The Impact of Clinical Presentation on One-Year Major Adverse Cardiac Events in Patients With Angiographically Defined Moderate to Severe Calcified Coronary Artery Disease
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/26.00022. Epub April 13, 2026.
Abstract
Objectives. The authors aimed to investigate the impact of clinical presentation on 1-year major adverse cardiac events (MACE) in patients with angiographically defined moderate or severe calcified coronary artery disease.
Methods. The authors conducted a retrospective, single-center cohort study in patients with moderate to severe coronary calcification who underwent percutaneous coronary intervention (PCI) between June 2020 and July 2021 with either chronic coronary syndrome (CCS) or acute coronary syndrome (ACS). The primary endpoint was 1-year MACE, a composite of all-cause mortality, myocardial infarction (MI), and ischemia-driven revascularization. Cox proportional hazards regression was used.
Results. Seven hundred twenty-four patients were included: 420 patients with ACS and 304 patients with CCS. The median age was 72.0 years. There were relatively more men in the CCS cohort (76.0% vs 66.2%, P = .005). Procedure time was longer in the context of CCS (88 minutes [25th-75th percentile: 56-104] vs 78 minutes [66-121], P < .001). Application of atherectomy and lithotripsy was similar in both cohorts. No significant difference was observed in the cumulative incidence of MACE between the ACS (15.0%) and CCS (16.4%) groups (HR, 0.94; 95% CI, 0.65-1.36; P = .74). Secondary outcomes, such as all-cause mortality, MI, and ischemia-driven revascularization, did not result in statistical significance between the groups.
Conclusions. The cumulative incidence of 1-year MACE and its composites did not show statistically significant difference between patients with moderate or severe calcification in the context of CCS or ACS. This suggests that the clinical presentation of patients with calcified coronary arteries does not significantly affect their outcomes.
Introduction
Calcified coronary lesions (CCLs) are considered one of the most challenging atherosclerotic lesions to treat. CCLs are characterized by their low compliance, which can lead to stent underexpansion;1 therefore, adequate plaque modification is crucial before stenting.2,3 The introduction of atherectomy and lithotripsy devices has improved procedural success for patients with CCLs and new generations of drug-eluting stents have reduced the incidence of in-stent restenosis in patients with CCLs.4-6 Nevertheless, calcified lesions still show a higher risk of lesion-related events compared with non-calcified lesions (NCL).7-10 CCLs are commonly observed in patients with chronic coronary syndrome (CCS); however, they can also be present in individuals with acute coronary syndrome (ACS), with patients with CCS having a higher degree of calcification on average.11
Despite the well-established association between coronary artery calcification and poor clinical outcomes, the differences in outcomes between patients with coronary artery calcification presenting with ACS vs CCS have not been well characterized. The goal of this study is to compare outcomes in patients with moderate or severe CCLs presenting with ACS vs CCS.
Methods
Study design and patient selection
A retrospective, single-center cohort study was conducted at the Erasmus Medical Center in Rotterdam, the Netherlands. Patients were eligible for inclusion if they were 18 years or older, underwent percutaneous coronary intervention (PCI) between December 6, 2020, and November 7, 2021, and had angiographically confirmed moderate or severe calcification, scored post-procedure by consensus from 2 interventional cardiologists (AS and WD), in any coronary artery, not necessarily the culprit lesion. Patients were excluded if medical data were unavailable, if the patient underwent a transcatheter aortic valve implantation simultaneously with the index procedure, or if the patient presented to the hospital with out-of-hospital cardiac arrest or shock. The study was approved by the Medical Ethics Committee of the Erasmus University Medical Center.
Baseline and procedural characteristics assessment
Patients were divided into 2 groups: (1) patients with CCS and (2) patients with ACS, including ST-segment elevation myocardial infarction (STEMI), non-ST-segment elevation myocardial infarction (NSTEMI), and unstable angina. Coronary angiography was evaluated by an interventional cardiologist post-procedure to identify and classify all lesions. Angiographically moderate calcification was defined as visible opacifications during the cardiac cycle before contrast injection affecting one side of the artery, while angiographically severe calcification was defined as visible opacification without cardiac motion prior to contrast injection affecting both sides of the artery.
