A Rare Case of Campylobacter-Associated Hallux Osteomyelitis Treated with Novel Biologic Tendon Interposition
Campylobacter species are common causes of gastroenteritis but are rarely implicated in osteomyelitis, particularly in the peripheral skeleton. This case report describes a presumed hematogenous Campylobacter osteomyelitis of the hallux in an immunocompetent adult, complicated by coexisting crystal arthropathy and culture-negative findings. A novel biologic tendon interposition and functional proprioceptive reconstruction technique was used following joint excision, resulting in durable healing and preserved hallux function.
Key Takeaways
- Campylobacter osteomyelitis is rare and diagnostically challenging: Extraintestinal Campylobacter infections may present as culture-negative osteomyelitis, requiring clinicians to rely on temporal associations, imaging findings, and clinical judgement—especially in patients with recent enteritis.
- Crystal arthropathy does not exclude infection: Gout-like findings can coexist with infectious or post-infectious osteomyelitis, potentially delaying diagnosis and emphasizing the importance of maintaining a broad differential in atypical monoarthritis.
- Biologic tendon interposition can preserve function after aggressive debridement: Reconstructive techniques that restore balanced flexor–extensor tension and proprioceptive feedback may offer durable functional outcomes following joint excision for complex infectious and inflammatory forefoot pathology.
Campylobacter species are among the most frequent bacterial causes of gastroenteritis worldwide, typically transmitted through contaminated food or water. Although the gastrointestinal illness is usually self-limited, extraintestinal infection is uncommon and generally occurs in individuals with underlying medical conditions or transient bacteremia following enteric infection.1,2 Osteomyelitis due to Campylobacter is especially rare, with only a small number of cases reported in the literature, most involving the spine or large joints.3-6 Peripheral skeletal involvement is exceedingly unusual.
Published cases of Campylobacter-associated osteomyelitis are scarce and usually involve either vertebral osteomyelitis due to Campylobacter jejuni in immunocompetent adults, long-bone osteomyelitis in the immunocompromised, or prosthesis-associated infection. A 2023 case describes an immunocompetent adult with epidural abscess.3 In 2024, a literature review summarized 6 Campylobacter jejuni vertebral cases.4 Cervical vertebral osteomyelitis was also described in 2021, which parallels our case, as both had negative bone and blood cultures but positive stool polymerase chain reaction (PCR).5 Beyond C. jejuni, Campylobacter fetus has repeatedly caused vertebral osteomyelitis or spondylodiscitis.6,7 A case report describes long-bone disease in 2-sided femoral osteomyelitis in the setting of acquired hypogammaglobulinemia.8 Prosthesis-associated osteomyelitis has been reported in a hip arthroplasty in a patient with acquired immunodeficiency syndrome (AIDS).9 Pediatric involvement is rare but includes a probable C. jejuni osteomyelitis in a 14-month-old child.10 Collectively, the literature suggests vertebral predilection, hematogenous seeding after enteritis, frequent culture negativity, and host factors such as immunosuppression or prosthesis as recurrent themes.
Accurate microbiologic identification of Campylobacter remains challenging because of its fastidious growth requirements. The organism thrives only under microaerophilic conditions and typically requires selective enrichment media and a narrow range of temperature and oxygen concentration for successful isolation. Even under optimal circumstances, isolation from nonenteric specimens is difficult, and culture sensitivity decreases further following prior antibiotic exposure or in mixed-flora samples.11-13 As a result, false-negative cultures are not uncommon, and the true frequency of invasive Campylobacter infections may be underestimated. Molecular techniques such as PCR and 16S ribosomal RNA sequencing have improved diagnostic yield but are not universally available in clinical practice.14
Recent advances in limb reconstruction and amputation science have demonstrated that re-establishing physiologic tension between agonist and antagonist muscle groups can restore more natural proprioceptive feedback and improve functional motor control. The agonist–antagonist myoneural interface (AMI) model, pioneered in amputee studies, shows that surgically coupling opposing muscle groups allows reciprocal lengthening and contraction patterns that reactivate afferent signaling from muscle spindles and Golgi tendon organs, improving limb position sense and reducing reliance on visual cues for movement.15-19 Translating this principle to joint-preserving surgery, biologic tendon interposition constructs can similarly maintain dynamic tension across a joint, acting not only as a soft-tissue spacer but also as a potential proprioceptive bridge that preserves the physiologic communication between flexor and extensor mechanisms. This concept informed the reconstructive approach described in the present case, in which interposed tendon tissue was used to create a biologic spacer and functional coupling after debridement of infected and degenerated articular structures.
