Bispecific Antibody Treatments for Multiple Myeloma: A Hybrid Academic-Community Cancer Center’s Outpatient Step-up Dosing Care Model
Abstract
Teclistamab and talquetamab are bispecific antibody (BsAb) therapies approved for relapsed/refractory multiple myeloma (RRMM). It is important to understand how practices evolve BsAb step-up dosing (SUD) in outpatient care models that improve patient access while ensuring safety. A qualitative interview was conducted with hematologists at Atrium Health Wake Forest Baptist Comprehensive Cancer Center to understand their processes and experiences with administering teclistamab/talquetamab SUD in an outpatient setting. Atrium Health hematologists used Hospital-at-Home (HAH) programs to administer outpatient SUD, thereby reducing health care burden and increasing access to these therapies. Patient eligibility for outpatient SUD was determined by presence of caregiver support, proximity to Atrium Health’s main campus, and clinical characteristics. Hematologists and HAH staff jointly work to monitor vitals, and HAH paramedics conduct home visits between step-up doses. Atrium Health hematologists developed adverse event management processes, including guidance for at-home medications, nurse triage, and emergency department protocols. These findings showed that outpatient SUD for teclistamab/talquetamab is feasible and can be safely implemented for selected patients with RRMM. This care model may serve as an example to inform the development of outpatient SUD programs at other centers.
Introduction
The recent US Food and Drug Administration (FDA) approval of numerous immunotherapies has drastically improved the treatment landscape for multiple myeloma (MM). Bispecific antibody therapies (BsAbs) have become a new standard of care for patients with relapsed or refractory multiple myeloma (RRMM). Approved in October 2022, teclistamab is the first approved bispecific B-cell maturation antigen (BCMA) directed CD3 T-cell engager for the treatment of RRMM in the US. Before teclistamab was approved, the median overall survival (OS) for patients with RRMM after triple-class exposure (ie, exposure to a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 antibody) was estimated at 12 months, with median progression-free survival (PFS) estimated at 4 months and an overall response rate (ORR) of 40%.1 In the long-term analysis of the teclistamab clinical trial MajesTEC-1, the median OS was 22.2 months and median PFS was 11.4 months during a 30.4-month median follow-up.2 Patients receiving teclistamab achieved an ORR of 63%, where 46.1% achieved complete response or better, and the median duration of response was 24 months for all responders.2
In August 2023, 2 additional BsAbs were approved by the FDA for adult patients who have received at least 4 prior lines of therapy and triple-class exposure for RRMM: talquetamab (bispecific GPRC5D-directed CD3 T-cell engager) and elranatamab (bispecific BCMA-directed CD3 T-cell engager).3,4
BsAbs have a specific adverse event (AE) profile and may cause cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). To reduce the risk and severity of these AEs, the FDA advises that patients be hospitalized during step-up dosing (SUD) to gradually administer the initial doses until the target therapeutic dose is reached. However, hospitalization presents a burden on health care systems, straining limited resources, such as hospital beds and medical personnel. Further, hospitalization requirements may limit access to teclistamab among patients living in rural areas. To overcome these challenges, some health care practices have shifted to an outpatient SUD model.
Atrium Health is an integrated community health care system that includes the Levine Cancer Institute in central Charlotte, North Carolina, along with over 25 other cancer care locations. After the initial approval of teclistamab, hematologists at Atrium Health Wake Forest Baptist Comprehensive Cancer Center (hereafter “Atrium Health”) recognized the need to alleviate the health care resource burden associated with inpatient SUD and began exploring an outpatient SUD model.5 Atrium Health used its Hospital-at-Home (HAH) program, which was developed in March 2020 at the onset of the COVID-19 pandemic, to address hospital overcrowding to monitor patients undergoing outpatient SUD. The HAH program was built on an existing hospital transition program and implemented to provide at-home health services to patients with SARS-CoV-2.6,7 The program included both virtual and in-person services to high-acuity patients who would otherwise require hospitalization, including daily virtual physician visits, vital sign monitoring, and the ability to perform lab checks and give intravenous medications and oxygen as needed. By September 2020, Atrium Health had permanently staffed the HAH program and expanded it to patients with non-COVID-19 diagnoses.
Early in their experience with outpatient SUD administration, Atrium Health experienced challenges, including identifying appropriate patient candidates, ensuring dedicated staff for 24-hour remote monitoring of treatment-related AEs, and navigating costs and reimbursement issues.8 Atrium Health hematologists overcame these challenges using existing resources to launch a remote monitoring program for administering teclistamab and talquetamab SUD in an outpatient setting.
