Transcatheter Treatment of a Large Saphenous Vein Graft Aneurysm
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
J INVASIVE CARDIOL 2026. doi:10.25270/jic/26.00075. Epub April 13, 2026.
A 70-year-old man presented with persistent exertional angina. He had undergone coronary artery bypass grafting (CABG) 14 years ago, with a left internal mammary artery graft (LIMA) to the left anterior descending artery (LAD) and a saphenous vein Y-graft (SVG) to the first obtuse marginal (OM1) and second diagonal (D2) branches.
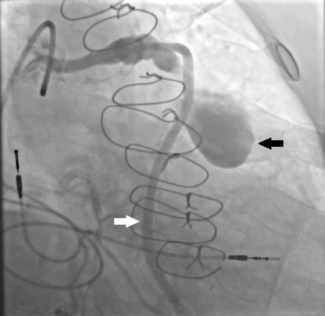
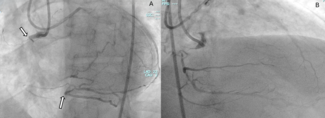
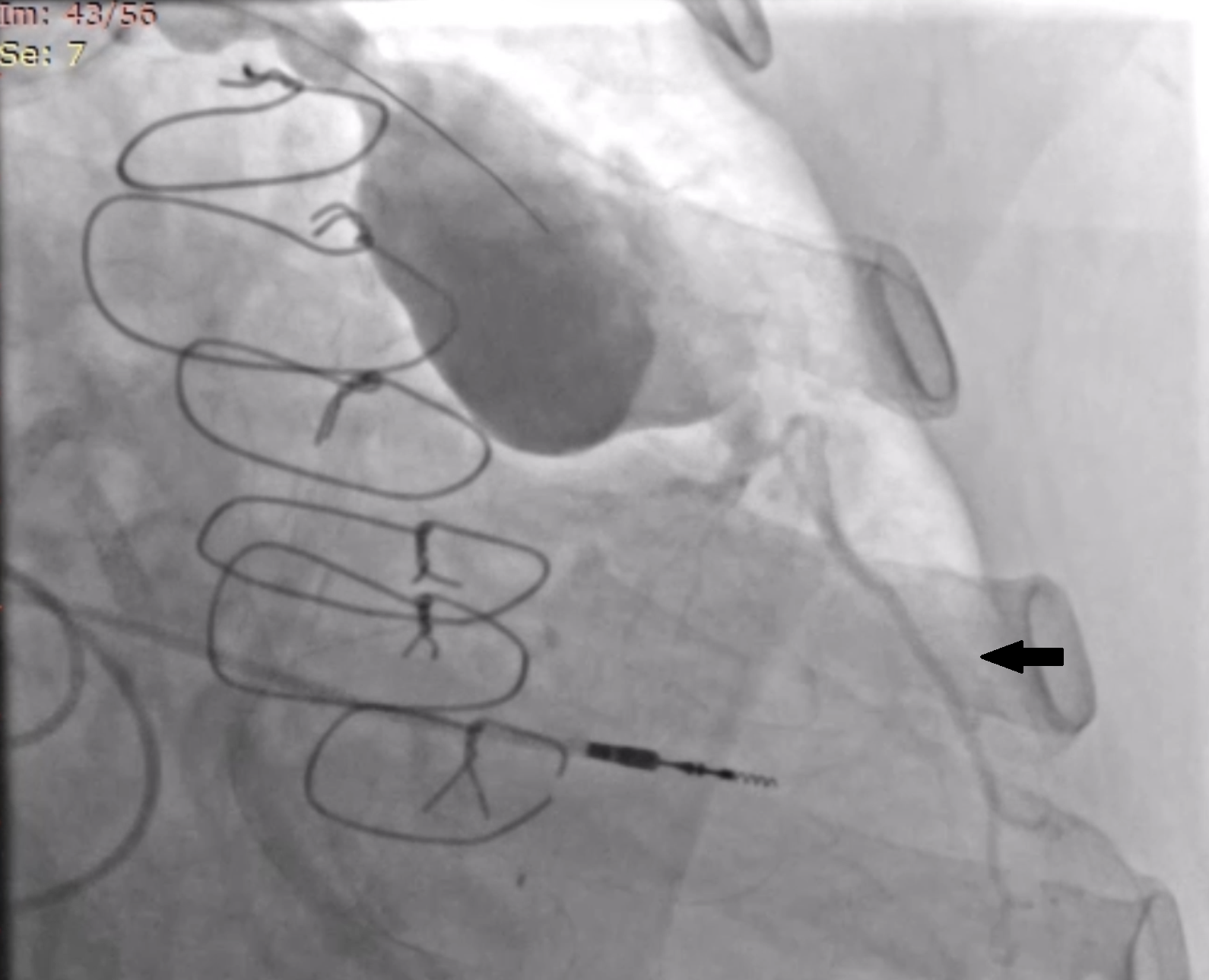
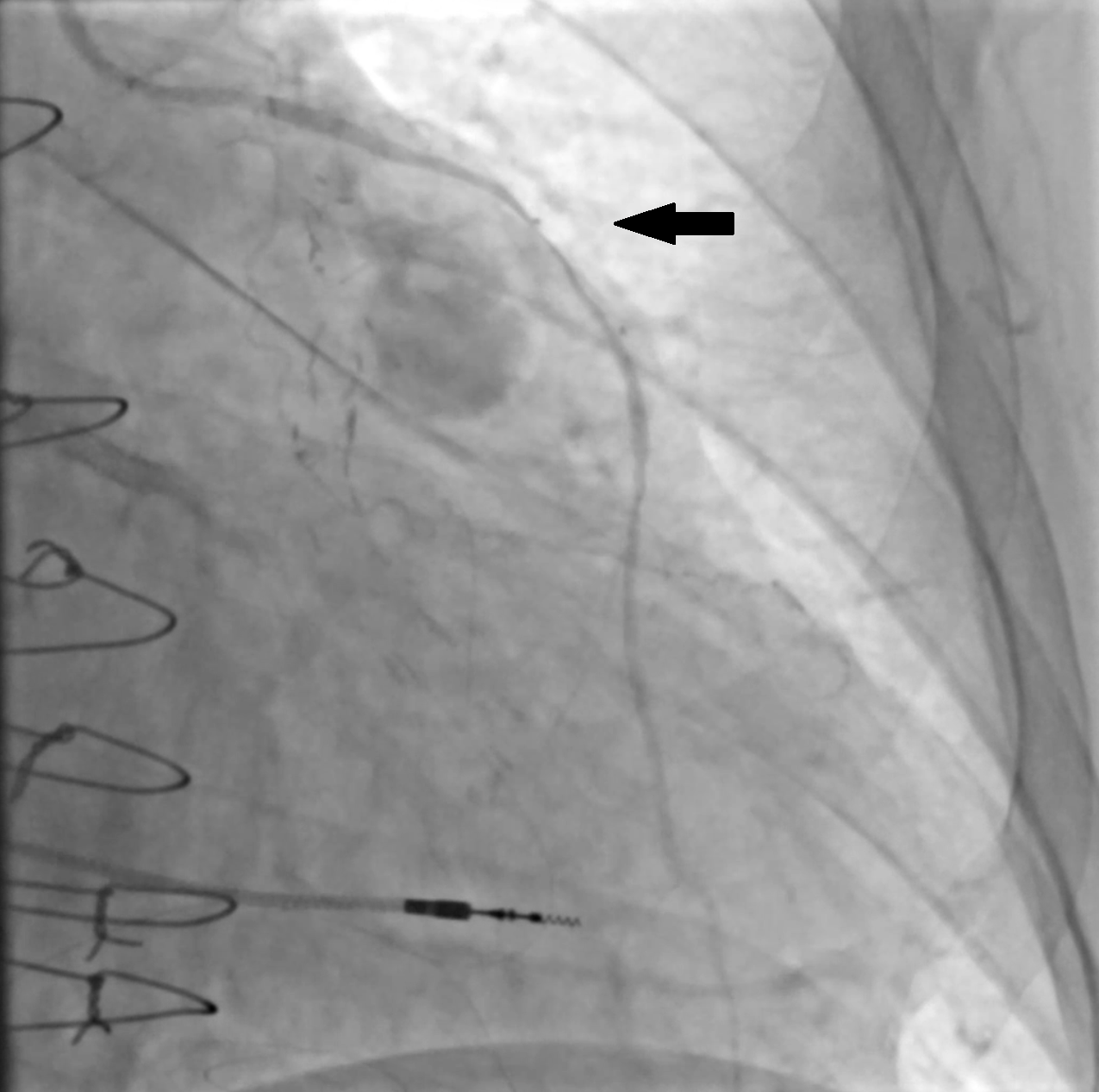
Baseline coronary angiography revealed a large aneurysm of the SVG to D2, with a patent SVG to OM1 proximal to the origin of the aneurysm (Figure 1, Videos 1 and 2). Through a left coronary bypass (LCB) guide catheter, a guide extension catheter was advanced inside the aneurysm sack to allow visualization of the distal vessel segment, demonstrating a small caliber vessel (Figure 2, Video 3). The LIMA graft was patent; however, possible external compression of the graft by the SVG aneurysm was identified (Figure 3, Video 4).
