Comparison of Excimer Laser Coronary Atherectomy as a Sole Device or as Part of a Multimodality Technique
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
Abstract
Objectives. There are limited data on the use of excimer laser coronary atherectomy (ELCA) in conjunction with other calcium modification devices (intravascular lithotripsy [IVL], rotational/orbital atherectomy [RA/OA]). The aim of this analysis was to compare the use of ELCA as a sole device for coronary intervention with ELCA in combination with additional calcium modification devices.
Methods. This was a retrospective analysis of all patients treated with ELCA (either as a sole modification device or in conjunction with another calcium modification device) at a single high-volume center. Data and comparisons between ELCA alone and each of the combination therapies (with IVL, with RA/OA, with both IVL and RA/OA) were presented and compared using statistical methods appropriate to the data type.
Results. This analysis included 98 interventions using ELCA (67 as a sole device, 22 with IVL, 6 with RA/OA, 3 with IVL and RA/OA). ELCA alone or in conjunction with IVL were most utilized for in-stent restenosis/underexpansion compared with ELCA in conjunction with RA/OA +/-IVL, which were used more frequently for uncrossable/calcified lesions. The frequency of coronary artery perforation across the entire cohort was 4.1%. Target vessel revascularization frequency was 9.2%, and target vessel myocardial infarction was 3.1% at a median of 1051 days, with no statistically significant differences between the device groups.
Conclusions. ELCA combination therapies have a potential role in certain complex cases and though these are associated with higher risk, they can be safely performed in selected centers using radial access with good medium-term outcomes.
Introduction
The use of multimodality coronary plaque modification techniques has grown recently. This is partly due to the increasing complexity of cases because of aging populations, but also due to the development of newer modification techniques, particularly intravascular lithotripsy (IVL).1 There has been a rapid uptake in the use of IVL and it already has a central role in the calcium modification algorithm because of its ease of use, excellent safety profile, and efficacy.2-5 While the adoption of IVL has been widespread, excimer laser coronary atherectomy (ELCA) has been available for decades; however, because of the cost of acquiring/maintaining the console and consumable catheters, as well as the learning curve, this technology has mainly been limited to a few centers.6 One of the main practical advantages of ELCA is that these devices can be delivered over a standard 0.014-inch workhorse wire, which means that the device can be used when other, bulkier devices cannot cross or when it is difficult to deliver other atherectomy wires.6
There has been much interest in the combined use of atherectomy devices (both rotational and orbital) with IVL because of the practical and potential synergistic advantages of these combinations.7-10 The main advantage of this combination approach is the ability of atherectomy devices to create a channel to deliver the bulkier IVL device. There are, however, circumstances for which either orbital (OA) or rotational atherectomy (RA) are not well suited. ELCA may have a role in these cases, whether as a sole device or in conjunction with IVL, OA, or RA.11 Furthermore, a developing indication for IVL is for the treatment of underexpanded stents due to calcification. ELCA also has an established role in these cases, and, therefore, there has been interest in the combination of ELCA and IVL for this indication.6,12,13 Despite the potential clinical utility of a combination IVL and ELCA strategy, there is only a single case report describing its use and limited data on the use of ELCA with RA/OA or ELCA with IVL and OA/RA.1,14-16
The aim of this single-center analysis was to review the use and outcomes of a combination therapy using ELCA, whether that be ELCA combined with IVL (termed ELCA shock), ELCA combined with OA/RA (termed RASER), or ELCA combined with both IVL and RA/OA (termed RASER shock) compared with cases treated with ELCA alone.
Methods
Study participants and data collection
This analysis retrospectively included all patients treated with ELCA alone and ELCA combination therapies at the Royal Bournemouth Hospital (University Hospital Dorset, United Kingdom). All patients treated with ELCA from the introduction of IVL in January 2019 until the end of September 2024 were included in this analysis. In the cases of combination therapy using ELCA, the devices must have been used for treatment of the same lesion to be considered a combination therapy. Approval for the analysis was received from the local audit/service evaluation group. Baseline demographics, comorbidities, and procedural details were obtained from the clinical record, which is contemporaneously documented as part of the British Cardiovascular Intervention Society database. Each clinical record was then reviewed for further procedural details, specifically relating to the use of ELCA and IVL/OA/RA and the timing and indication for use of each device. Further, each case was reviewed to assess whether there were any procedural complications, target vessel revascularization (TVR), target vessel myocardial infarction (TVMI), or mortality during clinical follow-up.