Antiplatelet therapy
All patients in the study received dual antiplatelet therapy (DAPT) consisting of acetylsalicylic acid in combination with one of the following antiplatelet agents: clopidogrel, ticagrelor, or prasugrel. The selection of antiplatelet therapy and the duration of treatment were based on current clinical guidelines, but the final decision was at the discretion of the treating cardiologist. For patients with CCS, clopidogrel was typically prescribed for a duration of 6 months. For patients with NSTEMI or unstable angina, ticagrelor was the preferred choice for a 12-month period. In patients with STEMI, prasugrel was generally administered for 12 months. However, these regimens were flexible and could be adjusted based on individual patient characteristics, comorbidities, or physician preference.
Data collection
Baseline characteristics, procedural information, and follow-up were extracted from electronic health records from the hospital. Data on all-cause mortality were extracted from the municipal civil registry. Data on outcomes were further collected through a written patient questionnaire or a telephone survey.
Study outcomes
The primary endpoint was defined as major adverse cardiac events (MACE), a composite of all-cause mortality, myocardial infarction (MI), and ischemia-driven revascularization at 1 year. Additionally, secondary endpoints were the individual components of MACE, target lesion revascularization (TLR), and target vessel revascularization (TVR).
Statistical analysis
Continuous variables were depicted as mean ± standard deviation or median and 25th to 75th percentile (P25-P75), depending on whether a variable was normally distributed. Normality was determined with the Shapiro-Wilk test. Differences between unpaired continuous variables were tested with the unpaired t-test or the Mann-Whitney U test. Categorical variables were expressed as numbers with percentages. The differences between unpaired categorical variables were tested with the chi-squared test or the Fisher’s exact test, as appropriate, and Fisher-Freeman-Halton exact test for tables larger than 2 x 2.
Event rates were displayed as cumulative incidences, which were calculated with use of Kaplan-Meier survival estimates. Cox proportional hazard models were used to investigate univariable associations of individual predictors, including clinical presentation, with MACE and with secondary endpoints. Subsequently, clinical presentation as well as all variables with a P-value of less than 0.10 in univariable analyses were entered into a multivariable Cox model. To prevent overfitting of the model, no more than 1 variable was entered for every 10 events. The proportional hazards assumption was assessed by log-log plots. Two-sided P-values of less than 0.05 were considered statistically significant. All analyses were conducted using SPSS v28.0.0.0 (IBM).
Results
Baseline characteristics
A total of 724 patients with either ACS (n = 420) or CCS (n = 304) were included in the study. Of the 420 patients with ACS, 123 (29.3%) presented with STEMI, 219 (52.1%) with NSTEMI, and 78 (18.6%) with unstable angina (Table 1). The median age of the patients in the ACS and CCS groups was 72.0 (range, 66.0-78.0 years) and 72.0 (range, 65.0-78.0 years), respectively (P = .67). There was no significant difference in diabetes, previous coronary artery bypass graft (CABG), previous stroke, or chronic kidney disease (CKD) between the 2 groups. Male gender was more prevalent in the CCS group, with 231 of 304 patients (76.0%) compared with 278 of 420 patients (66.2%) in the ACS group (P = .005). Furthermore, the prevalence of hypertension (HTN), hyperlipidemia (HLD), peripheral arterial disease (PAD), heart failure (HF), previous MI, and previous PCI was also higher in the CCS group: HTN: 271 (64.5%) vs 231 (76.0%), P = .001; HLD: 219 (52.1%) vs 182 (59.9%), P = .04; PAD: 66 (15.7%) vs 74 (24.3%), P = .004; HF: 49 (11.7%) vs 64 (21.1%), P = .002; previous MI: 113 (26.9%) vs 115 (37.8%), P = .002; and previous PCI: 134 (31.9%) vs 133 (43.8%), P = .001. The prevalence of smoking was higher in the ACS group: 90 (21.4%) vs 36 (11.8%), P < .001 (Table 1).