This study describes the Atrium Health care model for outpatient SUD administration of teclistamab and talquetamab and AE management, with the goal of informing other health care practices seeking to develop outpatient SUD care models. A semi-structured interview format was used to elicit information on processes and experiences with initiating outpatient teclistamab/talquetamab SUD for RRMM patients.
Methods
A semi-structured in-depth interview was conducted with 2 hematologists who spearheaded the development of the teclistamab/talquetamab outpatient SUD program at Atrium Health. Interview audio recordings were transcribed verbatim and imported into a qualitative data analysis software program. An a priori coding scheme was developed, with emerging themes identified using the constant comparative method.9 Overarching themes included Atrium Health’s teclistamab/talquetamab outpatient SUD model, AE management strategies, and perspectives on the future evolution of their care model. The transcript was coded independently by two members of the research team. The results from this qualitative study are summarized descriptively by themes.
Results
Outpatient SUD Model
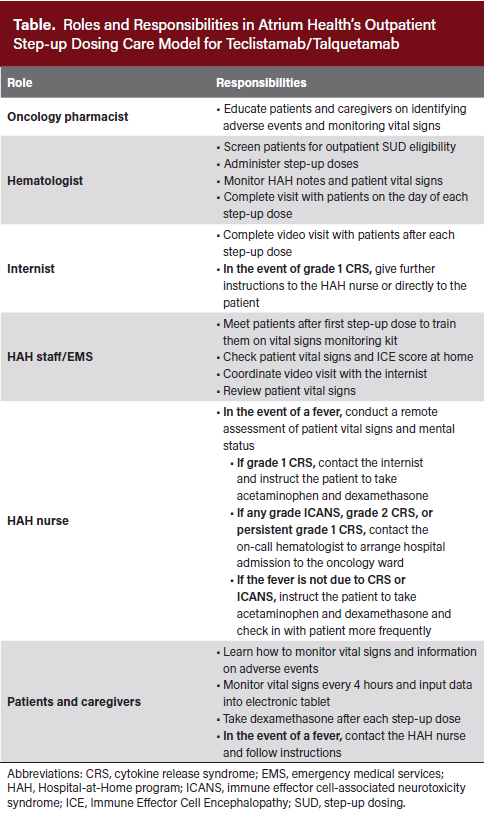 As the acuity of COVID-19 cases declined, Atrium Health hematologists saw an opportunity to use HAH for outpatient SUD of teclistamab and talquetamab. With increased capacity, HAH staff, including nurses, internists, and paramedics, completed a training session and received written guidelines on triaging patients experiencing CRS or ICANS (Table). To reduce the risk and ensure timely management of AEs, eligibility for outpatient SUD required patients to have a 24-hour at-home caregiver who could monitor symptoms during the SUD period, help with medications, and coordinate transportation. Patients were also required to stay within state lines (eg, within North Carolina) and within an approximate 1-hour radius of the Levine Cancer Institute (HAH radius). Additional considerations for patient selection include a score of 2 or greater on the Eastern Cooperative Oncology Group Performance Status Scale, creatinine clearance of at least 30 mL/min, and the absence of neutropenia, rapidly progressing disease, high tumor burden, or significantly elevated baseline inflammatory markers. However, eligibility decisions for outpatient SUD were ultimately at the treating physician’s discretion.
As the acuity of COVID-19 cases declined, Atrium Health hematologists saw an opportunity to use HAH for outpatient SUD of teclistamab and talquetamab. With increased capacity, HAH staff, including nurses, internists, and paramedics, completed a training session and received written guidelines on triaging patients experiencing CRS or ICANS (Table). To reduce the risk and ensure timely management of AEs, eligibility for outpatient SUD required patients to have a 24-hour at-home caregiver who could monitor symptoms during the SUD period, help with medications, and coordinate transportation. Patients were also required to stay within state lines (eg, within North Carolina) and within an approximate 1-hour radius of the Levine Cancer Institute (HAH radius). Additional considerations for patient selection include a score of 2 or greater on the Eastern Cooperative Oncology Group Performance Status Scale, creatinine clearance of at least 30 mL/min, and the absence of neutropenia, rapidly progressing disease, high tumor burden, or significantly elevated baseline inflammatory markers. However, eligibility decisions for outpatient SUD were ultimately at the treating physician’s discretion.
Below, we outline the operational workflow of the outpatient SUD program as implemented at Atrium Health. Before beginning treatment, an oncology pharmacist educates eligible patients on teclistamab/talquetamab, symptoms of treatment-related AEs, logistics of the HAH program, and at-home AE monitoring. Patients are instructed to contact the on-call HAH nurse in the event of fever or malaise. The pharmacist gives the patient a written handout summarizing this information.