Procedure
This was a retrospective analysis and, therefore, all aspects of the percutaneous coronary intervention (PCI) procedures were at the discretion of the treating interventional cardiologist. In our center, the use of intracoronary imaging (ICI) is well established and highly encouraged, particularly in complex PCI cases. Saline was the preferred ELCA flush strategy, with contrast flushes or blood only reserved for highly resistant lesions. The sizing of IVL balloons was at the discretion of the performing cardiologist but with the aim of a balloon to artery ratio of 1:1. Further, both the number of IVL balloons and pulses was also decided by the supervising cardiologist. This analysis includes both the standard Shockwave C2 IVL balloons (with 80 pulses available) and the Shockwave C2+ IVL balloons (with 120 pulses available) (Shockwave Medical). The RA (burr sizing, speed, and number of runs) and OA (speed and number of runs) strategies were similarly at the discretion of the supervising cardiologist.
Analysis
Descriptive statistics are reported as the number (percentage of the total) for categorical variables and as the median (interquartile range [IQR]) for continuous variables. Comparisons between ELCA alone and each of the combination therapies were undertaken using the chi-square test for categorical variables and the Kruskal-Wallis test for continuous variables. Cases in which patients had more than 1 qualifying event were analyzed on a per-case basis for procedural characteristics and procedural outcomes (complications/TVMI/TVR) and on a per-patient basis for baseline demographics and mortality. Comparisons between mortality and TVMI/TVR across the device combinations are presented using Kaplan Meier curves with log-rank analysis. The decision making behind each of the strategies is presented as a flow chart for each multimodality calcium modification strategy by clinical indication (calcification/uncrossable and in-stent restenosis/stent underexpansion). All analyses were performed using SPSS v29.0 (SPSS, IBM Corporation).
Results
Patient characteristics
There were 94 patients treated with ELCA alone or a combination therapy with ELCA between January 2019 and September 2024 for a total of 98 PCI procedures; 2 patients had 2 procedures, and 1 patient had 3 procedures. The cohort was predominantly male with 23 (24.5%) females and a median age of 72 years (IQR 65-79 years). Most patients in the cohort had a history of previous revascularization and significant cardiovascular comorbidity (Table 1). The only significant difference between the baseline characteristics was that patients treated with ELCA or ELCA shock were more likely to have had previous PCI (Table 1).