Key Details of the Case Presentation
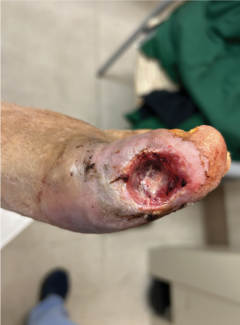
We present the case of a 55-year-old male patient with past medical history of tobacco use, liver steatosis, chronic obstructive pulmonary disease, nephrolithiasis, Lyme disease, and in 2008 had ehrlichiosis with Rocky Mountain spotted fever (RMSF) with spotted fever rickettsioses (SFR). The patient presented to the clinic in August 2025 with his right great toe edematous, erythematous, and exquisitely tender, especially the hallux interphalangeal joint. The patient had been referred to us and to a local rheumatologist for evaluation. The patient reported that beginning about 3 months prior, he first noticed the redness, swelling, and pain which had waxed and waned ever since and was presently in a flare. The patient explained that his right foot symptoms began around the time that he was hospitalized for Campylobacter infection in April 2025 which he contracted after eating an undercooked hamburger patty at a local fast-food restaurant. Campylobacter infection was initially detected from a stool sample with enzyme immunoassay (EIA) and confirmed with a culture.
Examination of the patient’s right foot revealed palpable pulses, intact gross and protective sensation, elevated temperature of the skin on the dorsum of the foot and hallux, with edema, erythema, induration of the right hallux extending from the interphalangeal joint into the dorsal medial forefoot.
We ordered lab work including complete blood count (CBC), basic metabolic panel (BMP), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and uric acid, all of which were unremarkable. Radiographs of the right foot demonstrated nonspecific subcutaneous edema, degenerative joint disease, and diffuse osteopenia, but were otherwise unremarkable. After a lengthy discussion of the risks and benefits, we obtained consent for joint aspiration. Using aseptic technique, an 18-gauge needle on a 5cc syringe aspirated the right hallux interphalangeal joint, which revealed chalky-white, gritty, crystals and blood-tinged synovial fluid. We replaced the syringe with the needle still in place intraarticularly and injected 0.5cc of dexamethasone sodium phosphate (10mg/mL). The patient tolerated the procedure well with no complications. The joint fluid aspirated was sent for culture and sensitivity testing as well as crystal analysis.
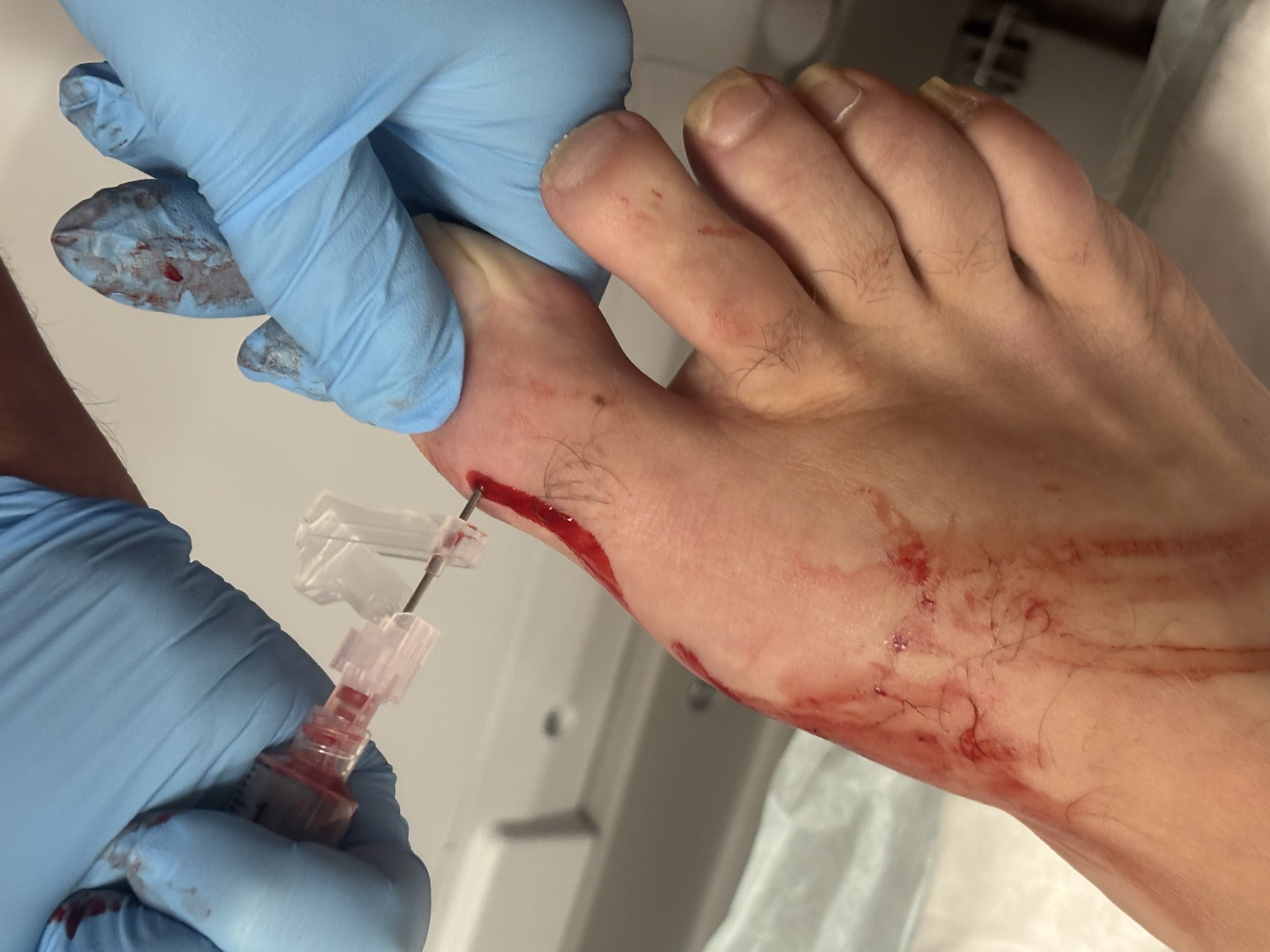
