/02676591231178413
16. Kmiec L, Holzamer A, Fischer M, et al. Protected complex percutaneous coronary intervention and transcatheter aortic valve replacement using extracorporeal membrane oxygenation in a high-risk frail patient: a case report. J Med Case Rep. 2020;14(1):163. doi:10.1186/s13256-020-02474-x
17. Marchese A, Tarantini G, Tito A, et al. Mechanical circulatory support and intravascular lithotripsy in high-risk patients undergoing percutaneous coronary intervention and transcatheter aortic valve replacement: a case series. Eur Heart J Case Rep. 2021;5(12):ytab498. doi:10.1093/ehjcr/ytab498
18. Généreux P, Piazza N, Alu MC, et al; VARC-3 WRITING COMMITTEE. Valve Academic Research Consortium 3: updated endpoint definitions for aortic valve clinical research. Eur Heart J. 2021;42(19):1825-1857. doi:10.1093/eurheartj/ehaa799
19. Ghrair F, Omran J, Thomas J, et al. Outcomes of concomitant percutaneous coronary interventions and transcatheter aortic valve replacement. Arch Med Sci Atheroscler Dis. 2020;5:e284-e289. doi:10.5114/amsad.2020.103092
20. Tran Z, Hadaya J, Downey P, et al. Staged versus concomitant transcatheter aortic valve replacement and percutaneous coronary intervention: a national analysis. JTCVS Open. 2022;10:148-161. doi:10.1016/j.xjon.2022.02.019
21. Zghouzi M, Osman H, Erdem S, et al. In-hospital outcomes of combined coronary revascularization and transcatheter aortic valve implantation in inpatient nationwide analysis. Curr Probl Cardiol. 2024;49(1 Pt A):101913. doi:10.1016/j.cpcardiol.2023.101913
22. Okuyama K, Jilaihawi H, Makkar RR. Leaflet length and left main coronary artery occlusion following transcatheter aortic valve replacement. Catheter Cardiovasc Interv. 2013;82(5):E754-E759. doi:10.1002/ccd.25059
23. Spina R, Khalique O, George I, Nazif T. Acute left main stem coronary occlusion following transcatheter aortic valve replacement in a patient without recognized coronary obstruction risk factors: a case report. Eur Heart J Case Rep. 2018;2(4):yty112. doi:10.1093/ehjcr/yty112
24. Seco M, Forrest P, Jackson SA, et al. Extracorporeal membrane oxygenation for very high-risk transcatheter aortic valve implantation. Heart Lung Circ. 2014;23(10):957-962. doi:10.1016/j.hlc.2014.05.006
25. Farag M, Malik IS. Mechanical circulatory support for percutaneous coronary intervention in high-risk patients undergoing transcatheter aortic valve replacement. Eur Heart J Case Rep. 2022;6(2):ytac037. doi:10.1093/ehjcr/ytac037