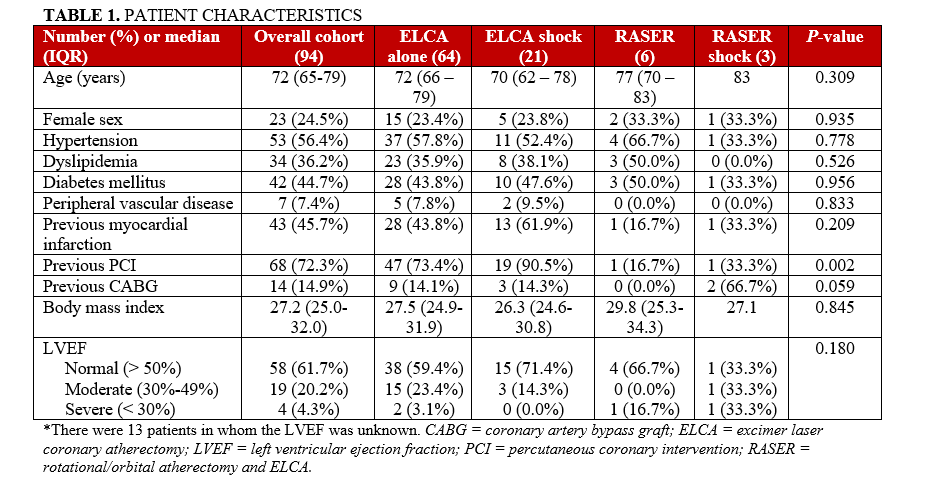
Procedural characteristics
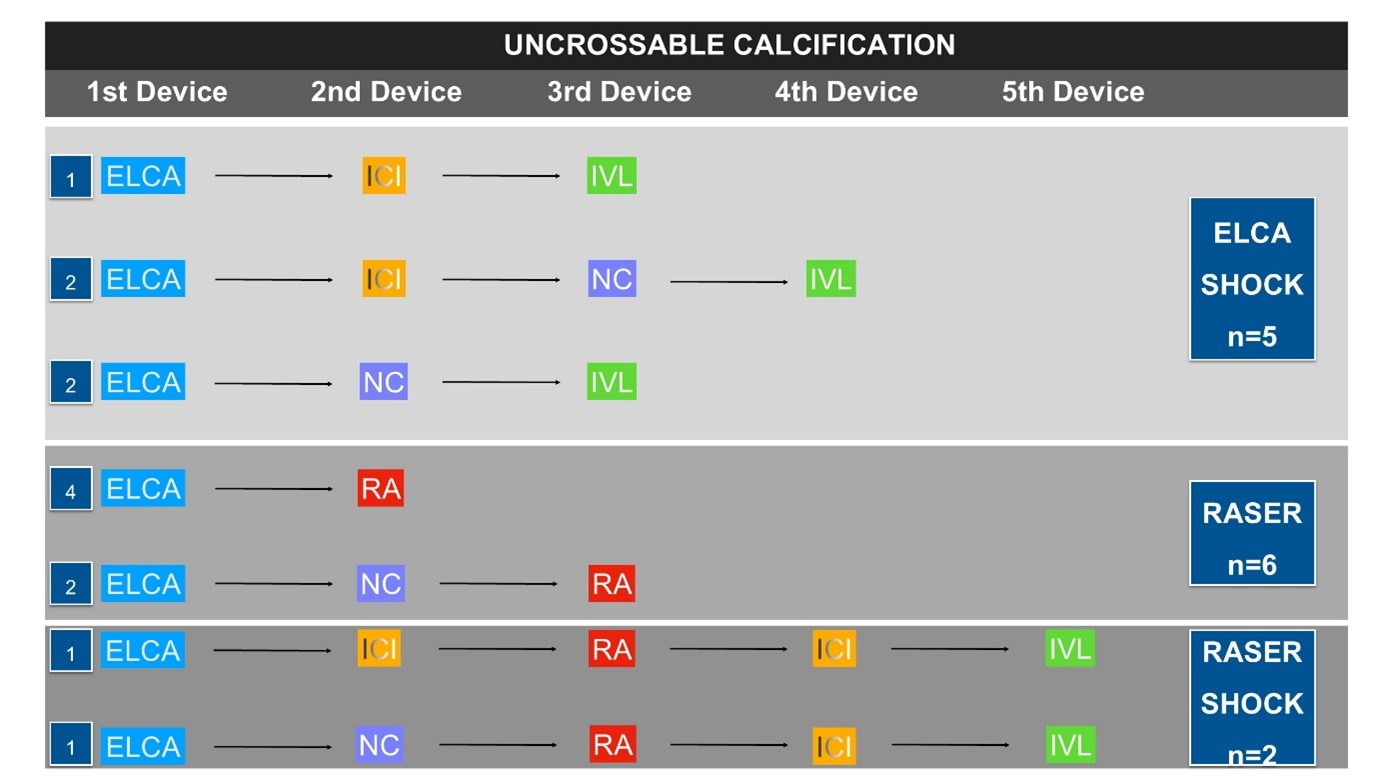
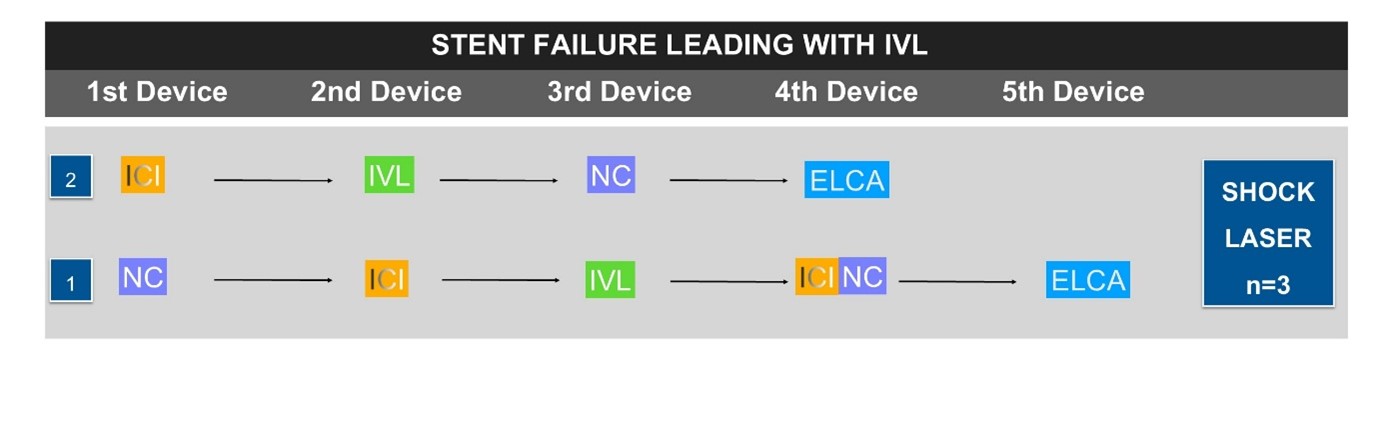
Unsurprisingly, most of these cases were planned electives (n = 67 [68.4%]). The indication for device utilization varied between the different device combinations. ELCA and ELCA shock were most frequently used in stent underexpansion/restenosis (n = 35 [52.3%] and n = 17 [77.3%], respectively). RASER and RASER shock tended to be utilized in uncrossable/undilatable calcified lesions (n = 6 [100.0%] and n = 2 [66.7%], respectively). Only ELCA as a single device was utilized in thrombus in 8 (11.9%) cases (Table 2). The left anterior descending artery (LAD) was the most frequent PCI target, followed by the right coronary artery (RCA) (n = 40 [40.8%] and n = 37 [37.8%], respectively). The majority of procedures were performed solely via the radial artery (n = 84 [85.7%]), although the frequency was significantly lower in the RASER shock cohort (n = 1 [33.3%]). Most cases were performed with a 6F guiding system (n = 79 [80.6%]) (Table 2). ICI was used in 77 (78.6%) cases, which was consistent