We issued the patient a prescription for ten days of doxycycline 100mg twice daily because the presentation of his symptoms was atypical and suggested possible infection. We advised the patient to follow up with his primary care provider for long-term management of gout and prescribed colchicine as the patient’s uric acid was 6.2 mg/dL and joint fluid aspirate was positive for monosodium urates. We recommended that he keep the previously arranged appointment with rheumatology to seek their additional expertise. The rheumatologist performed an extensive laboratory evaluation including ceruloplasmin, alpha-1 antitrypsin, hepatitis B surface antibody (HBsAb), hepatitis B surface antigen (HBsAg), hepatitis A total antibody and IgM, hepatitis C virus antibody (anti-HCV), anti–smooth muscle (actin) antibody, anti–mitochondrial M2 antibody, anti–liver kidney microsomal (anti-LKM) antibody, antinuclear antibody (ANA) by indirect immunofluorescence assay (IFA) with reflex testing, creatine kinase (CK), and a comprehensive autoimmune and rheumatologic biomarker panel including lupus, rheumatoid arthritis, Sjögren’s syndrome, mixed connective tissue disease, antiphospholipid antibody syndrome, systemic sclerosis, myositis, and thyroid autoantibodies, as well as antineutrophil cytoplasmic antibodies (ANCA) by IFA and HLA-B27 typing. All results were within normal limits or negative and no new treatments were initiated by the rheumatologist.
A month after our initial encounter with the patient, their rheumatologist reached out and informed us that the patient had initially improved for 3 weeks, but over the past week had developed recurrence of the edema and pain in his right great toe with new onset body aches, malaise, fatigue, chills, night sweats, right ankle and knee pain, and ascending erythematous streaking extending from the hallux interphalangeal joint up the lower leg. We then contacted the patient and advised him to come to the emergency department for evaluation.
Hospital Admission
The patient was seen in the Emergency Department and presented with a heart rate of 101 beats per minute, blood pressure of 152/92 mmHg, and afebrile. Labs obtained demonstrated leukocytosis and neutrophilia. Radiographs obtained of the right foot were again unremarkable. The patient was admitted for sepsis and septic arthritis and started on intravenous vancomycin and cefepime. Blood cultures were obtained at time of admission.
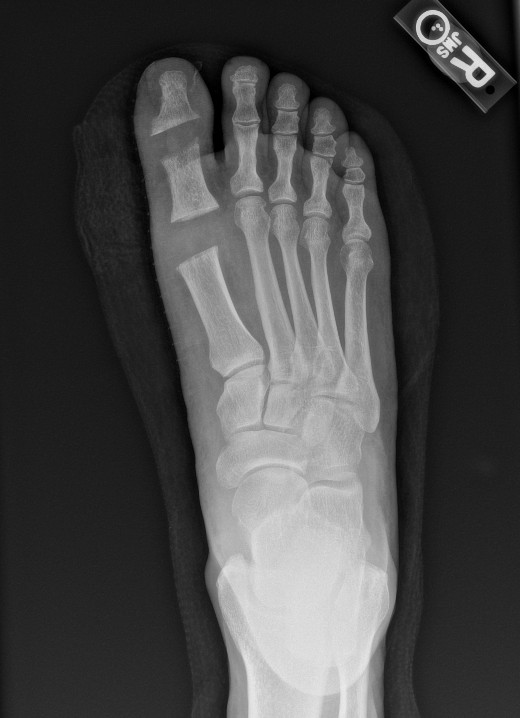
Subsequent magnetic resonance imaging (MRI) of the right foot demonstrated small effusions of the first metatarsal head with underlying bone marrow edema and enhancement. There was also low T1 signal along the medial cortex and subcortical marrow. Additionally, the MRI showed erosions on the plantar aspect of the head of the hallux proximal phalanx with bone marrow edema and enhancement. These MRI findings were suggestive of osteomyelitis.
