Clinical Pathways to Address Racial Disparities in Uterine Cancer: A SEER-Based Needs Analysis
Abstract
Racial disparities in uterine cancer stem from biological, clinical, and socioeconomic factors, yet the impact of clinical pathways in mitigating these factors remains largely unexamined. Using Surveillance, Epidemiology, and End Results (SEER) data and propensity score matching, we analyzed uterine cancer cases to assess disparities between non-Hispanic (NH) Black and White patients in hysterectomy rates and overall survival (OS). In Cohort 1 (N = 133 839), 10 952 NH Black patients were matched 1:1 with 10 952 NH White patients; NH Black patients had lower hysterectomy rates and shorter median OS (114 vs 170 months, P < .001). In Cohort 2 (N = 59 516), 9314 NH Black patients were matched 1:1 with 9314 NH White patients. Similar disparities persisted, with NH Black patients showing lower hysterectomy rates, higher mortality (30.67% vs 23.15%, P < .001), and higher risk of death (HR, 1.41; 95% CI, 1.34-1.49). Moreover, NH White patients lived a median of 4.5 years longer than NH Black patients, who also had higher mortality risk. These findings suggest that equity-focused clinical pathways that standardize hysterectomy recommendations and address social determinants, along with real-time monitoring, may help reduce racial disparities in uterine cancer and improve care of NH Black patients.
Introduction
Although overall cancer incidence and mortality have declined in the US, the incidence of uterine cancer continues to rise, with mortality approaching that of ovarian cancer, the deadliest gynecologic cancer.1-3 Non-Hispanic (NH) Black patients bear a disproportionate and growing burden of uterine cancer mortality, largely driven by higher prevalence of aggressive, nonendometrioid histologic subtypes that metastasize earlier and are associated with poorer survival.1,4-9 Structural inequities rooted in systemic racism and discrimination contribute to persistent disparities between NH Black and NH White patients.10 Early-life disadvantages, including poor maternal nutrition and socioeconomic hardship, further compound these disparities.2
Barriers that contribute to racial and ethnic disparities across the cancer care continuum include lower health literacy, less insurance coverage, limited access to quality care, and higher obesity.2,3,11-20 Additionally, some diagnostic tools, such as transvaginal ultrasonography, may lack adequate validation across diverse populations, potentially delaying diagnosis and treatment for NH Black women.2,21,22 Compared with NH White patients, NH Black patients are more likely to have aggressive tumor biology, higher rates of surgical complications, and significantly lower 5-year overall survival (OS; 63% vs 84%).2,3,14,23-28 Biological and environmental factors may also contribute; NH Black patients are more likely to be genetically predisposed to aggressive tumor histology and greater exposure to environmental carcinogens.2,29,30 Additional contributors include later-stage and higher-grade tumors at diagnosis, lower eligibility for and response to immunotherapy,6 higher comorbidity burden,2,29,31 and elevated obesity.15 Although histology and insurance status have been identified as key drivers of disparity through propensity score matching (PSM) analyses, these factors do not fully account for the persistent racial gap in outcomes, even when access to care is equal.1,2,26,32,33 Notwithstanding the National Comprehensive Cancer Network® (NCCN®) Clinical Practice Guidelines in Oncology (NCCN Guidelines®)34 and American College of Obstetricians and Gynecologists guidelines,35 NH Black patients are less likely to receive guideline-concordant care2,33 and more likely to undergo hysterectomy for benign conditions.27
Between 2015 and 2019, key advances in uterine cancer management emerged, including refined surgical staging, sentinel lymph node mapping, adjuvant therapies for high-risk disease, and targeted treatments for HER2/neu-positive tumors.36 Innovations in combination therapies, molecular profiling, and immunotherapy have further improved outcomes.36 Yet, NH Black patients continue to experience worse OS due to a confluence of aggressive tumor biology, treatment disparities, socioeconomic barriers, and potential biological differences.32 They are also more likely to undergo abdominal surgery rather than minimally invasive hysterectomy and to receive first-line radiation and chemotherapy for locoregional disease.2,32,33,37 Although clinical pathways have improved the consistency and quality of care, their potential to mitigate racial disparities in uterine cancer treatment and outcomes remains underexplored. Integrating equity-focused pathways that standardize surgical recommendations and incorporate social determinants of health (SDOH) may help reduce these disparities.
This study aimed to evaluate differences in first-line hysterectomy recommendation and receipt between NH Black and NH White patients with a diagnosis of uterine cancer during 2 periods: 2000-2017 (Cohort 1) and 2015-2021 (Cohort 2). A secondary objective was to compare OS by race.
Methods
This retrospective registry study used 2 overlapping cohorts from the Surveillance, Epidemiology, and End Results (SEER) data cycles of 2022 (Cohort 1, 2000-2017) and 2023 (Cohort 2, 2015-2021) to examine racial/ethnic differences in surgical recommendation, receipt, and OS outcomes between NH Black and NH White patients.38 Cohort 1 accounted for longer-term trends, whereas Cohort 2 focused on patients diagnosed during recent staging and therapeutic advances. Cohort 1 data covered 2000 to 2020, with diagnoses reported up to 2017. Cohort 2 data covered 2015 to 2021, with diagnoses reported up to 2021. Data related to cause of death were based on state death certificates; survival represented patients who did not die of the specified cause.