Atrium Health uses a 3-day interval schedule for step-up doses (ie, days 1, 4, and 7) with flexibility to ensure all doses are taken during the week when they would otherwise fall on a weekend. Before or shortly after the patient receives an initial teclistamab/talquetamab step-up dose in the outpatient infusion center, an HAH paramedic meets the patient and caregiver at home to provide a monitoring kit. The kit includes a thermometer, blood pressure cuff, wearable device for continuous pulse-oximetry, heart rate, and respiratory rate monitoring, and an electronic tablet for recording vital signs for the HAH team to review.
Atrium Health partnered with Current Health, a telehealth platform, to enable virtual monitoring of patient vital signs and 24/7 communication with the nursing staff through the provided tablet. The paramedic trains the patient and caregiver on proper use of the Current Health system and all devices of the monitoring kit. Patients and/or caregivers check temperature and blood pressure every 4 hours while awake (and more frequently if they feel febrile or unwell) and enter the details into the tablet. The wearable device continuously monitors the patient’s vital signs, including blood oxygen saturation, respiratory rate, and heart rate. HAH staff remotely monitor vital signs every 4 hours via a patient dashboard and receive alerts for any significant abnormalities. HAH staff also educate patients and caregivers on the signs and symptoms of potential AEs and advise them to contact the on-call HAH nurses with any concerns and questions. The HAH staff can escalate questions to the hematologists if any complexities arise.
In our study, an initial assessment of the HAH approach showed that none of the 18 patients who received outpatient SUD of either teclistamab or talquetamab with HAH monitoring developed ICANS or high-grade (grade 3 or higher) CRS.5 Eleven patients developed CRS, of which 4 developed grade 2 CRS and required hospital admission. All CRS cases resolved, and patients continued to receive outpatient SUD. The 18 patients in this cohort were hospitalized for a total of 19 days compared with an estimated 116 total days that would have been necessary under traditional inpatient SUD monitoring.5
Management of AEs
For management of AEs, the patient is provided with acetaminophen, dexamethasone, acyclovir, and trimethoprim/sulfamethoxazole to take home. The patient is instructed to take 8 mg of prophylactic dexamethasone the day after each SUD to mitigate the onset and severity of CRS and ICANS. Acetaminophen should be taken as-needed as instructed by the HAH staff. Patients are seen by a hematologist either in-person at the clinic or via video visit on the days of teclistamab/talquetamab SUD administration.
On days when the patient does not receive teclistamab/talquetamab SUD treatment, a paramedic visits the patient at home to perform a basic physical examination, record vital signs, assess their Immune Effector Cell Encephalopathy (ICE) score, and coordinate a virtual visit with an HAH internist. On the days between SUD, patients are billed only for the virtual HAH visit. Vital signs and HAH visit notes are uploaded to the patient’s electronic medical record and are accessible to Atrium Health hematologists for review between the days of SUD administration and for future reference. If no major concerns arise between doses, Atrium Health hematologists review this information on the day of the next SUD.
In the event of a fever, patients first contact the on-call HAH nurse, who will guide the patient through a focused remote assessment including vital signs and ICE score. If grade 1 CRS is confirmed, the nurse contacts the HAH internist for further instructions and to initiate the fever protocol, which includes acetaminophen and an additional 8 mg of dexamethasone if the fever persists or recurs. In the event of a fever, the nurse checks in with patients more frequently to ensure that it is improving. Per the protocol, the HAH internist virtually instructs the patient to check blood pressure and blood oxygen saturation more frequently when they feel febrile to evaluate adequate response to treatments and monitor progression to grade 2 CRS.
If a patient progresses to grade 2 CRS or has persistent grade 1 CRS that does not respond to at-home interventions, hospital admission is arranged for further management with tocilizumab and other supportive care measures, which may include intravenous fluids, supplemental oxygen, and evaluation for infection. Any grade of ICANS warrants hospital admission for treatment and further monitoring. The HAH nurse/internist contacts the on-call hematologist to ensure the patient is admitted to the oncology ward. If the patient is admitted overnight to the emergency department (ED) first, HAH staff notifies the on-call hematologist, who advises the ED providers on patient management and typically recommends administering tocilizumab per the patient’s care plan. While awaiting consultation, ED providers begin basic stabilization and obtain imaging and laboratory cultures. HAH staff may also contact the patient’s primary hematologist (during business hours) or the on-call inpatient hematology attending physician (24/7 access) for additional guidance on AE management.