across the different devices, except in RASER (n = 2 [33.3%]) (Table 2). Cutting ballons (CB)/scoring balloons were also frequently utilized (n = 48 [49.0%]), although this was 0.0% in RASER cases. Figures 1 through 3 highlight the decision-making steps at each stage of the multimodality calcium modification cases. ICI forms a major part of the PCI guidance process of these procedures, but, unsurprisingly, it is used later in the uncrossable cohort compared with the stent-failure group, where it is almost universally used as a first device.
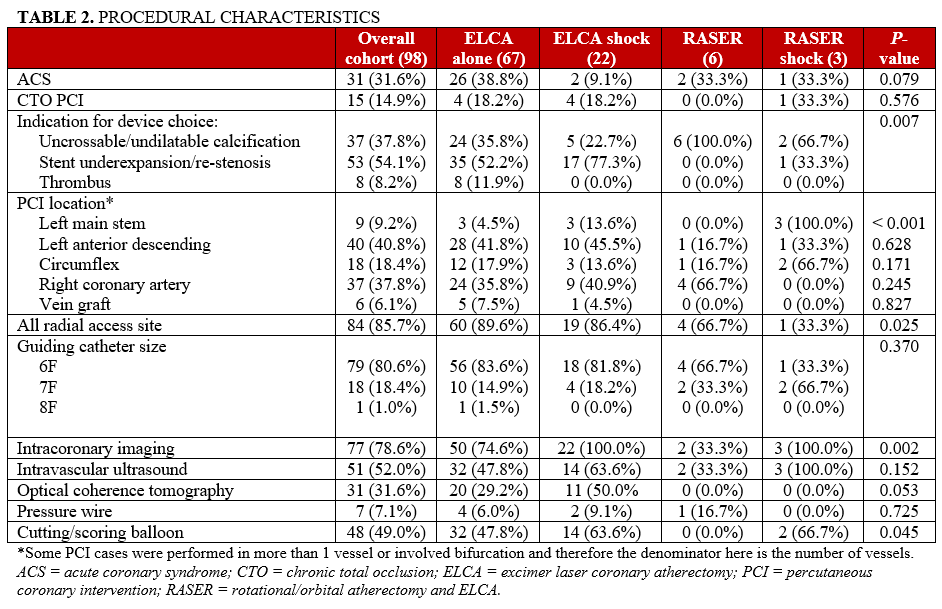

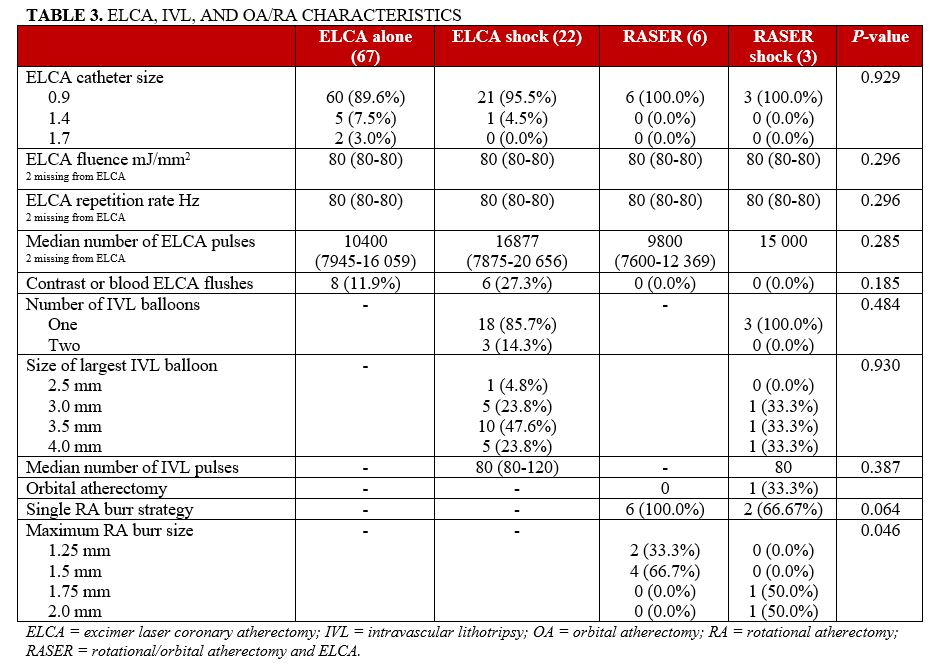
ELCA, IVL, and RA/OA characteristics
A 0.9-mm ELCA catheter was the most frequently utilized (n = 90 [91.8%]) and, as a result, the median fluence was 80 mJ/mm2 and the median repetition frequency was 80 Hz (Table 3). A single IVL balloon was used in 21 (87.5%) cases, incorporating IVL with a median of 80 pulses. There was a single case of OA included in the analysis, which formed part of a RASER shock strategy during which OA was used at both low and high speed. The RA burr size was larger in the RASER shock cohort compared with the RASER group (Table 3). There was no difference in terms of complications between the different device combinations (Table 4).