Both SEER databases use American Community Survey (ACS) 5-year estimates, covering 26.5% of the US population. County-level data link cancer cases and deaths to static attributes (collected at a single point) and time-dependent attributes (collected at different points). The ACS cycle for SEER 2022 and 2023 provides detailed demographic, social, economic, and housing data for county attributes. Rural-urban continuum codes distinguish metropolitan counties from adjacent nonmetropolitan counties by population. The time to first-line treatment (hysterectomy) was capped at 24 months in both cohorts. The SEER variable RX Summ—Surg Prim Site (1998+) provided surgical codes for hysterectomies, based on the SEER Program Coding and Staging Manual 2023, surgical codes Corpus Uteri C540–C559.39 Surgical decisions were based on tumor stage, using the SEER variable Summary Stage 2000 (1998-2017), which simplifies stage categories by in situ, localized, regional, distant, and unknown. Surgical recommendation and receipt were captured using the variable “reason no cancer-directed surgery,” which specified whether and why hysterectomy was or was not recommended and/or performed.
Cohort 1 included NH Black or NH White women aged 18 years or older with a diagnosis of first and only malignancy between 2000 and 2017, with known stage and histology. Exclusions were unknown tumor grade, median household income, metropolitan proximity, benign/borderline malignancies, or autopsy diagnoses. Median household income was dichotomized using the 2021 US Census estimate of $70 000.40 Histology was categorized as corpus uteri carcinoma and carcinosarcoma, corpus uteri adenosarcoma, or corpus uteri sarcoma. Cohort 2 included NH Black or NH White women aged 18 years or older with microscopically confirmed uterine cancer diagnosed between 2015 and 2021. Exclusions were benign/borderline malignancies or autopsy diagnoses. Median household income was dichotomized using the average 2015-2021 US Census estimate of $65 000.41 To ensure standardized diagnostic data, we included only patients with microscopically confirmed uterine cancer. To better represent tumor histology, uterine cancer was categorized as endometrioid, nonendometrioid, sarcoma, or other (see Supplement).42 Geographic US regional data were not available in SEER for 2023.
We conducted descriptive and comparative analyses for both cohorts before and after balancing baseline characteristics. Patients were matched 1:1 on age at diagnosis, median household income, proximity to metropolitan area, marital status, and primary tumor site, histology, stage, and grade. Propensity scores were calculated using logistic regression to estimate the probability of each patient receiving hysterectomy based on age at diagnosis, median household income, proximity to metropolitan area, marital status, and primary tumor site, histology, stage, and grade.43 Ages at diagnosis and death were expressed using mean (standard deviation [SD]) values. Because SEER reports ages 90 years or older as categorical, age 90 was used as the threshold for categorizing patient age. In Cohort 2, the ages at diagnosis of 527 patients and at death of 464 patients were capped at 90 years. For both cohorts, the Wilcoxon rank-sum test compared continuous variables, and chi-square tests compared categorical variables. Kaplan-Meier curves and log-rank tests measured OS. Survival times were calculated from the month and year of diagnosis to the month and year of death or last follow-up, whichever occurred first.
As there were more NH White patients than NH Black patients in both cohorts, we chose greedy, nearest-neighbor 1:1 PSM using age at diagnosis, median household income (Cohort 1, < $70 000 vs ≥ $70 000; Cohort 2, < $65 000 vs ≥ $65 000), metropolitan proximity (population < 250 000 vs ≥ 250 000), marital status, primary tumor site per International Classification of Diseases (ICD), Oncology 3rd edition44 codes based on ICD-10, tumor histology, tumor stage, and tumor grade. We used PSM to balance demographic and clinical covariates to evaluate whether and to what extent treatment differences had occurred between NH Black and NH White patients. After matching, absolute standardized mean differences (SMDs) were calculated to quantify the remaining imbalances; values of 0.10 or less indicated balance between the racial groups.45 We also compared hysterectomy rates between NH White and NH Black patients pre- and post-PSM. Aside from variables reported as “missing,” “no match,” “other,” “unknown,” or “unstaged,” data were complete for all patients. A 2-sided test at α = 0.05 was significant; all analyses were performed using Statistical Analysis Software (SAS) version 9.4 (SAS Institute).