Perceptions of Patient Response to Outpatient SUD Program
Hematologists report that patients seem enthusiastic about the opportunity to receive teclistamab/talquetamab in an outpatient setting, with some patients temporarily moving closer to the hospital to meet outpatient SUD requirements, avoiding an inpatient stay. Informal patient feedback of the teclistamab/talquetamab outpatient SUD program has been positive, with hematologists noting that the patients appreciate the communication between the hematology team and HAH staff.
Outpatient SUD Implementation Challenges and Considerations
The HAH outpatient SUD program is an adaptable model that can expand to include other BsAbs for the treatment of RRMM. However, the level of monitoring required for outpatient SUD may pose challenges for institutions and community oncology practices with limited resources. Alternative measures to facilitate outpatient SUD include administering prophylactic tocilizumab and using remote patient monitoring (RPM). Prophylactic tocilizumab has been shown to reduce the incidence and severity of CRS in patients treated with BsAbs for MM.10 The broader use of prophylactic tocilizumab can enable practices with limited resources to safely administer outpatient SUD. RPM offers another option for practices that cannot implement an at-home health service.11 An institution could implement a BsAb administration model in which on-call providers review the vitals virtually and respond to patient calls. Alternatively, some producers of RPM technology have begun to offer an additional service in which they provide the monitoring and triage of the RPM results for patients receiving outpatient transplant and cellular therapy treatments.
Quality Improvement and Expansion of Outpatient SUD
Atrium Health hematologists regularly review processes and outcomes through the HAH program to support continuous quality improvement. Initially, dexamethasone was administered only prophylactically; however, after reviewing data on hospital admissions, the outpatient SUD protocol was revised to include dexamethasone doses as required in addition to prophylactic use.
At the time of the interview, BsAbs were typically initiated for patients at the infusion center of the Atrium Health Levine Cancer Institute in Charlotte, NC. After successfully completing teclistamab SUD, patients transitioned to the clinic closest to their home for ongoing treatment. As of May 2025, Atrium Health began outpatient SUD administration for patients who live within the 1-hour HAH radius at select regional sites within North Carolina (eg, Concord, Huntersville, and Monroe) and plans to expand to more sites in the future. A new outpatient transplant and cell therapy unit is available for patients with persistent grade 1 or 2 CRS to receive tocilizumab and potentially return home for continued HAH monitoring instead of being admitted.
In addition, a recent retrospective analysis from the Atrium Health Levine Cancer Institute comparing outpatient and inpatient SUD outcomes found similar rates of CRS and ICANS incidence between both cohorts, and all instances of CRS and ICANS in the outpatient cohort were low grade.12 The outpatient cohort also had fewer hospitalization days, indicating lower health care resource utilization. These findings demonstrate that BsAb SUD can be safely administered in an outpatient model that reduces the burden on health care resources.
Conclusion
This study provides a real-world model of outpatient SUD administration for teclistamab/talquetamab, demonstrating that SUD for teclistamab/talquetamab can be safely administered with AE management in an outpatient setting for eligible patients. Outpatient SUD models allow for flexibility in treatment administration, thereby increasing patient access to expanded treatment options without needing scheduled hospitalization.
Atrium Health hematologists leveraged the existing health care system infrastructure to support teclistamab/talquetamab initiation and used existing resources to develop a program tailored to meet the needs of their practice and patients. As other health care systems look to adopt BsAb therapies for their patients with RRMM, this care model can help inform best practices for developing outpatient SUD models that ensure quality care.
Key Clinical Summary
- Atrium Health’s Hospital-at-Home program supports outpatient step-up dosing (SUD) for bispecific antibodies (BsAbs) teclistamab and talquetamab in patients with multiple myeloma, providing continuous remote vital sign monitoring, scheduled home paramedic visits, and 24/7 nurse access between step-up doses.
- The outpatient SUD model for bispecific antibody therapy defines patient eligibility criteria and outlines standardized protocols for adverse event monitoring, at-home management of side effects such as cytokine release syndrome (CRS), and clear thresholds for hospital admission.
- Atrium Health’s outpatient oncology care model demonstrates that SUD for bispecific antibodies in multiple myeloma can be safely and feasibly delivered outside the hospital, reducing the need for routine inpatient hospitalization while maintaining high-quality cancer care.
Clinical Pathway Category: Infrastructure & Innovation
This study advances the infrastructure & innovation clinical pathway category by outlining a structured Hospital-at-Home outpatient model for bispecific antibody step-up dosing that aligns with evidence-based safety standards and defined care protocols. By embedding clear eligibility criteria, monitoring processes, and adverse event pathways into practice, it offers a scalable framework that enhances oncology care delivery and supports integration into clinical pathways.