Although the MRI findings were concerning for osteomyelitis, the diagnostic picture remained complex. Infection had been suspected early in the clinical course; however, repeated blood and joint cultures obtained up to that point had remained negative. Additionally, the patient’s known history of gout raised the possibility that the cortical erosions and marrow changes seen on MRI could also represent inflammatory osseous damage from crystal arthropathy rather than true infection. Given this overlap and the absence of microbiologic confirmation, we elected to pursue repeat aspiration and culture prior to proceeding with operative debridement or bone biopsy. This approach allowed an additional opportunity to identify an organism, strengthen diagnostic certainty, and avoid subjecting the patient to surgical excision without clearer evidence of infection. When aspiration again failed to yield definitive culture results, surgical intervention was ultimately pursued for source control and diagnostic confirmation.
After a lengthy discussion of the risks and benefits, we obtained consent for right hallux interphalangeal joint and metatarsophalangeal joint aspiration. Using aseptic technique, we used an 18-gauge needle on a 20cc syringe to perform the aspirations, which demonstrated blood-tinged serous fluid negative for purulence and crystals. The patient tolerated the procedure well with no complications. The fluid was sent for pathology and culture testing.
Preliminary results from the blood cultures obtained at admission and on the aspirated joint fluid were negative however and it was decided to take the patient to the operative room and surgically excise the diseased bone identified on MRI based on high clinical suspicion for infection for source control, pathology, and culture.
Surgical Intervention
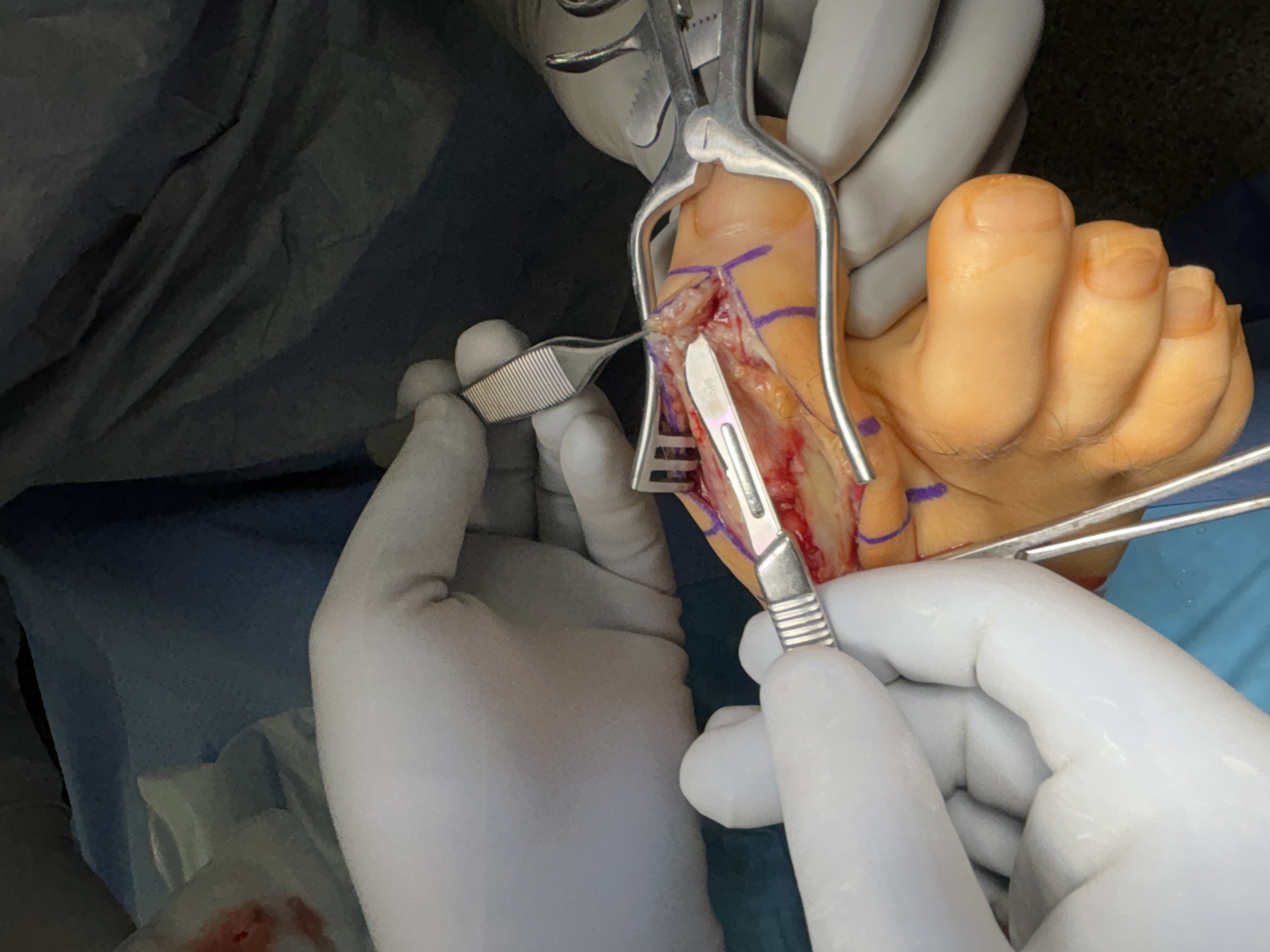
Our linear incision began dorsal and medial to the extensor tendon, one centimeter distal to the hallux interphalangeal joint and extending a centimeter proximal to the head of the first metatarsal. Using blunt and sharp dissection, we carried the incision down deep to bone. Using sharp dissection, we then reflected the joint capsule and soft tissues away from the hallux interphalangeal joint and the first metatarsal phalangeal joint. Using a sagittal saw, we resected the head of the first metatarsal at the neck, and passed it from the surgical field. We then used a sagittal saw to resect the base of the hallux proximal phalanx, and also passed it from the surgical field. Both sections of resected bone were sent for gross pathology, culture and sensitivity testing. Using sharp dissection, we excised the sesamoid complex in its entirety. Pulse lavage thoroughly irrigated the area.