Results
Cohort 1
Cohort 1 included 133 839 patients with uterine cancer (11.50% NH Black, 88.50% NH White) diagnosed between 2000 and 2017 (data not shown). Of them, 102 534 were eligible for PSM (10 960 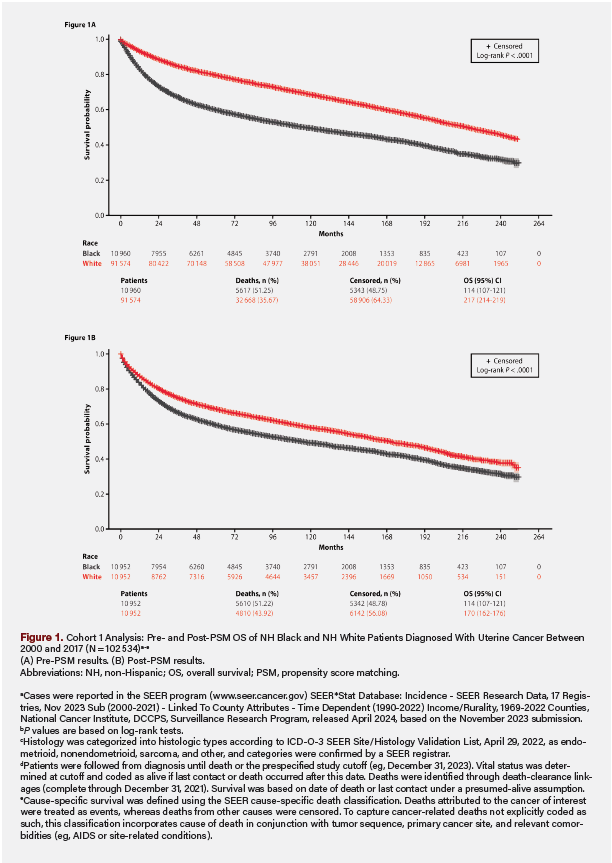 NH Black and 91 574 NH White), resulting in a 1:1-matched cohort of 21 904 patients (10 952 NH Black and 10 952 NH White). Pre-PSM, the SMDs were as high as 0.59. Higher proportions of NH Black than NH White patients had incomes less than $70 000 (64.64% vs 46.96%), lived in counties with populations of at least 250 000 (83.43% vs 77.41%), resided in the Southern US (47.56% vs 20.64%), and had never married (30.98% vs 16.15%). They also had higher rates of stage IV disease (14.81% vs 6.66%), grade 3/4 tumors (50.20% vs 25.99%), and sarcoma histology (16.31% vs 5.82%). Additionally, a larger proportion of NH Black patients were not recommended for surgery (8.74% vs 3.41%) and did not receive it (10.19% vs 4.20%), and had higher all-cause (51.25% vs 35.67%) and uterine cancer-related (34.29% vs 17.0%) mortality (all P < .001). NH Black patients also had lower first-line total hysterectomy rates across disease stages (75.64% vs 84.91%) and shorter median OS (114 months [range, 107-121 months] vs 217 months [range, 214-219 months]; all P < .001) (Figure 1). Post-PSM, demographic and tumor characteristics were balanced between the groups (all SMDs were ≤ .07). Compared with NH White patients, NH Black patients still had lower total hysterectomy rates (75.63% vs 79.45%) and shorter median OS (114 months [range, 107-121 months] vs 170 months [range, 162-176 months]) (all P < .001) (Figure 1).
NH Black and 91 574 NH White), resulting in a 1:1-matched cohort of 21 904 patients (10 952 NH Black and 10 952 NH White). Pre-PSM, the SMDs were as high as 0.59. Higher proportions of NH Black than NH White patients had incomes less than $70 000 (64.64% vs 46.96%), lived in counties with populations of at least 250 000 (83.43% vs 77.41%), resided in the Southern US (47.56% vs 20.64%), and had never married (30.98% vs 16.15%). They also had higher rates of stage IV disease (14.81% vs 6.66%), grade 3/4 tumors (50.20% vs 25.99%), and sarcoma histology (16.31% vs 5.82%). Additionally, a larger proportion of NH Black patients were not recommended for surgery (8.74% vs 3.41%) and did not receive it (10.19% vs 4.20%), and had higher all-cause (51.25% vs 35.67%) and uterine cancer-related (34.29% vs 17.0%) mortality (all P < .001). NH Black patients also had lower first-line total hysterectomy rates across disease stages (75.64% vs 84.91%) and shorter median OS (114 months [range, 107-121 months] vs 217 months [range, 214-219 months]; all P < .001) (Figure 1). Post-PSM, demographic and tumor characteristics were balanced between the groups (all SMDs were ≤ .07). Compared with NH White patients, NH Black patients still had lower total hysterectomy rates (75.63% vs 79.45%) and shorter median OS (114 months [range, 107-121 months] vs 170 months [range, 162-176 months]) (all P < .001) (Figure 1).
Cohort 2
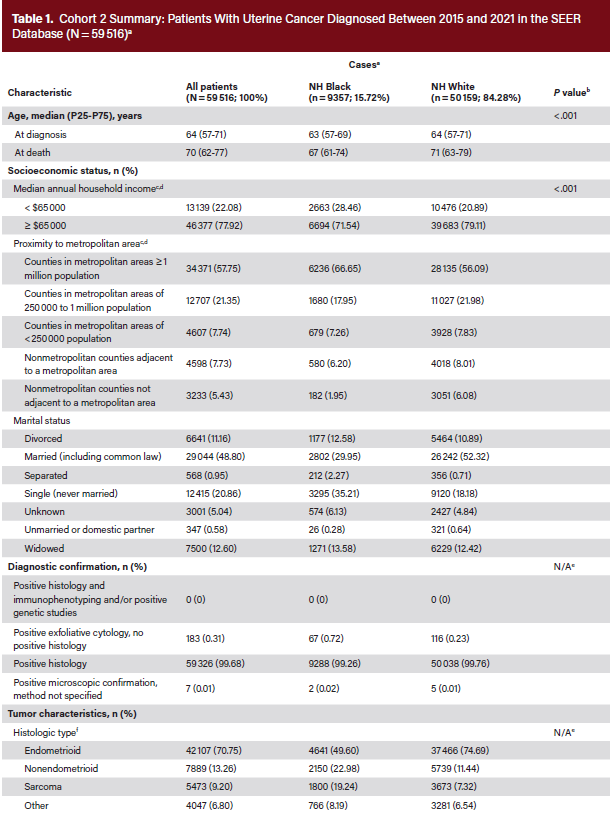
Cohort 2 included 59 566 patients who met initial eligibility criteria. After excluding 50 patients due to the coding of nonuterine tumors, 59 516 patients remained, of whom 9357 (15.72%) were NH Black and 50 159 (84.28%) were NH White (Table 1). The median (25th-75th percentiles) age at diagnosis was 63 years (range, 57-69 years) for NH Black patients and 64 years (range, 57-71 years) for NH White patients. Higher proportions of NH Black patients lived in metropolitan areas with populations of at least 1 million (66.65% vs 56.09%).