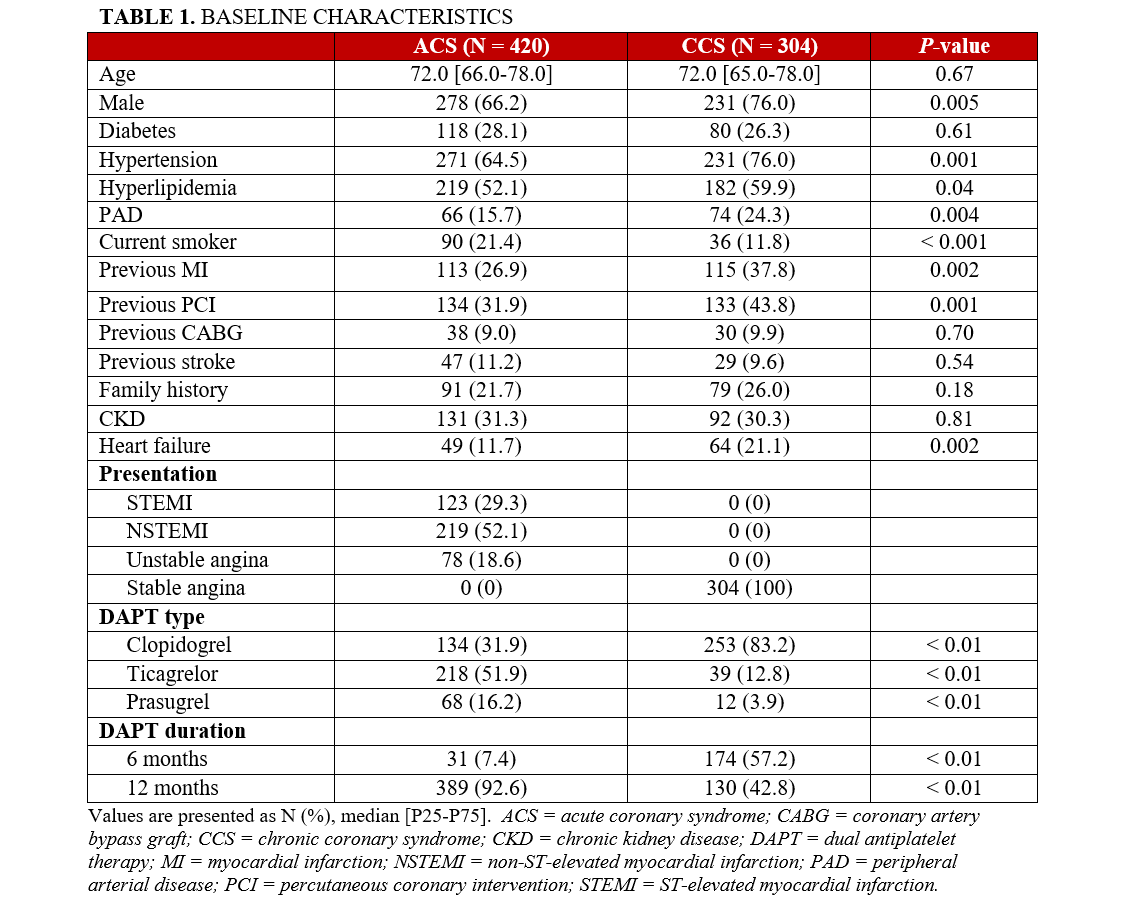
With respect to DAPT, an expected significant difference was found in the types of antiplatelet therapy used between the ACS and CCS groups. For patients with ACS, 134 (31.9%) were treated with clopidogrel, 218 (51.9%) with ticagrelor, and 68 (16.2%) with prasugrel. In contrast, the CCS group had a much higher proportion of patients on clopidogrel and a lower proportion on ticagrelor and prasugrel (253 [83.2%], 39 [12.8%], and 12 [3.9%], respectively) (all P < .01). DAPT duration also differed significantly between the groups. In the ACS group, 389 patients (92.6%) received DAPT for 12 months, while in the CCS group, only 130 patients (42.8%) received DAPT for the same duration. Conversely, a higher proportion of patients with CCS, 174 patients (57.2%), received DAPT for 6 months, compared with just 31 (7.4%) of the patients with ACS (both P < .01) (Table 1).
Procedural characteristics
Radial access was used more often in the ACS group compared with the CCS group (92.6% vs 84.9%, P = .001). A 6F catheter was more common in the ACS group and larger sheaths were more common in the context of CCS (6.0% vs 13.8%, P < .001). The median procedural time in the ACS group was 78 minutes (range, 56-104 minutes) compared with 88 minutes (range, 66-121 minutes) in the CCS group (P < .001). Twenty-seven patients (6.4%) in the ACS group received mechanical hemodynamic support during the PCI compared with 11 patients (3.6%) in the CCS group (P = .13). The contrast use, the number of stents per patient, the total length of stents, and the lesion classification were similar between the 2 groups (Table 2).