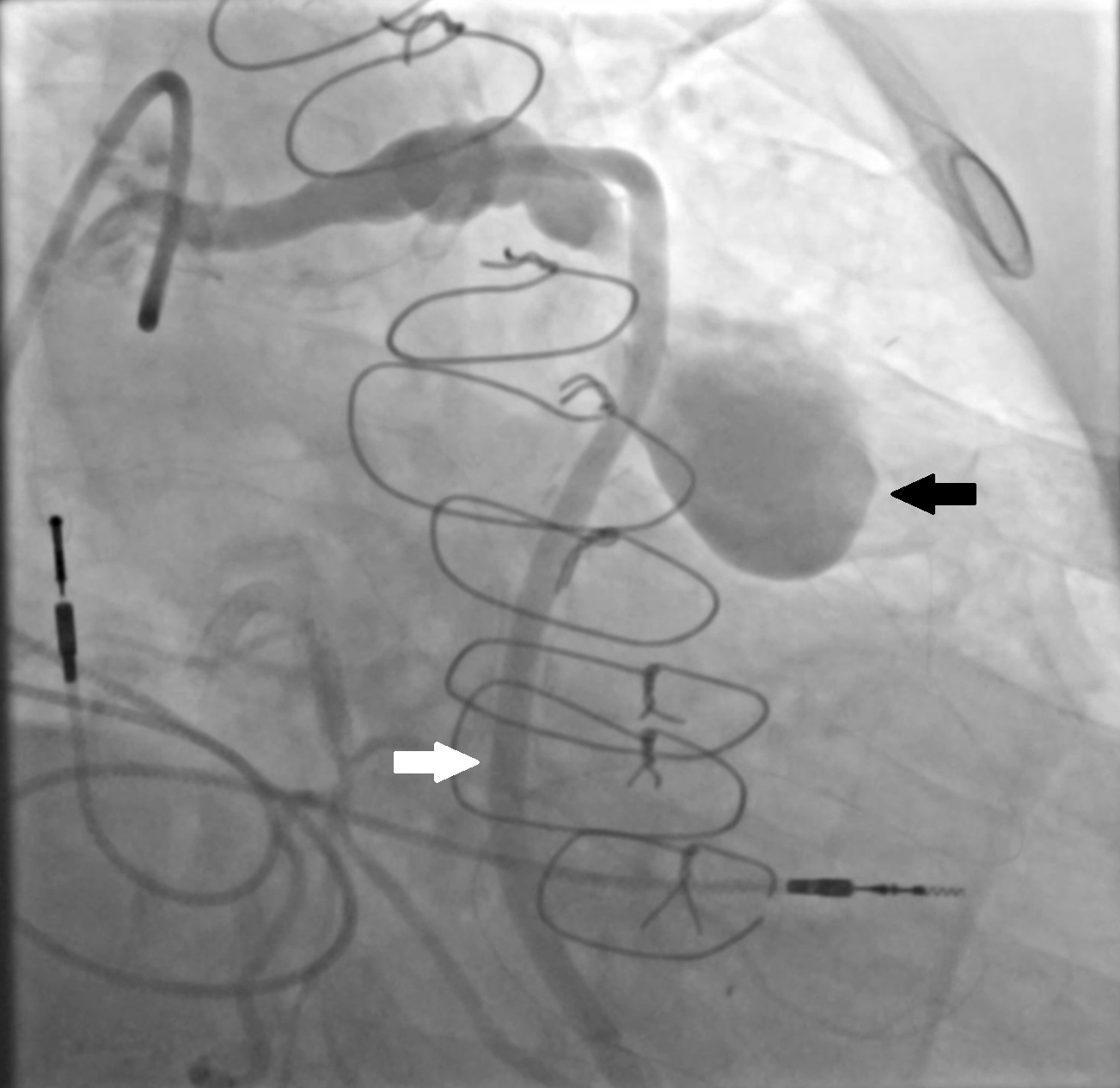
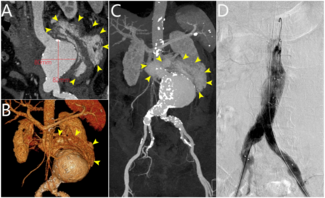
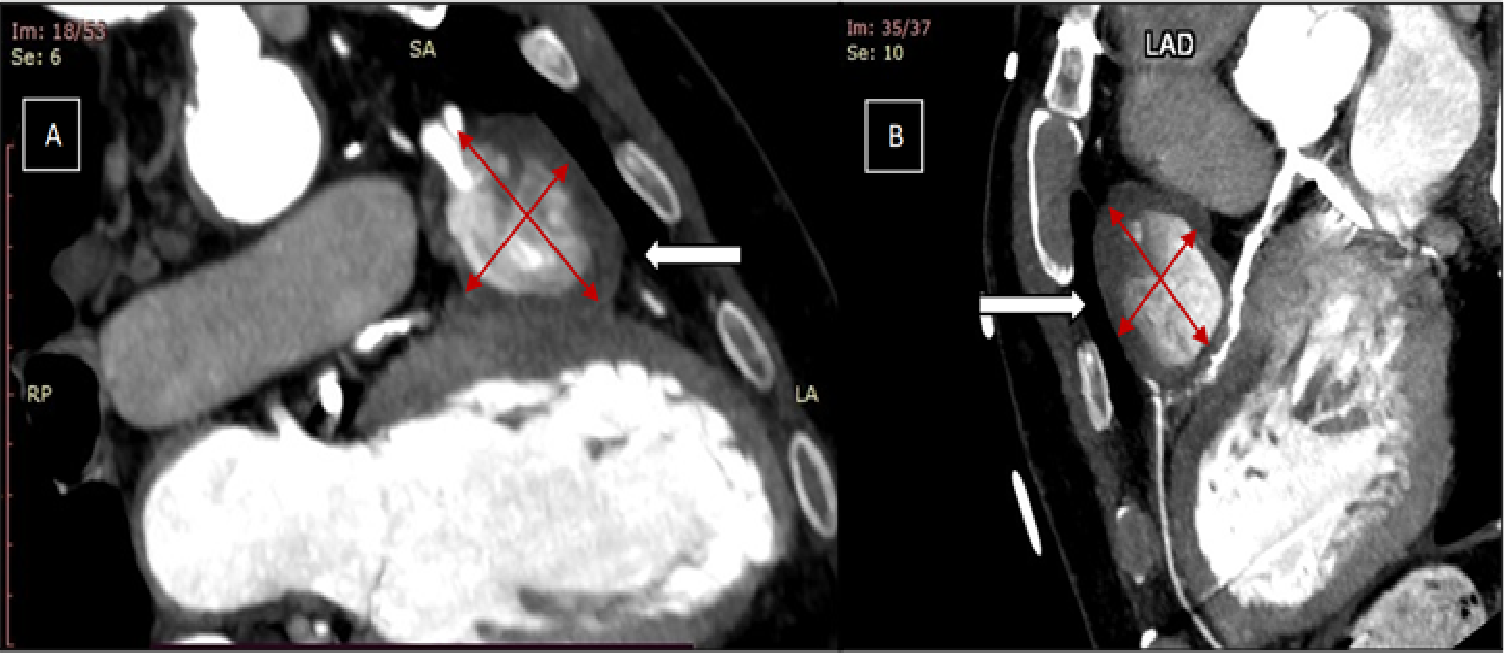
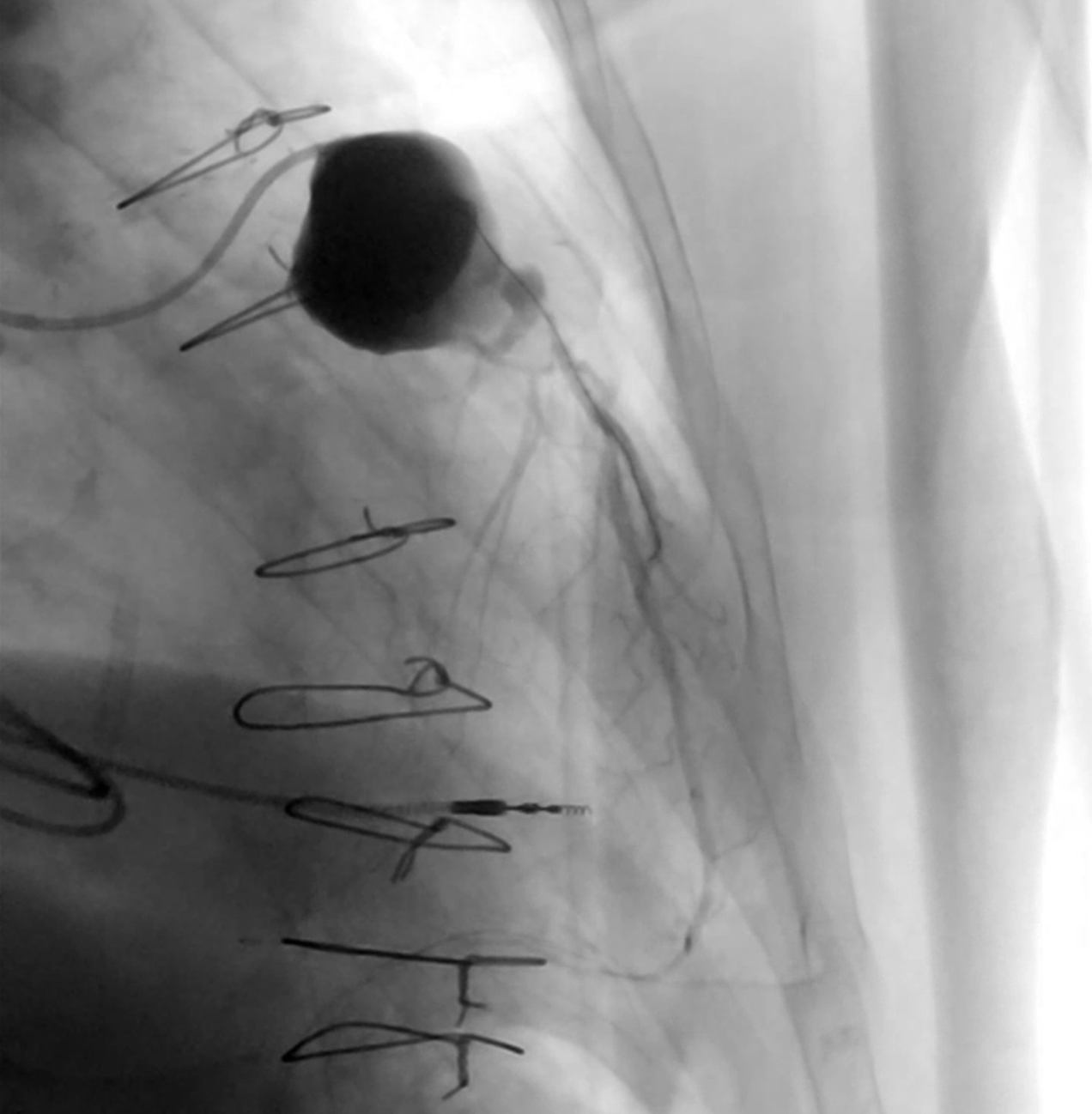
Coronary computed tomography angiography confirmed the presence of a 51 x 39-mm partially thrombosed SVG aneurysm (Figure 4). Transcatheter closure was attempted utilizing a 6F LCB guide catheter to engage the degenerated SVG. Using a 6F guide extension catheter and a Finecross microcatheter (Terumo), a 0.014-inch workhorse guidewire was advanced through the aneurysm into the D2 (Figure 5, Video 5). A 4.0 x 10-mm Prestige Plus peripheral vessel coil (Balt) was deployed, causing successful occlusion of the outflow segment of the aneurysm (Figure 6, Video 6). Subsequently, multiple similar coils were deployed, with successful occlusion of the inflow segment (Figure 7, Video 7).