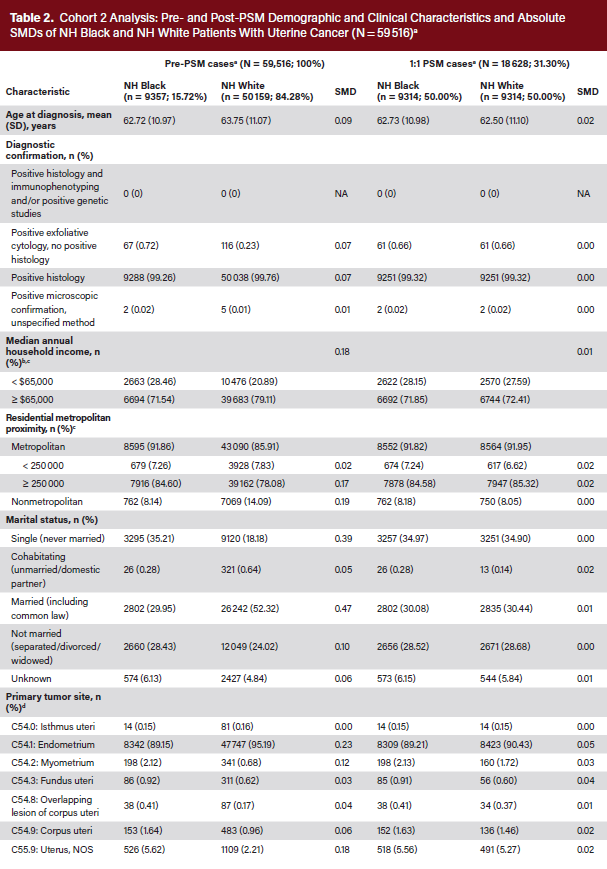
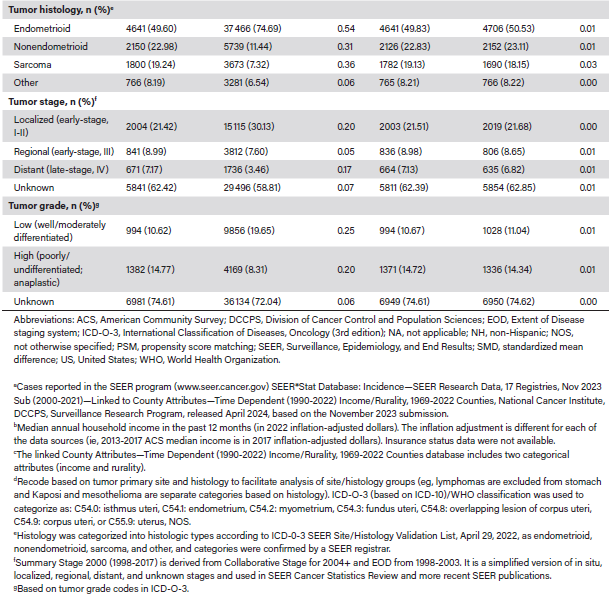
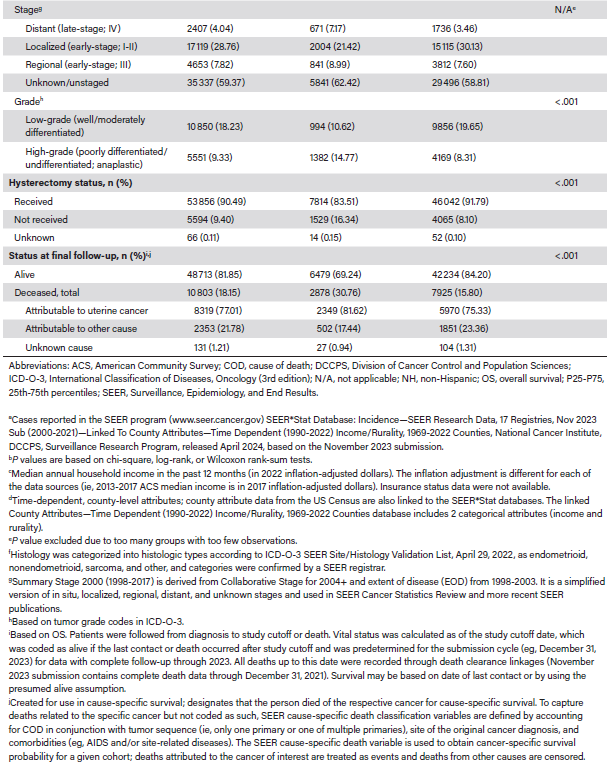
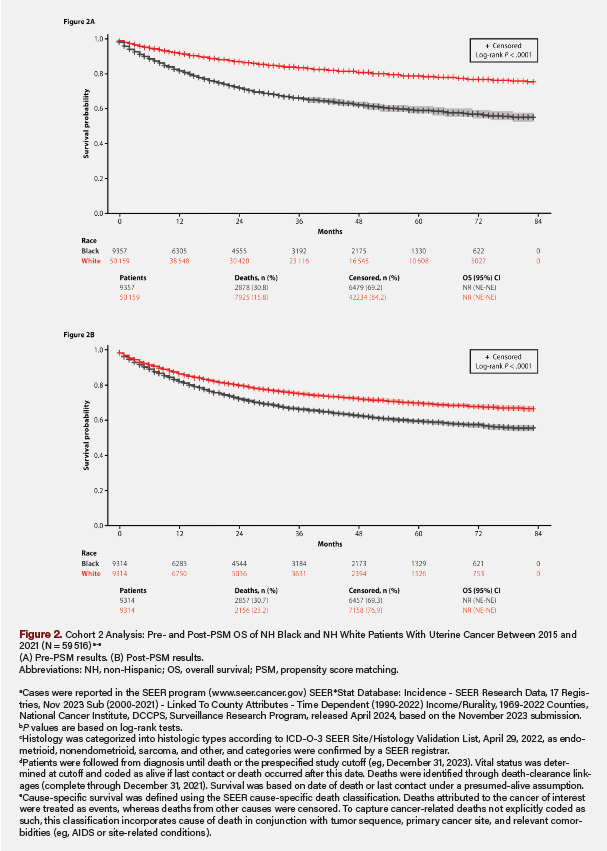 Pre-PSM, the absolute SMDs were as high as 0.54 (Table 2). The mean (SD) ages at diagnosis were 62.72 (10.97) and 63.75 (11.07) years, respectively. Higher proportions of NH Black than NH White patients lived in metropolitan areas with populations of at least 250 000 (84.60% vs 78.08%), had incomes less than $65 000 (28.46% vs 20.89%), and had never married (35.21% vs 18.18%). They also had higher rates of aggressive tumor histology (nonendometrioid, 22.98% vs 11.44%; sarcoma, 19.24% vs 7.32%), stage IV disease (7.17% vs 3.46%), and grade 3/4 tumors (14.77% vs 8.31%). Hysterectomy was not recommended for 14.24% of NH Black patients and 6.88% of NH White patients (Table 3). Additionally, 83.51% of NH Black vs 91.79% of NH White patients received a hysterectomy, of whom 72.21% vs 83.54% had total surgical removal (P < .001). By stage, total hysterectomy occurred in 84.4% of NH White vs 90.3% of NH Black patients for localized (P < .001), 74.91% vs 81.72% for regional (P < .001), and 36.96% vs 43.49% for distant (P = .004) disease, respectively. The Kaplan-Meier median OS was not reached (NR), and mortality was 30.76% for NH Black versus 15.80% for NH White patients (P < .001). NH Black patients also had lower survival probability (Figure 2) and higher risk of death (hazard ratio [HR], 2.26; 95% CI, 2.17-2.36; P < .001) (Table 3). By tumor histology, OS was NR (not estimable; [NE]-NE) for endometrioid, NR (range, 75-NE months) for nonendometrioid, 34 (range, 31-37) months for sarcoma, and NR (NE-NE) for other histologic types (Figure 3).