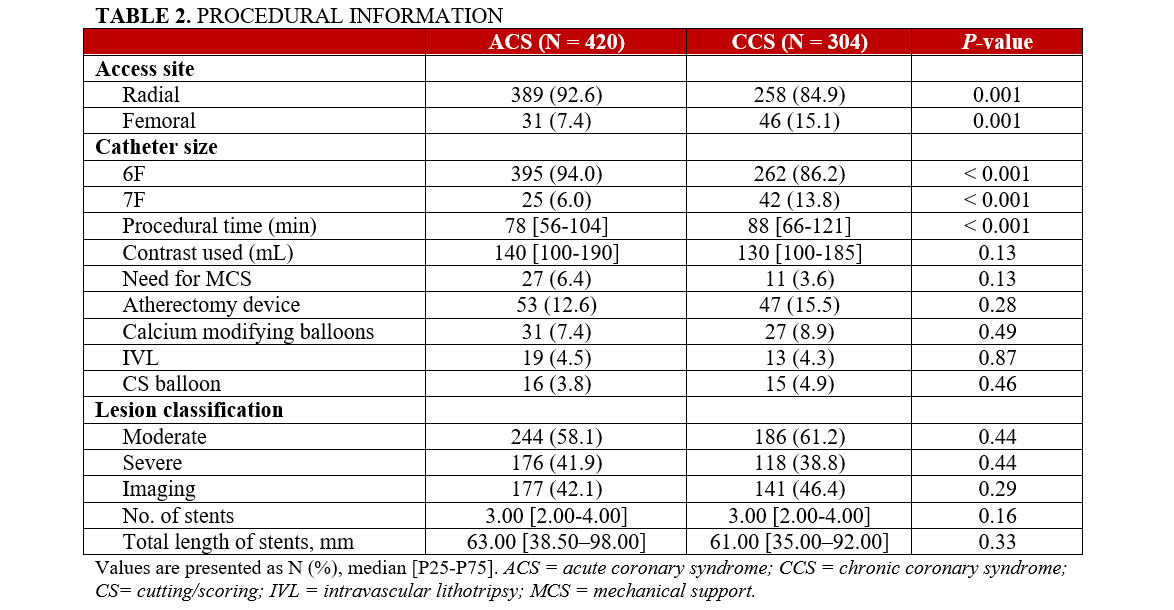
There was no significant difference in the use of dedicated tools to treat CCLs between the 2 groups, with 53 patients (12.6%) in the ACS group receiving treatment with an atherectomy device compared with 47 patients (15.5%) in the CCS group (P = .28), and 31 patients (7.4%) in the ACS group receiving treatment with a modified balloon compared with 27 patients (8.9%) in the CCS group (P = .49) (Table 2).
Outcomes
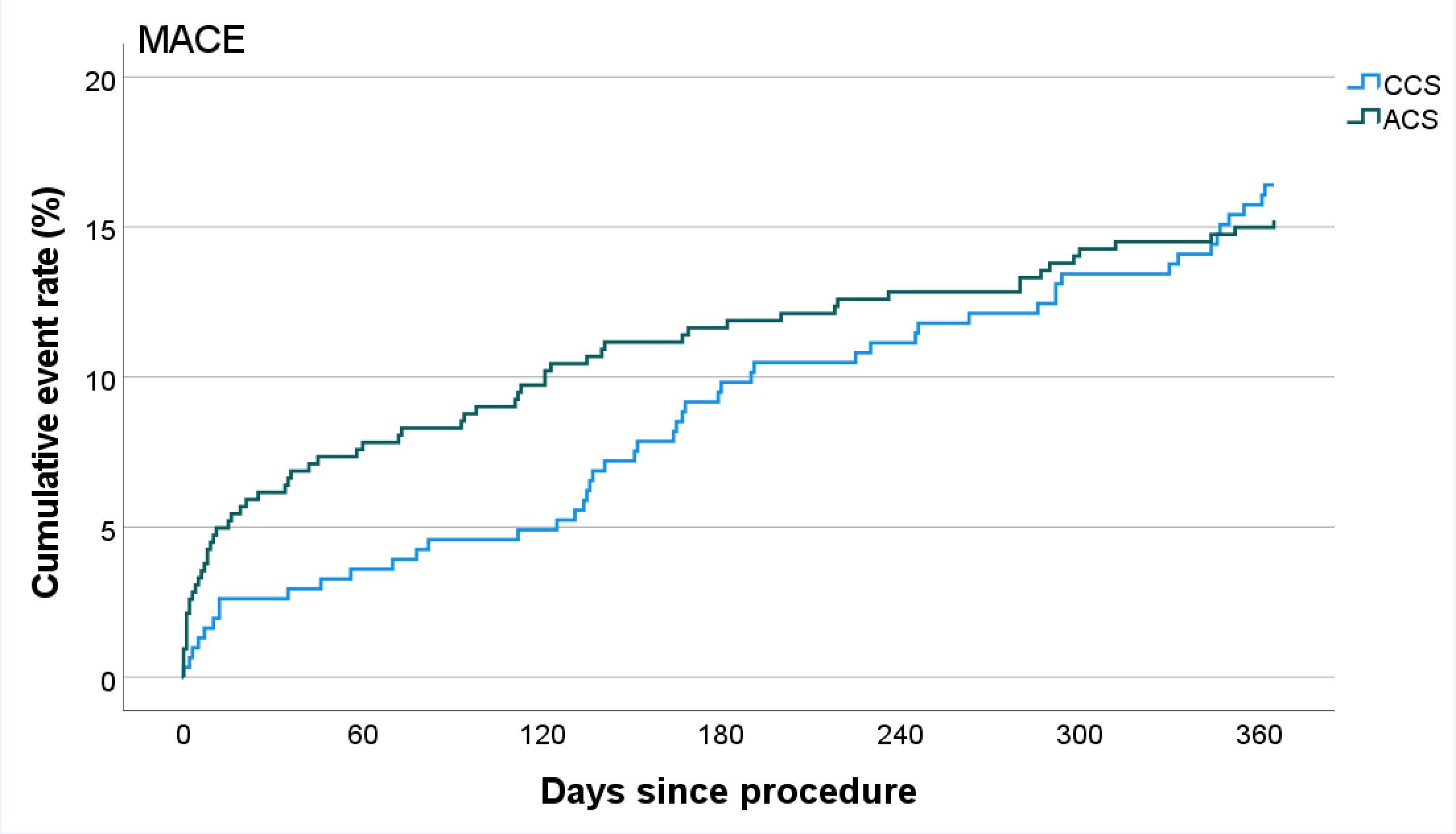
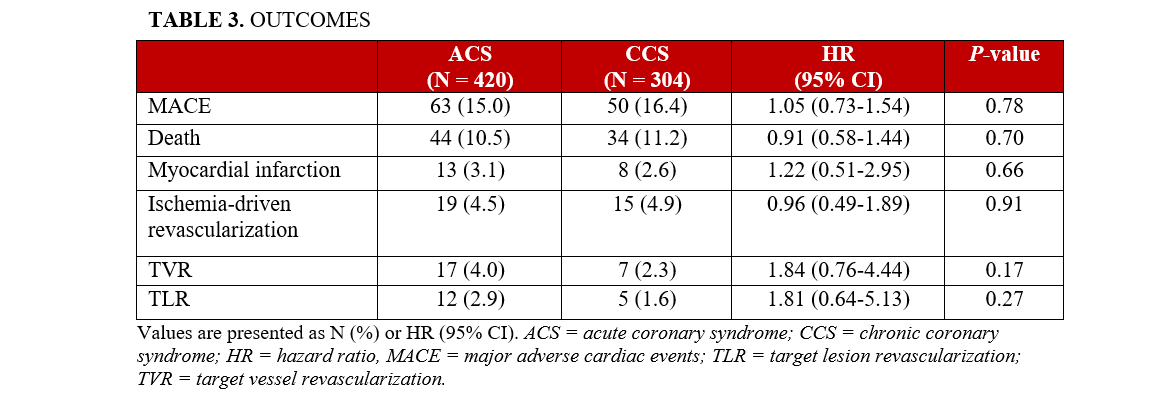
Follow-up was completed in 710 patients (98.1%). No significant difference in the cumulative incidence of MACE between the ACS and CCS groups was observed at 1 year (Figure), with 63 (15.0%) and 50 (16.4%) MACE events at 1 year, respectively (HR, 0.94; 95% CI, 0.65-1.36; P = .74). Additionally, there was no significant difference in the components of MACE or the secondary outcome measures between the groups with regard to the following incidences: death (44 [10.5%] vs 34 [11.2%] [HR, 0.91; 95% CI, 0.58-1.44; P = .70]), MI (13 [3.1%] vs 8 [2.6%] [HR, 1.22; 95% CI, 0.51-2.95; P = .66]), TVR (17 [4.0%] vs 7 [2.3%] [HR, 1.84; 95% CI, 0.76-4.44; P = .17]), ischemia-driven revascularization (19 [4.5%] vs 15 [4.9%] [HR, 0.96; 95% CI, 0.49-1.89; P = .91]), and TLR (12 [2.9%] vs 5 [1.6%] [HR, 1.81; 95% CI, 0.64-5.13; P = .27]) (Table 3).