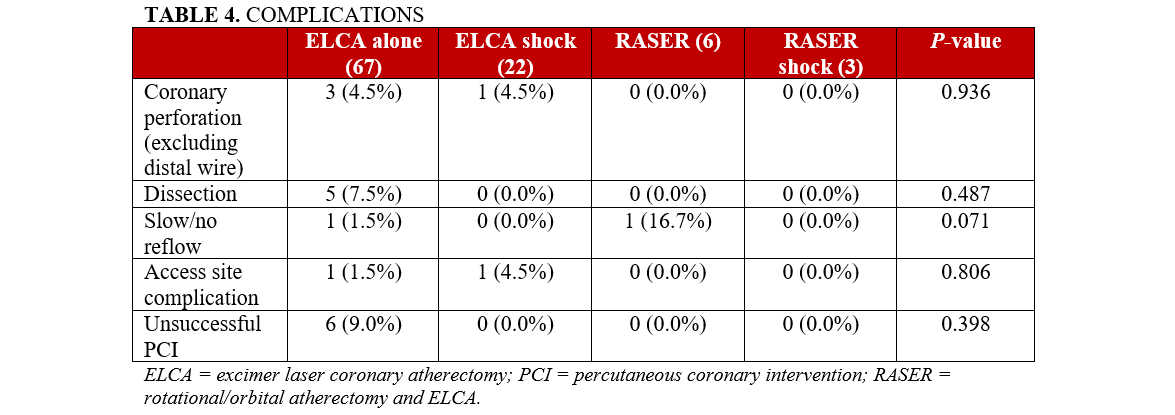
Procedural and medium-term outcomes
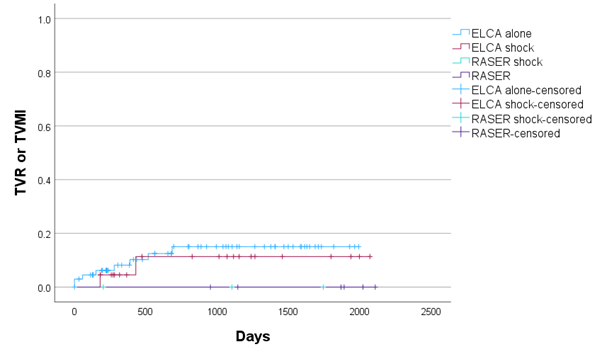
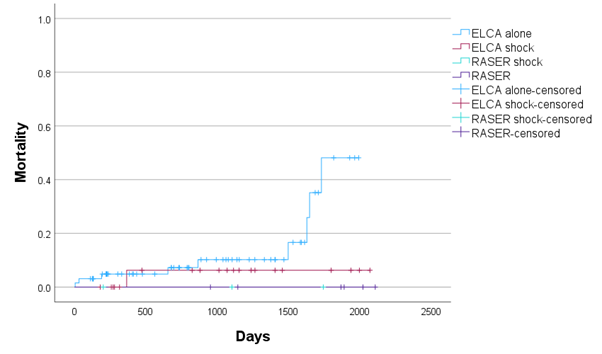
There were 6 (6.1%) cases where it was not possible to complete the procedure because of the complexity of the disease. There were 4 cases of coronary perforation with an additional distal wire perforation. Two of the coronary perforations required covered stent implantation and both patients subsequently died in hospital. Upon review, 2 cases of coronary perforation were caused by postdilatation, one was caused during ELCA use and one was caused either during ELCA or predilatation. At the median clinical follow-up of 1074 days (IQR 411-1585 days), there were 10 deaths but no significant differences between the device combinations (Figure 4). There were 9 cases of TVR and 3 cases of TVMI, with no differences between the device combinations when TVMI and TVR were considered separately or combined first event (Figure 5).
Discussion
This retrospective single-center analysis of patients treated with either ELCA alone or ELCA as part of a combination therapy shows that, while these techniques are only utilized infrequently for complex higher risk cases, they are associated with durable outcomes despite this complexity. ELCA as part of a combination therapy is reserved for complex in-stent restenosis/underexpanded stents and uncrossable/undilatable calcified lesions.