Pre-PSM, the absolute SMDs were as high as 0.54 (Table 2). The mean (SD) ages at diagnosis were 62.72 (10.97) and 63.75 (11.07) years, respectively. Higher proportions of NH Black than NH White patients lived in metropolitan areas with populations of at least 250 000 (84.60% vs 78.08%), had incomes less than $65 000 (28.46% vs 20.89%), and had never married (35.21% vs 18.18%). They also had higher rates of aggressive tumor histology (nonendometrioid, 22.98% vs 11.44%; sarcoma, 19.24% vs 7.32%), stage IV disease (7.17% vs 3.46%), and grade 3/4 tumors (14.77% vs 8.31%). Hysterectomy was not recommended for 14.24% of NH Black patients and 6.88% of NH White patients (Table 3). Additionally, 83.51% of NH Black vs 91.79% of NH White patients received a hysterectomy, of whom 72.21% vs 83.54% had total surgical removal (P < .001). By stage, total hysterectomy occurred in 84.4% of NH White vs 90.3% of NH Black patients for localized (P < .001), 74.91% vs 81.72% for regional (P < .001), and 36.96% vs 43.49% for distant (P = .004) disease, respectively. The Kaplan-Meier median OS was not reached (NR), and mortality was 30.76% for NH Black versus 15.80% for NH White patients (P < .001). NH Black patients also had lower survival probability (Figure 2) and higher risk of death (hazard ratio [HR], 2.26; 95% CI, 2.17-2.36; P < .001) (Table 3). By tumor histology, OS was NR (not estimable; [NE]-NE) for endometrioid, NR (range, 75-NE months) for nonendometrioid, 34 (range, 31-37) months for sarcoma, and NR (NE-NE) for other histologic types (Figure 3).