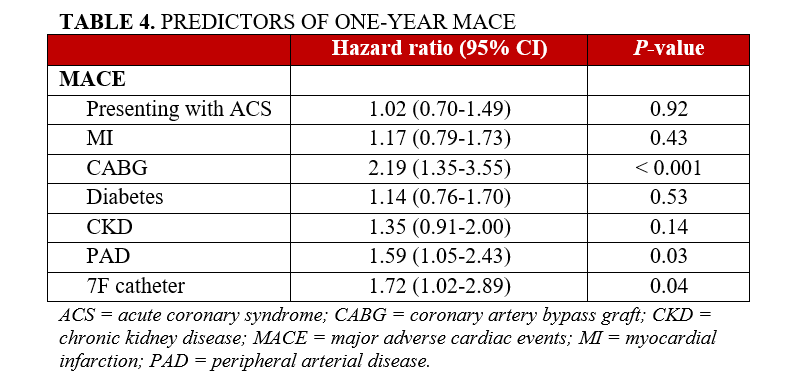
The multivariable analysis identified that clinical presentation was not associated with MACE at 1-year. Furthermore, previous MI, diabetes, and CKD were also not associated with 1-year MACE. In contrast, a history of CABG (HR, 2.19; 95% CI, 1.35-3.55; P < .001), a history of PAD (HR, 1.59; 95% CI, 1.05-2.43; P = .03), and usage of a 7F catheter (HR, 1.72; 95% CI, 1.02-2.89; P = .04) were identified as significant predictors of 1-year MACE (Table 4).
Discussion
The primary finding of this study was that patients with moderately or severely calcified coronary arteries exhibited similar 1-year cumulative incidence of MACE in the context of CCS and ACS. Additionally, we observed no significant differences in the incidence of individual components of 1-year MACE, or ischemia-driven revascularization or TLR, between the 2 groups.
The 1-year MACE rates found in our cohort (15.0%) corroborate with previous studies (16.1%-16.9%) focusing on patients with moderately or severely calcified coronary arteries. However, our study population exhibited a lower rate of MI and ischemia-driven revascularization (3.1% and 4.5% vs 8.0%-8.9% and 7.5%-9.4%, respectively) and higher all-cause mortality rates than those reported in other registries (10.5% vs 4.6%).8,12 This discrepancy may stem from differences in baseline demographics, as our registry generally included an older population with a higher degree of comorbidity, possibly causing more complex procedures. The longer average procedural time (82 vs 46 minutes) also suggests a higher degree of complexity in our procedures compared with those registries.12
Our findings suggest that among patients with significant coronary calcification, clinical presentation may not affect 1-year outcomes. This is contrary to earlier studies showing worse outcomes for ACS compared with CCS in patients with calcified coronary disease.13-15 It appears that the presence of coronary calcification overrules the relative importance of clinical presentation in determining outcomes. CCLs are known to be independent predictors of adverse events, including TLR and all-cause mortality, in patients undergoing PCI.7-10 These lesions may pose considerable procedural challenges, resulting in reduced procedural efficacy, increased complication risks, and compromised long-term prognosis.3
These findings align with those of Genereux et al,16 who investigated 2-year outcomes in patients with calcified lesions undergoing PCI. While the primary aim of their study was to assess the impact of coronary calcification on 2-year outcomes, they also reported data on how clinical presentation influenced outcomes, specifically MACE, mortality, MI, and revascularization. Consistent with our study, their results indicated no significant differences in 2-year outcomes between patients presenting with ACS or stable coronary artery disease in the context of coronary calcification.16
A possible explanation for the similar outcomes observed in ACS and CCS patients with CCLs lies in procedural differences. In our study, femoral access and 7F sheaths were more common in the CCS group. Femoral access, compared with radial access, has been associated with higher rates of MACE including mortality, major bleeding, and vascular complications and similar higher rates of MACE are seen when 7F sheaths are compared with 6F sheaths.17,18 Moreover, the median procedural time was longer in the CCS group, despite a similar number of stents per procedure in both groups, suggesting that lesions in the CCS group were more complex and challenging to treat. These procedural factors could partially account for the comparable outcomes observed between the two groups. Importantly, the use of dedicated atherectomy tools was similar in both groups.