Attention was then directed to the hallux interphalangeal joint where we noted chalky white-yellow deposits, a sample of which we sent for crystal analysis. Using a sagittal saw, we resected the head of the proximal phalanx was resected at the neck and the base of the distal phalanx, which we passed from the surgical field. Both of these additional sections of resected bone were sent for gross pathology, culture and sensitivity testing. Pulse lavage again thoroughly irrigated the site.
Following complete surgical debridement of infected and gout-involved bone and soft tissue, we directed our attention to reconstructing both of the joint spaces and restoring balanced tendon mechanics. To achieve this, we fashioned a biologic spacer by interposing slips of the patient’s native flexor and extensor tendons into the prepared joint space. We rolled the tendon tissue secured it centrally within the defect, providing a soft-tissue buffer that preserved anatomic spacing while mitigating direct osseous contact. The residual ends of the flexor and extensor tendons were then tensioned and anchored to opposing sides of the joint to recreate a physiologic agonist–antagonist relationship. This construct was designed to maintain reciprocal length–tension dynamics during motion, potentially enhancing proprioceptive signaling and soft-tissue balance across the reconstructed site. Resorbable antibiotic loaded cement beads (Stimulan Rapid Cure) were then prepared according to the manufacture’s recommendations using vancomycin and gentamicin and implanted into the space. The joint was ranged intraoperatively to confirm smooth excursion and absence of impingement before layered closure. The incision was then closed with layered sutures and the right hallux was splinted in a rectus slightly dorsiflexed position with sterile dressings. The patient was offloaded in a cast boot.
Surgical Aftercare and Specimen Results
Following surgery, the patient remained hospitalized for 3 more days receiving continuous IV antibiotic therapy, infectious disease consultation, and rehabilitation. Patient’s condition dramatically improved following the surgery with the pain, erythema, and swelling fully resolving. Following discharge from the hospital, the patient was seen on a regular basis outpatient for follow-up care until completely healed. The patient was non-weight bearing for 4 weeks then transitioned to weight-bearing in a CAM boot for three weeks, then transitioned to full unrestricted weightbearing.
Pathology from the excised bone of the first metatarsophalangeal joint revealed acellular amorphous material suggestive of gout that was nonpolarizable. Analysis of the interphalangeal joint excised bone and metatarsophalangeal joint excised bone showed extensive bone marrow fibrosis with neutrophil infiltration, confirming osteomyelitis. Bone cultures yielded no definitive findings.