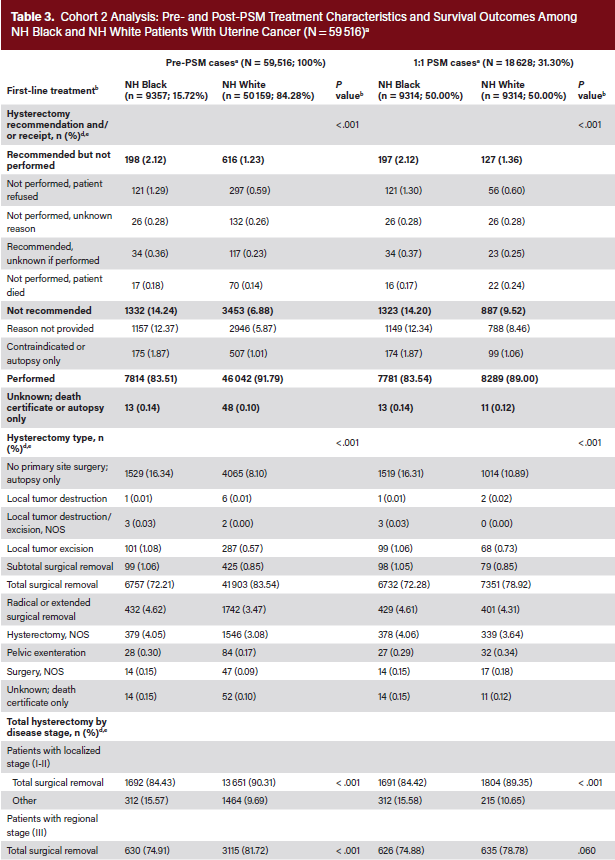
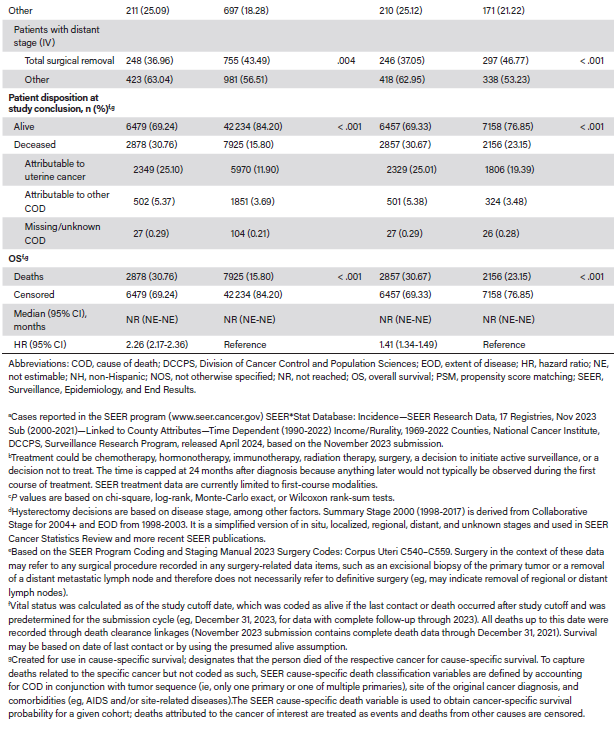
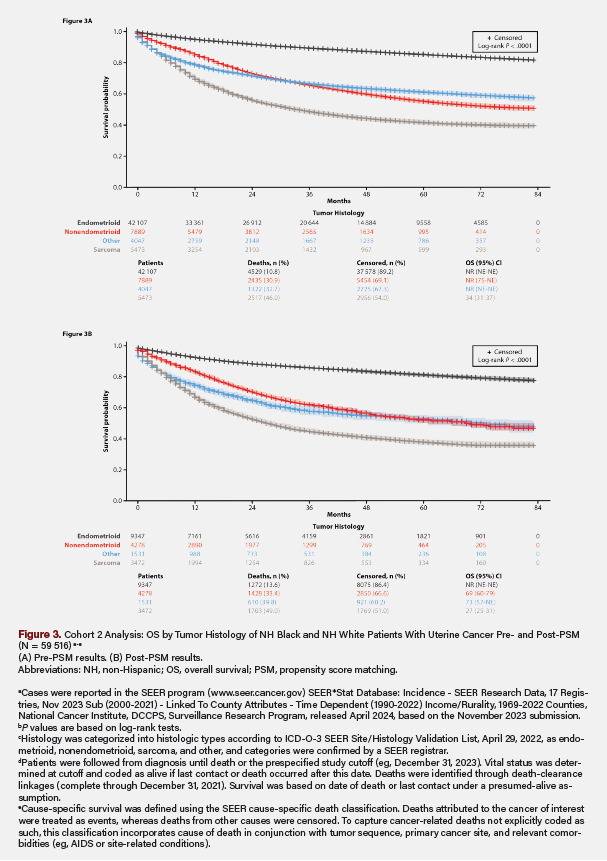 Post-PSM, 9314 NH Black patients were matched with 9314 NH White patients, with between-group SMDs ranging from 0 to 0.05 (Table 2). The proportions of NH Black and NH White patients with incomes less than $65 000, living in counties with populations of at least 250 000, and having never married were similar, as were the clinical characteristics of stage IV disease, grade 3/4 tumors, and sarcoma histology. Hysterectomy was not recommended for 14.20% of NH Black and 9.52% of NH White patients (Table 3). Additionally, 83.54% of NH Black vs 89.00% of NH White patients received a hysterectomy, of whom 72.28% vs 78.92% had total surgical removal (P < .001). By stage, total hysterectomy occurred in 84.42% of NH Black vs 89.35% of NH White patients for localized (P < .001), 74.88% vs 78.78% for regional (P = .060), and 37.05% vs 46.77% for distant (P < .001) disease, respectively. The Kaplan-Meier median OS was NR, and mortality was 30.67% for NH Black vs 23.15% for NH White patients (P < .001). NH Black patients also had lower survival probability (Figure 2) and higher risk of death (HR, 1.41; 95% CI, 1.34-1.49; P < .001) (Table 3). By tumor histology, OS was NR (NE-NE) for endometrioid, 69 months (range, 60-79 months) for nonendometrioid, 27 months (range, 25-31 months) for sarcoma, and 73 months (range, 57-NE months) for other histologic types (Figure 3).