An additional notable distinction between the ACS and CCS groups in our study is that the CCS group exhibited a higher degree of comorbidity. This increased comorbidity may contribute to the similar outcomes observed between the 2 groups, as the greater complexity and frailty associated with the higher degree of comorbidity in CCS patients could partially explain the similar rate of onset of events.12-22
The findings of this study highlight the significant influence of CCLs on 1-year outcomes, underscoring the need for improved risk stratification and management strategies for such lesions. Advancements in PCI, including drug-eluting stents and specialized devices for calcified lesions, have significantly improved outcomes for patients with CCLs.4-6 However, further improvements in treatment approaches remain necessary to optimize care for this high-risk population.
Limitations
The present study exhibits several limitations related to its retrospective design, which inherently restricts the ability to draw definitive conclusions about causality. A notable limitation was the lack of data on post-procedure bleeding rates and periprocedural MI, as cardiac enzyme measurements are not routinely performed in patients with CCS. Furthermore, the use of specific devices for the treatment of calcified lesions in this study introduces the potential for operator and selection bias. Variation in operator expertise and experience with these devices can impact the outcomes observed. Lastly, calcium severity was scored with angiography, which could lead to underscoring of lesions when compared with intracoronary imaging.
Conclusions
This study suggests that patients presenting with ACS and CCLs exhibit similar 1-year clinical outcomes to those with CCS and CCLs. This indicates that moderate to severe coronary calcifications may drive clinical outcomes over and above the clinical presentation. However, given the observational design, further trials are required to validate this finding.
Affiliations and Disclosures
Argisjt Mkrtchjan, BSc; Eleni Ntantou, MD; Alexandros A. Siskos, MD; Jacob J. Elscot, BSc; Jeroen M. Wilschut, MD; Rutger-Jan Nuis, MD, PhD; Roberto Diletti, MD, PhD; Joost Daemen, MD, PhD; Isabella Kardys, MD, PhD; Nicolas M. Van Mieghem, MD, PhD; Wijnand K. den Dekker, MD, PhD
From the Department of Cardiology, Erasmus MC, Cardiovascular Institute, Thorax Center, Rotterdam, the Netherlands.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Wijnand K. den Dekker, MD, PhD, Erasmus Medical Center, Department of Cardiology, Dr. Molewaterplein 40, 3015 GD, Rotterdam, the Netherlands. Email: w.dendekker@erasmusmc.nl
References
1. Kobayashi Y, Okura H, Kume T, et al. Impact of target lesion coronary calcification on stent expansion. Circ J. 2014;78(9):2209-2214. doi:10.1253/circj.cj-14-0108
2. Hill JM, Kereiakes DJ, Shlofmitz RA, et al; Disrupt CAD III Investigators. Intravascular lithotripsy for treatment of severely calcified coronary artery disease. J Am Coll Cardiol. 2020;76(22):2635-2646. doi:10.1016/j.jacc.2020.09.603
3. Angsubhakorn N, Kang N, Fearon C, et al. Contemporary management of severely calcified coronary lesions. J Pers Med. 2022;12(10):1638. doi:10.3390/jpm12101638
4. Abdel-Wahab M, Baev R, Dieker P, et al. Long-term clinical outcome of rotational atherectomy followed by drug-eluting stent implantation in complex calcified coronary lesions. Catheter Cardiovasc Interv. 2013;81(2):285-291. doi:10.1002/ccd.24367
5. Aksoy A, Salazar C, Becher MU, et al. Intravascular lithotripsy in calcified coronary lesions: a prospective, observational, multicenter registry. Circ Cardiovasc Interv. 2019;12(11):e008154. doi:10.1161/CIRCINTERVENTIONS.119.008154
6. Lee MS, Gordin JS, Stone GW, et al. Orbital and rotational atherectomy during percutaneous coronary intervention for coronary artery calcification. Catheter Cardiovasc Interv. 2018;92(1):61-67. doi:10.1002/ccd.27339