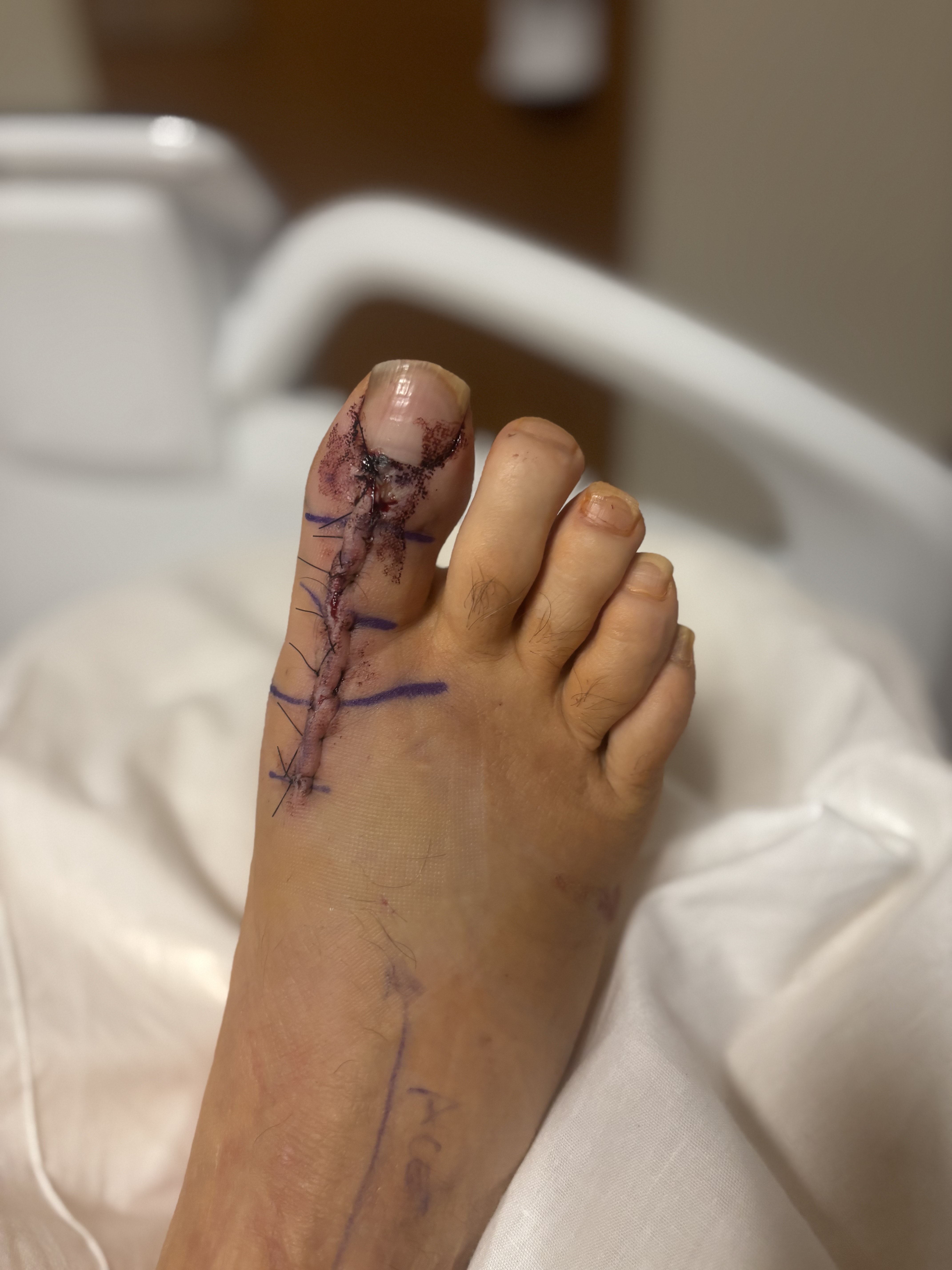
Observations and Lessons Learned
The unique pathophysiologic context of the timing of the infection, symptomatology, and systemic course lends itself to a presumed diagnosis of hematogenous Campylobacter osteomyelitis in the right foot.
Hematogenous osteomyelitis of the foot is uncommon in adults and usually arises secondary to bacteremia in the setting of predisposing systemic conditions like immunosuppression, diabetes, and peripheral vascular disease. Campylobacter infection is rare, and most reported infections occur in the gastrointestinal tract.1,2 Very few cases have been described of extraintestinal dissemination, and among those, osteoarticular involvement is typically in the spine or large joints.1-10 To our knowledge, presumed hematogenous Campylobacter osteomyelitis of the hallux has not been previously reported in association with gout.