Post-PSM, 9314 NH Black patients were matched with 9314 NH White patients, with between-group SMDs ranging from 0 to 0.05 (Table 2). The proportions of NH Black and NH White patients with incomes less than $65 000, living in counties with populations of at least 250 000, and having never married were similar, as were the clinical characteristics of stage IV disease, grade 3/4 tumors, and sarcoma histology. Hysterectomy was not recommended for 14.20% of NH Black and 9.52% of NH White patients (Table 3). Additionally, 83.54% of NH Black vs 89.00% of NH White patients received a hysterectomy, of whom 72.28% vs 78.92% had total surgical removal (P < .001). By stage, total hysterectomy occurred in 84.42% of NH Black vs 89.35% of NH White patients for localized (P < .001), 74.88% vs 78.78% for regional (P = .060), and 37.05% vs 46.77% for distant (P < .001) disease, respectively. The Kaplan-Meier median OS was NR, and mortality was 30.67% for NH Black vs 23.15% for NH White patients (P < .001). NH Black patients also had lower survival probability (Figure 2) and higher risk of death (HR, 1.41; 95% CI, 1.34-1.49; P < .001) (Table 3). By tumor histology, OS was NR (NE-NE) for endometrioid, 69 months (range, 60-79 months) for nonendometrioid, 27 months (range, 25-31 months) for sarcoma, and 73 months (range, 57-NE months) for other histologic types (Figure 3).
Discussion
Despite balancing baseline characteristics, disparities in treatment, mortality, and survival between NH Black and NH White patients persisted. Compared with the NH White patients, NH Black patients had lower rates of surgical recommendation and receipt, higher uterine cancer–related mortality, and shorter median OS. Although disease stage-specific surgical recommendations contributed to the lower hysterectomy rates among NH Black patients, racial disparities remained across all stages. From a clinical pathway perspective, these findings highlight a critical gap in the consistent application of evidence-based treatment protocols. Clinical pathways are designed to standardize care, reduce unwarranted variation, and improve outcomes; however, when pathways are not equitably implemented—or when they fail to account for SDOH—racial disparities may persist or even widen. Thus, clinical pathways in uterine cancer should incorporate equity-focused decision points, ensuring that recommendations, such as total hysterectomy, are guided by standardized criteria rather than influenced by implicit bias among NH White providers, racially differential patient–provider communication, or systemic health care barriers. Additionally, clinical pathways should embed social risk screening tools to identify patients at risk of delayed or suboptimal care due to socioeconomic factors; real-time monitoring and feedback mechanisms to track disparities in treatment delivery and outcomes across racial and ethnic groups will be instrumental. Pathway development should also engage patients and stakeholders from the communities they intend to serve to ensure that diverse needs, experiences, and tumor biology are reflected in care planning.
In this study, balancing baseline characteristics reduced—but did not eliminate—differences in first-line hysterectomy recommendation, receipt, type, and OS. Differences in total hysterectomy rates were most reduced between NH Black and NH White patients with stage IV disease. In Cohort 2, NH Black patients died younger than NH White patients (median [25th-75th percentiles] age, 67 [61-74] years vs 71 [63-79] years). Most deaths (77.01%) were related to uterine cancer, with higher proportions of NH White patients (23.36%) than NH Black (17.44%) patients dying from other causes, possibly due to receiving more guideline-concordant care.5 Patient treatment refusal contributed minimally to the racial disparity in uterine cancer survival, highlighting the need to understand why they refused treatment and investigate other factors, especially SDOH.46 In Cohort 1, median survival post-PSM for NH White patients was 4.5 years longer than for NH Black patients. In Cohort 2, mortality risk was higher for NH Black patients vs NH White patients (pre-PSM HR, 2.26; post-PSM HR, 1.41). Although insurance data were lacking, more NH Black patients had incomes below the median, lived in high-density urban areas, and had never married, contributing to poorer outcomes. Socioeconomic and structural factors at the ZIP‑code level are more strongly associated with access to high‑quality cancer care and survival than are individual genetic characteristics.47
Study Strengths and Limitations
This study benefited from the large cohort of SEER data, which is geographically diverse and represents the US population.48 The SEER data span several decades, are updated annually, and are widely published. Another strength is the study’s statistical methodology, which matched NH Black and NH White patients using 1:1 greedy, nearest-neighbor matching without replacement. Prior studies that did not apply PSM have reported contradictory findings, emphasizing its analytic value in producing valid, reliable, and generalizable data.49 The methodologic utility of PSM is further demonstrated in our study, wherein treatment and outcomes differed substantially and significantly before and after PSM. As PSM helps reduce confounding bias, its uptake is expanding in surgical outcome evaluations leveraging databases such as SEER.49
Limitations of this study include missing data due to postal address gaps or privacy restrictions, and the use of county-level rather than patient-level median household income reported in SEER. We averaged the 2015-2021 US Census data to categorize income levels; although these do not account for ancestry, this has minimal impact as NH Black households consistently have lower incomes than NH White households. Additionally, alignment between SEER- and Medicare claims-derived diagnosis dates for uterine cancer may vary due to data collection methods, reporting practices, and timing. Our findings were further limited by SDOH, treatment sequencing, insurance, and comorbidities not being analyzed, and biases from unmeasured treatment decision-making factors and interpretation by providers.50,51 We also could not study clinical covariates affecting surgical candidacy, approach, complications, or recovery, and the lack of systemic therapy data may have underestimated neoadjuvant and postsurgical treatment frequency.50,52 Moreover, survival differences among patients who did or did not undergo hysterectomy cannot be inferred without controlling for surgical recommendation factors.51 Classifying cause of death can also be challenging,52 with an estimated 20% to 30% of death certificates being incomplete or inaccurate.53 Misclassification bias was mitigated by accounting for tumor sequence and site-related diseases.52 We could not adjust for all potential confounders given these limitations.