7. Bourantas CV, Zhang YJ, Garg S, et al. Prognostic implications of coronary calcification in patients with obstructive coronary artery disease treated by percutaneous coronary intervention: a patient-level pooled analysis of 7 contemporary stent trials. Heart. 2014;100(15):1158-1164. doi:10.1136/heartjnl-2013-305180
8. Généreux P, Madhavan MV, Mintz GS, et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes. Pooled analysis from the HORIZONS-AMI (Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction) and ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) TRIALS. J Am Coll Cardiol. 2014;63(18):1845-1854. doi:10.1016/j.jacc.2014.01.034
9. Budoff MJ, Shaw LJ, Liu ST, et al. Long-term prognosis associated with coronary calcification: observations from a registry of 25,253 patients. J Am Coll Cardiol. 2007;49(18):1860-1870. doi:10.1016/j.jacc.2006.10.079
10. Copeland-Halperin RS, Baber U, Aquino M, et al. Prevalence, correlates, and impact of coronary calcification on adverse events following PCI with newer-generation DES: findings from a large multiethnic registry. Catheter Cardiovasc Interv. 2018;91(5):859-866. doi:10.1002/ccd.27204
11. Beckman JA, Ganz J, Creager MA, Ganz P, Kinlay S. Relationship of clinical presentation and calcification of culprit coronary artery stenoses. Arterioscler Thromb Vasc Biol. 2001;21(10):1618-1622. doi:10.1161/hq0901.095554
12. Sharma SK, Bolduan RW, Patel MR, et al. Impact of calcification on percutaneous coronary intervention: MACE-trial 1-year results. Catheter Cardiovasc Interv. 2019;94(2):187-194. doi:10.1002/ccd.28099
13. Alcock RF, Yong AS, Ng AC, et al. Acute coronary syndrome and stable coronary artery disease: are they so different? Long-term outcomes in a contemporary PCI cohort. Int J Cardiol. 2013;167(4):1343-1346. doi:10.1016/j.ijcard.2012.04.011
14. Montalescot G, Ongen Z, Guindy R, et al; RIVIERA Investigators. Predictors of outcome in patients undergoing PCI. Results of the RIVIERA study. Int J Cardiol. 2008;129(3):379-387. doi:10.1016/j.ijcard.2007.07.127
15. Landes U, Kornowski R, Assali A, et al. Predictors of long term outcomes in 11,441 consecutive patients following percutaneous coronary interventions. Am J Cardiol. 2015;115(7):855-859. doi:10.1016/j.amjcard.2015.01.009
16. Généreux P, Redfors B, Witzenbichler B, et al. Two-year outcomes after percutaneous coronary intervention of calcified lesions with drug-eluting stents. Int J Cardiol. 2017;231:61-67. doi:10.1016/j.ijcard.2016.12.150
17. Mamas MA, Ratib K, Routledge H, et al; British Cardiovascular Intervention Society and the National Institute for Cardiovascular Outcomes Research. Influence of arterial access site selection on outcomes in primary percutaneous coronary intervention: are the results of randomized trials achievable in clinical practice? JACC Cardiovasc Interv. 2013;6(7):698-706. doi:10.1016/j.jcin.2013.03.011
18. Grossman PM, Gurm HS, McNamara R, et al; Blue Cross Blue Shield of Michigan Cardiovascular Consortium (BMC2). Percutaneous coronary intervention complications and guide catheter size: bigger is not better. JACC Cardiovasc Interv. 2009;2(7):636-644. doi:10.1016/j.jcin.2009.05.012
19. Zhang F, Wong C, Chiu Y, et al. Prognostic impact of comorbidity measures on outcomes following acute coronary syndrome: a systematic review. Int J Clin Pract. 2021;75(10):e14345. doi:10.1111/ijcp.14345
20. Sud M, Han L, Koh M, et al. Low-density lipoprotein cholesterol and adverse cardiovascular events after percutaneous coronary intervention. J Am Coll Cardiol. 2020;76(12):1440-1450. doi:10.1016/j.jacc.2020.07.033
21. Mamas MA, Fath-Ordoubadi F, Danzi GB, et al. Prevalence and impact of co-morbidity burden as defined by the Charlson co-morbidity index on 30-day and 1- and 5-year outcomes after coronary stent implantation (from the Nobori-2 study). Am J Cardiol. 2015;116(3):364-371. doi:10.1016/j.amjcard.2015.04.047
22. Jalali A, Hassanzadeh A, Najafi MS, et al. Predictors of major adverse cardiac and cerebrovascular events after percutaneous coronary intervention in older adults: a systematic review and meta-analysis. BMC Geriatr. 2024;24(1):337. doi:10.1186/s12877-024-04896-4