The chronologic association between the patient’s acute Campylobacter enteritis and subsequent onset of monoarticular inflammation raises strong suspicion for a postinfectious of reactive process that evolved into osteomyelitis.
Campylobacter is well documented to trigger reactive arthritis, particularly in genetically predisposed individuals (notably those positive for HLA-B27). The organism’s lipooligosaccharides share molecular mimicry with human gangliosides, contributing to immune-mediated inflammation that may persist long after clearance of the intestinal infection.20-22 In this case, the chronicity and localization of pain, together with radiographic and MRI findings of cortical erosion and marrow enhancement, support hematogenous seeding or immune-mediated destruction of bone.
An additional diagnostic challenge arose from the coexistence of crystal arthropathy. The aspiration of chalky, white crystals favored a diagnosis of gout; however, it didn’t exclude concomitant infection. Both conditions may present with erythema, swelling, and elevated inflammatory markers, complicating early differentiation. Although multiple cultures from blood, joint aspirate, and bone were negative, the absence of microbial growth does not preclude infection, particularly following antibiotic exposure. The patient’s rapid clinical improvement following debridement and broad-spectrum antibiotic therapy further supports an infectious or postinfectious component.
Surgical management was indicated for source control and diagnostic clarification. By utilizing a biologic tendon interposition technique designed to preserve balanced flexor-extensor tension and proprioceptive feedback, the reconstructed hallux maintained functional range of motion and stability. This approach supported durable wound closure and restoration of pain-free, functional use of the hallux, demonstrating the potential benefit of biologically coupled tendon reconstruction in complex infectious and inflammatory forefoot pathology. The patient progressed to full healing without postoperative complication or recurrence of infection. The use of bioabsorbable calcium sulfate antibiotic beads provided high local antibiotic concentrations to the surgical site, mitigating the risk of recurrence. The patient’s subsequent complete resolution of symptoms and radiographic stability on follow-up lend further weight to the presumption of an infectious etiology.
Limitations
Despite the temporal and clinical association between the patient’s documented Campylobacter enteritis and subsequent osteomyelitis, the lack of organism recovery from bone or blood cultures precludes absolute causation. Culture-negative osteomyelitis remains a diagnostic challenge, particularly for fastidious organisms such as Campylobacter, whose isolation requires specialized media, microaerophilic conditions, and prompt handling which are factors that may not have been optimized om this case. Even if cultures were successful, advanced molecular assays such as PCR or 16s RNA sequencing were not available in the rural health setting where this case took place.
The patient’s HLA-B27 negativity further complicates interpretation, as reactive arthritis secondary to Campylobacter jejuni typically occurs in genetically predisposed individuals. This finding suggests that the pathogenesis in this case may have involved direct hematogenous seeding or a non–HLA-B27-mediated inflammatory cascade. Diagnostic certainty was further limited by the coexistence of crystal arthropathy, which can clinically and radiographically mimic infection and may have delayed early surgical sampling.
Despite these constraints, the consistent chronology, imaging findings, and full resolution following surgical and antibiotic management strongly support presumed Campylobacter-associated osteomyelitis in a non–HLA-B27 host.
Conclusion
This case highlights the diagnostic complexity of overlapping crystal, infectious, and reactive arthropathy. It underscores the need for clinicians to maintain a broad differential diagnosis in atypical presentations of monoarthritis, especially in patients with recent gastrointestinal infections. While culture-negative osteomyelitis may pose diagnostic uncertainty, clinical judgment grounded in temporal association, imaging findings, and therapeutic response remains essential.
Arthur Evensen, DPM, CWSP is a podiatric surgeon and wound care specialist at Golden Valley Memorial Healthcare in Clinton, Missouri.
Jaime Moore, AGNP is a nurse practitioner at Golden Valley Memorial Healthcare in Clinton, Missouri.
Disclosures
The authors of this article declare no conflict of interest. The companies involved had no role in the design of the study; in the collection, analyses, or interpretation of date; in the writing of the manuscript, or in the decision to publish the results.
Disclosure of Use of Artificial Intelligence (AI)
The authors disclose that artificial intelligence (AI) based tools were used in the preparation of this manuscript solely to aid in literature organization, language refinement, and reference verification. All conceptual design, clinical content, interpretation, and final synthesis were conducted and verified by the authors. Human oversight was maintained throughout, and the authors accept full responsibility for the accuracy, integrity, and originality of the work. The manuscript reflects independent clinical judgment and human intellectual contribution at every stage.