Conclusion
Patient race may influence clinical research, guidelines, and treatment decisions in ways that clinical pathways have not yet anticipated or addressed.47 Racial disparities in hysterectomy recommendation and OS outcomes suggest either a lack of adherence to published guidelines or absence of those guidelines in institutional disease-specific clinical pathways that are monitored for compliance. Implementing or refining evidence-based pathways may help ensure equitable treatment decisions, such as consistent criteria for recommending hysterectomy regardless of race or socioeconomic status. Developing quality metrics within clinical pathways that track disparities in treatment recommendations and outcomes may enable real-time monitoring and timely intervention; these pathways should incorporate SDOH (eg, income, urban density) to personalize care and address barriers. For example, real-time equity dashboards created by diverse, interdisciplinary teams that maintain continuously updated SDOH data are ideal for monitoring patient outcomes, guiding policy, and reducing inequities.54 Notably, effective patient navigation programs that address barriers faced by underserved women within a given practice can serve as an essential link between patients and the health care system,55 alongside the use of clinician decision support tools.56 Routine electronic patient-reported outcome monitoring with clinician alerts improves quality of life, reduces acute care use, extends time on treatment, and increases OS.57 Models of care should consider social risk and enhance navigation, service integration, surveillance data, and data integration.58 By considering these factors, health care systems can move toward more equitable, data-driven, and patient-centered care, which not only aligns with the goals of clinical pathway implementation but also addresses the broader imperative of reducing racial disparities in cancer outcomes.
This article has supplementary material, which can be accessed here.
Key Clinical Summary
- Surveillance, Epidemiology, and End Results (SEER) analysis found significant racial disparities in uterine cancer treatment and survival, with NH Black patients receiving fewer hysterectomies and experiencing worse overall survival compared with matched NH White patients.
- After propensity-score matching, Black vs White disparities in uterine cancer outcomes persisted, with median overall survival of 114 vs 170 months, mortality rates of 30.67% vs 23.15%, and a death hazard ratio of 1.41 (95% CI, 1.34-1.49).
- Equity-focused clinical pathways may help reduce uterine cancer disparities by standardizing surgical recommendation criteria, integrating social determinants of health screening, and implementing disparity monitoring systems to trigger patient navigation and targeted interventions.
Clinical Pathway Category: Outcome Measurements
This study supports the clinical pathway category of outcome measurements by identifying measurable racial disparities in hysterectomy rates and overall survival among uterine cancer patients using robust population data. Its findings reinforce the need for equity-focused, evidence-based pathways with real-time monitoring to standardize care, reduce treatment variation, and improve oncology outcomes.
Author Information
Authors: Alexandrina Balanean, MPH, MWC1; Yolaine Jeune-Smith, PhD2; Parisa Asgarisabet, PhD, MBA1; Camryn Craig, BS1; Harlen Hays, MPH1; JaLyna Laney, RN1; Ajeet Gajra, MD, MBBS, FACP3; Bruce Feinberg, DO1
Affiliations: 1Cardinal Health Inc, Dublin, OH; 2Trinity Medical LLC, Chicago, IL; 3Hematology-Oncology Associates of CNY, East Syracuse, NY
Contributions:
Alexandrina Balanean, MPH, MWC:
- Conception and design; acquisition, analysis, and interpretation of data; drafting of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
- Had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; conducted and is responsible for the data analysis.
Yolaine Jeune-Smith, PhD:
- Conception and design; interpretation of data; editorial review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Parisa Asgarisabet, PhD, MBA:
- Design; analysis and interpretation of data; statistical and editorial review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
- Had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; conducted and is responsible for the data analysis.
Camryn Craig, BS:
- Design; analysis and interpretation of data; statistical and editorial review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
- Had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; conducted and is responsible for the data analysis.
Harlen Hays, MPH:
- Design; analysis and interpretation of data; statistical review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
JaLyna Laney, RN:
- Design; analysis and interpretation of data; clinical review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ajeet Gajra, MD, MBBS, FACP:
- Design; analysis and interpretation of data; clinical review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Bruce Feinberg, DO:
- Design; analysis and interpretation of data; clinical review of the manuscript; final approval of the version to be published; and accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Address correspondence to:
Alexandrina Balanean, MPH, MWC
Lead Publication Scientist, Strategic Research
Specialty Networks, a Cardinal Health Company
Chicago, IL (remote)
773.610.4520 (US Central Time)
Email: alexandrina.balanean@cardinalhealth.com
Disclosures: All authors conducted this research as part of their employment at Cardinal Health; no external funding was sought or received. A.B., P.A., and C.C. are salaried employees of Cardinal Health. H.H., J.L., and B.F. are salaried employees of and own stock in Cardinal Health. Y.J.-S. was a salaried employee of and owned stock in Cardinal Health at the time of this study. She is currently employed by Trinity Medical, LLC. A.G. was a salaried employee of and owned stock in Cardinal Health at the time of this study. He is currently employed by Hematology-Oncology Associates of CNY.
Funders: All authors conducted this research as part of their employment at Cardinal Health; no external funding was sought or received.
Acknowledgment: Microsoft Copilot was used for assistance with research and throughout the manuscript for paraphrasing and word count reduction; all output was checked for accuracy and edited as necessary by the senior author.