Author Information
Authors: Christopher J. Ferreri, MD1; Peter M. Voorhees, MD1; Marvin Knight 2; Meaghan Roach, MPH2,3; Richard Murphy, BA2,3; Amal Jamaleddine, BA2,3; Nicole Bariahtaris, BA2; Cindy Varga, MD1
Affiliations: 1Atrium Health Levine Cancer Institute, Wake Forest University School of Medicine, Charlotte, NC; 2Atrium Health Hospital at Home, Charlotte, NC; 3Precision AQ, New York, NY
Address correspondence to:
Christopher J. Ferreri, MD
Atrium Health Levine Cancer Institute,
Wake Forest University School of Medicine,
1021 Morehead Medical Drive, Charlotte, NC 28204
Christopher.Ferreri@atriumhealth.org
Disclosures: C.F. declares consultancy for Johnson and Johnson, and stock ownership for Affimed. P.V. declares consultancy for Abbvie, AstraZeneca, BMS, Karyopharm Therapeutics, Snofi, Regeneron, Johnson and Johnson, GSK, and Kite Pharma. C.V. declares speaker honoraria for Johnson and Johnson, and research for K36 Therapeutics, Arcellx and Kite Pharma. M.K. declares no non-financial or commercial, proprietary, or financial interest to disclose. R.M., A.J., and N.B. are employees of Precision AQ. M.R. is a former employee of Precision AQ.
References
1. Dhanasiri S, Hollier-Hann G, Stothard C, Dhanda DS, Davies FE, Rodriguez-Otero P. Treatment patterns and outcomes in triple-class exposed patients with relapsed and refractory multiple myeloma: findings from the multinational ITEMISE study. Clin Ther. 2021;43(11):1983-1996.e3. doi:10.1016/j.clinthera.2021.09.013
2. Garfall A, Nooka A, van de Donk NWCJ, et al. Long-term follow-up from the phase 1/2 MajesTEC-1 trial of teclistamab in patients with relapsed/refractory multiple myeloma. J Clin Oncol. 2024;42(Suppl 16):7540. doi:10.1200/JCO.2024.42.16_suppl.7540
3. FDA grants accelerated approval to elranatamab-bcmm for multiple myeloma. US Food and Drug Administration. 2023. Accessed January 31, 2026. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-elranatamab-bcmm-multiple-myeloma
4. FDA grants accelerated approval to talquetamab-tgvs for relapsed or refractory multiple myeloma. US Food and Drug Administration. 2023. Accessed January 31, 2026. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-talquetamab-tgvs-relapsed-or-refractory-multiple-myeloma
5. Varga C, Ahmed F, Knight M, et al. Prophylactic dexamethasone and remote monitoring for patients with relapsed refractory multiple myeloma (RRMM) receiving bispecific antibodies (BsAb): experience at a single institution. Blood. 2024;144:4720. doi:10.1182/blood-2024-204361
6. Sitammagari K, Murphy S, Kowalkowski M, et al. Insights from rapid deployment of a "virtual hospital" as standard care during the COVID-19 pandemic. Ann Intern Med. 2021;174(2):192-199. doi:10.7326/M20-4076
7. Raths D. How Atrium Health scaled up hospital at home. Healthcare Innovation. February 12, 2024. Accessed January 31, 2026. https://www.hcinnovationgroup.com/population-health-management/home-based-care/article/53096226/how-atrium-health-scaled-up-hospital-at-home
8. Derman BA, Roach M, Lin D, et al. Panel interview of oncology practices with emergent experience of teclistamab in the real world: the TecPIONEER study. Curr Med Res Opin. 2024;40(6):1053-1058. doi:10.1080/03007995.2024.2352856
9. Glaser BG, Strauss AL. The Discovery of Grounded Theory: Strategies for Qualitative Research. Transaction Publishers; 2009.
10. Kowalski A, Lykon J, Diamond B, et al. Tocilizumab prophylaxis for patients with multiple myeloma treated with bispecific antibodies. Blood Adv. 2025;9(19):4979-4986. doi:10.1182/bloodadvances.2025016911
11. Rajeeve S, Wilkes M, Zahradka N, et al. Effectiveness of remote patient monitoring in enabling outpatient step-up dosing for bispecifics at a large academic cancer center in the USA. Blood. 2025;146(suppl 1):2751. doi:10.1182/blood-2025-2751
12. Ferreri CJ, Knight M, Davis D, et al. Prophylactic dexamethasone (dex) with outpatient step-up dosing (SUD) of bispecific antibodies (BsAb) in multiple myeloma (MM) vs standard of care (SOC) inpatient observation. J Clin Oncol. 2025;43(suppl 16):e19506. doi:10.1200/JCO.2025.43.16_suppl.e19506