References
1. Kirk KF, Boel J, Nielsen HL. Vertebral osteomyelitis caused by Campylobacter jejuni in an immunocompetent patient. Gut Pathog. 2023;15(1):61. doi:10.1186/s13099-023-00589-2
2. Greminger S, Strahm C, Notter J, Martens B, Helfenstein SF, Den Hollander J, Frischknecht M. Vertebral osteomyelitis with Campylobacter jejuni – a case report and review of the literature of a very rare disease. J Bone Joint Infect. 2024;9:59-65. doi:10.5194/jbji-9-59-2024
3. Awada B, Hindy JR, Chalfoun M, Kanj SS. Cervical osteomyelitis potentially caused by Campylobacter fetus. J Infect Public Health. 2021;14(11):1233-1236. doi:10.1016/j.jiph.2021.08.017
4. Yamashita K, Aoki Y, Hiroshima K. Pyogenic vertebral osteomyelitis caused by Campylobacter fetus subsp. fetus. Spine (Phila Pa 1976). 1999;24(3):582-584.
5. Olaiya D, Cheng PM, Khan M, et al. Campylobacter fetus spondylodiscitis: a case report and literature review. IDCases. 2018;14:e00465.
6. Hartman J, Westerman M, Wagenaar JFP. Two-sided femoral Campylobacter jejuni osteomyelitis in acquired hypogammaglobulinemia. BMC Infect Dis. 2020;20:199.
7. Peterson MC, Kaye D, Hsu JJ, et al. Campylobacter jejuni osteomyelitis in a prosthetic hip with bacteremia. Clin Infect Dis. 1993;16(3):439-440.
8. Vandenberg O, Van Driessche E, Dupont J, et al. Possible Campylobacter jejuni osteomyelitis in a 14-month-old child. Int J Infect Dis. 2003;7(2):164-165.
9. Linton D, Lawson AJ, Owen RJ, Stanley J. Campylobacter jejuni and Campylobacter coli: factors affecting their detection and isolation in the laboratory. Clin Microbiol Rev. 2015;28(3):687-720.
10. Fitzgerald C, Nachamkin I, Moran AP, et al. Challenges in Campylobacter diagnostics. Eur J Clin Microbiol Infect Dis. 2019;38(11):2033-2042.
11. Bullman S, Corcoran D, O’Leary M, et al. Campylobacter bacteremia and the difficulties of laboratory diagnosis. Diagn Microbiol Infect Dis. 2017;88(3):229-234.
12. Mandrell RE, Le Clerc JE, Vizzini AM, et al. Application of PCR and 16S rRNA sequencing to improve Campylobacter detection in clinical specimens. J Clin Microbiol. 2019;57(12):e01145-19.
13. Hameed F, Malik M, Khan SA, et al. Molecular identification of Campylobacter species in culture-negative infections: diagnostic challenges and emerging solutions. Front Cell Infect Microbiol. 2020;10:243.
14. Parisi A, Tiengo C, Diletta V, et al. Advances in molecular diagnostics for Campylobacter species: from PCR to next-generation sequencing. Pathogens. 2022;11(7):803.
15. Srinivasan SS, Carty MJ, Calvaresi AE, et al. Agonist–antagonist myoneural interface: regenerating physiological muscle dynamics after amputation. Sci Transl Med. 2017;9(386):eaam7817.
16. Herr HM, Carty MJ. Restoration of sensory feedback in human amputees through a regenerating agonist–antagonist myoneural interface. Sci Transl Med. 2019;11(512):eaaw0935.
17. Clites TR, Carty MJ, Srinivasan SS, et al. Proprioception from a neurally controlled lower-extremity prosthesis. Sci Transl Med. 2018;10(443):eaap8373.
18. Clites TR, Herr HM, Srinivasan SS, et al. The Ewing amputation: the first human implementation of the agonist–antagonist myoneural interface. Plast Reconstr Surg. 2018;141(2):453-462. doi:10.1097/PRS.0000000000004065
19. Hoh DJ, Carty MJ, Srinivasan SS, et al. Advanced surgical concepts in limb amputation: the emerging role of the agonist–antagonist myoneural interface. J Plast Reconstr Aesthet Surg. 2021;74(2):272-280. doi:10.1016/j.bjps.2020.09.063
20. Hannu T. Reactive arthritis. Best Pract Res Clin Rheumatol. 2011;25(3):347-357.
21. Yuki N. Molecular mimicry between gangliosides and lipooligosaccharides of Campylobacter jejuni isolated from patients with Guillain–Barré syndrome. Ann Neurol. 1997;42(5):791-799.
22. Kaakoush NO, Castaño-Rodríguez N, Mitchell HM, Man SM. Global epidemiology of Campylobacter infection. Clin Microbiol Rev. 2015;28(3):687-720.
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Podiatry Today or HMP Global, their employees, and affiliates.