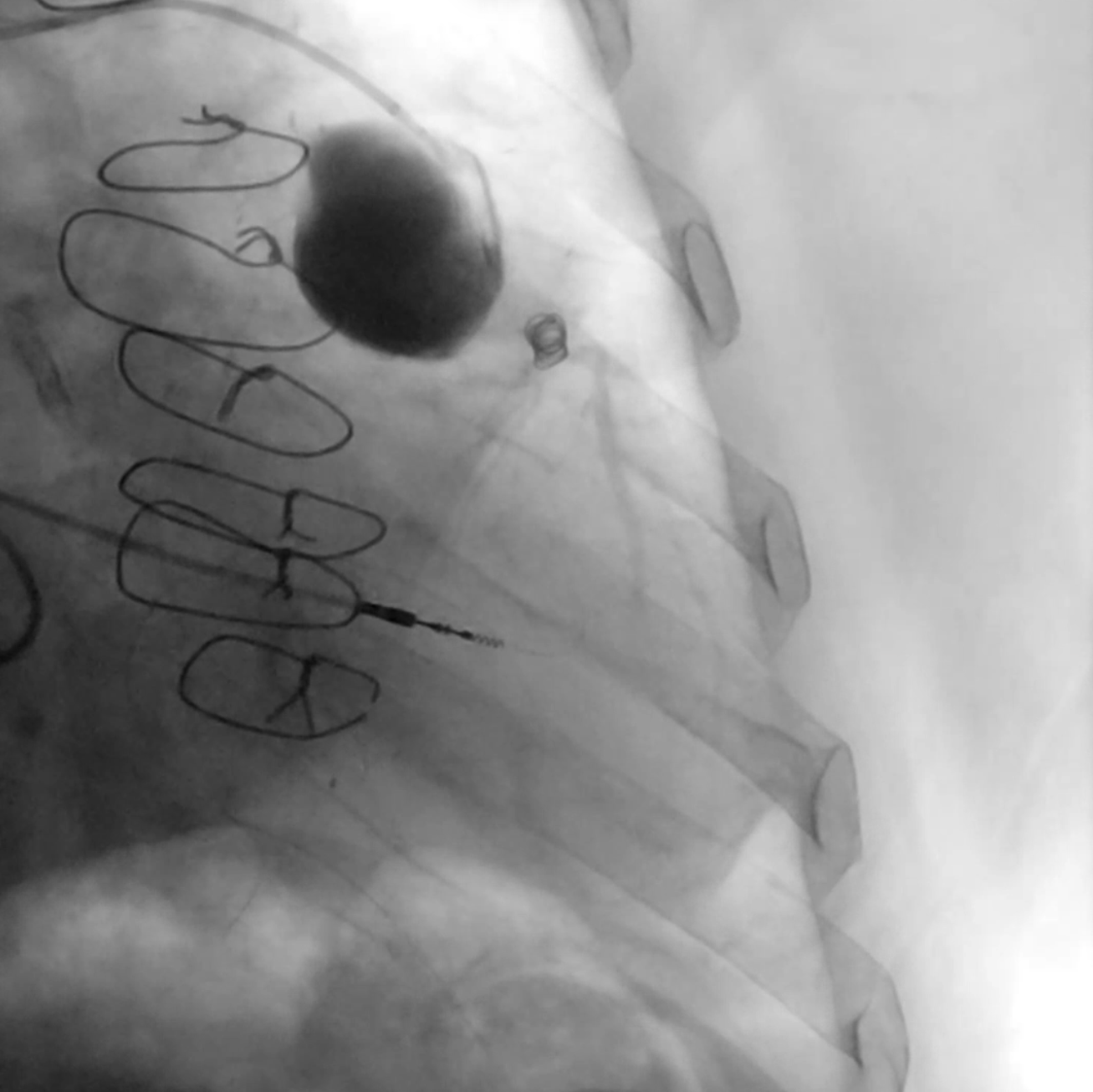
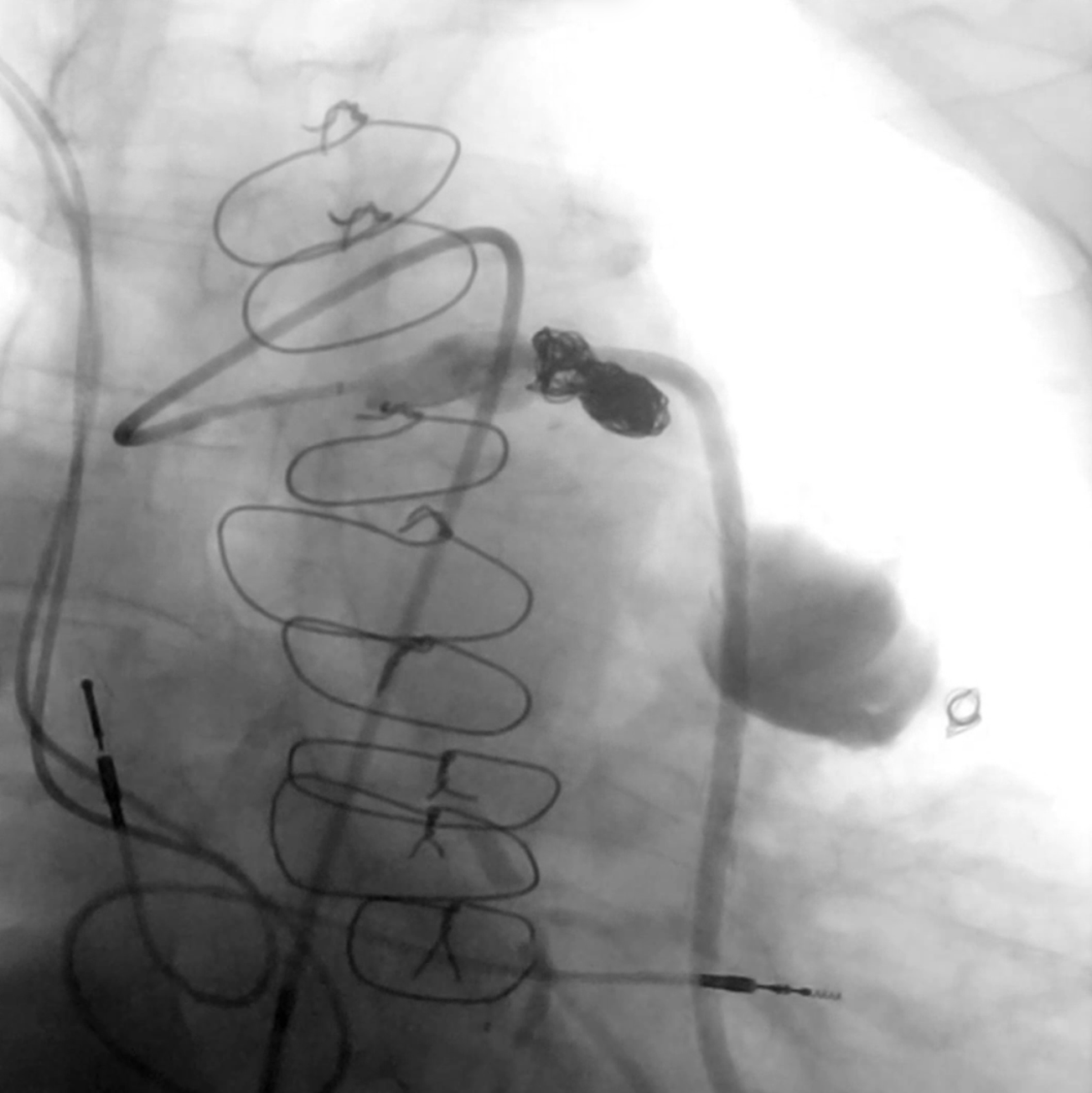
The patient was discharged uneventfully on day 2. Unfortunately, he developed chest pain and out-of-hospital cardiac arrest a few hours after discharge. Despite resuscitation efforts, he passed away. Postmortem examination showed anterior wall myocardial infarction, probably attributed to external compression of the LIMA by the aneurysm sack.
Affiliations and Disclosures
Antonios Lysandrou, MD1; Konstantinos Kalogeras, MD, PhD1; Maria Ioanna Gounaridi, MD, PhD1; George E. Zakynthinos, MD1; Ioannis Gialamas, MD1; Nikolaos Vythoulkas-Biotis, MD1; Efstratios Katsianos, MD1; Chrysovalantis Psathas, MD2; Gerasimos Siasos, MD, PhD1; Emmanouil Vavouranakis, MD, PhD1
From the 13rd Department of Cardiology, Sotiria Chest Diseases Hospital, Medical School, National and Kapodistrian University of Athens, Athens, Greece; 2Mediterraneo Hospital, Glyfada, Greece.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Consent statement: The authors confirm that informed consent was obtained from the patient for the intervention described in the manuscript and for the publication thereof, including any and all images.
Address for correspondence: Antonios Lysandrou, MD, 3rd Department of Cardiology, Sotiria Chest Diseases Hospital, Medical School, National and Kapodistrian University of Athens, 152 Mesogion Avenue, Athens 11527, Greece. Email: ant.lysandrou@hotmail.com