A combination strategy for managing complex calcification has been utilized in a number of different forms. Each of these strategies aims to utilize the crossing ability of 1 calcium modification tool (atherectomy devices) to allow delivery of a bulkier device that offers a different modification mechanism (eg, IVL or CBs). There are a number of case reports and series demonstrating that the use of RA and OA devices with IVL is safe and associated with good outcomes, but, to date, there are no randomized data.7-10,17,18 Similarly, the combination of RA with CBs, termed rota-cut, was found to be potentially efficacious in some published cohorts; however, in the first randomized study (ROTA-CUT), there was no difference in the use of rota-cut compared with RA performed with standard balloons.19,20
While there has been considerable interest in the combination of RA and OA devices with IVL or CBs, there have been few reports of combinations with ELCA, which is likely driven by the small number of centers with access to this device. The British Cardiovascular Interventional Society (BCIS) presented a cohort of all cases treated with RA and ELCA (termed RASER) from their database.15 The frequency of RASER in the database was low (0.02%), demonstrating the limited access and niche use of this device. The use of a RASER strategy was associated with a higher frequency of complications compared with the rest of the cohort, but there was no increase in in-hospital major adverse cardiovascular events.15
The data regarding the potential combination of ELCA and IVL are particularly limited. Until the recent ROLLER COASTR trial, there was only a single case report where the combination of ELCA and IVL was utilized.12 In that case, a proximal stent had been implanted and then the patient was brought back for treatment of a more distal calcified lesion. An IVL balloon would not cross this lesion and, because of the recent proximal stent, RA or OA would not be favorable; thus, ELCA was utilized to create a channel for the IVL balloon. This case demonstrates the potential role of ELCA in creating a channel for the delivery of the IVL balloon in situations for which other atherectomy devices are less suited.11,14 In the recently published ROLLER COASTR trial, 171 patients with moderate-severe calcification were randomized to either IVL, RA, or ELCA. In this trial, 11.7% of cases were performed, with 2 of these devices demonstrating the important role of combination therapies in calcium modification.18 Interestingly, the majority of these dual modality cases involved a combination with ELCA (60.0%). RA was utilized in cases where there was difficulty crossing the lesion, and IVL was used in cases of undilatable lesions.18
In our cohort, most cases were able to be performed with sole radial access utilizing a 6F guiding system, which highlights one of the potential advantages of a combination strategy.1 Conceptually, the combination with IVL allows the use of smaller RA burr sizes because of the dual modality10; this has been demonstrated in previous cohorts. However, in our current analysis, the opposite was seen—the patients treated with RASER had smaller RA burr sizes when compared with the RASER shock cohort. While this does not support one of the potential conceptual advantages of multimodality calcium modification strategies involving IVL, the numbers of both RASER and RASER shock in this analysis are too small to confirm this conceptual advantage.
The figures demonstrating the decision making behind the ELCA combination therapies highlight the central use of ICI for decision making. The data are overwhelmingly clear: ICI is required for complex PCI cases, and our analysis highlights the value of this at each stage of the decision-making process.21-23 Unsurprisingly, ICI was used at different stages in the cases of stent failure vs uncrossable lesions, with almost all the stent-failure cases having ICI before any treatment unless the lesion was uncrossable. We would consider the use of ICI essential to understand the mechanisms behind stent failure. Furthermore, the stent-failure flow diagrams highlight the complexity of the decision-making strategies within this cohort. This observation suggests that, especially given the relatively low volume of complex stent-failure cases, it is appropriate to reserve ELCA for a few centers that can develop expertise in stent failure and, as such, receive regional referrals rather than multiple centers performing smaller volumes of these cases.
In our cohort, 3 out of a total of 37 cases with uncrossable/undilatable calcification involved the use of ELCA shock. Most of the remaining cases were performed with ELCA alone (64.9%). ELCA is a very useful technique for uncrossable lesions because it can be delivered on a standard 0.014-inch workhorse wire and, as such, is particularly helpful in creating a channel in cases in which it would not be possible to deliver a microcatheter or to direct wire to deliver other atherectomy devices. The effect of ELCA on calcium is restricted to the sonic wave effect rather than ultraviolet light, which passes through and does not alter the calcium.6 The sonic wave itself can be amplified using blood or contrast to provide more potent outward mechanical disruption.6 Despite this ability, ELCA sometimes cannot deliver enough modification to deeper calcification to provide an optimal result. This is reflected in the 35.1% of calcification cases in our series that required combination therapy (with IVL alone, 13.5%; part of RASER/RASER shock, 21.6%). The authors would recommend the use of ELCA in coronary calcification mainly for cases where the calcification is uncrossable with a microcatheter or where it is not possible to direct wire for other atherectomy devices.
The majority of ELCA shock/RASER shock cases in our series were for the treatment of underexpanded stents. ELCA is a well-established modality for the treatment of underexpanded stents, and there are growing data that IVL is also an effective strategy for treating underexpanded stents due to calcification.6,12,13,24,25 Beyond ELCA providing a channel for delivery of the IVL balloon, it is plausible that the combination of these strategies with their different action mechanisms for stent underexpansion may result in improved procedural and clinical outcomes for these patients. While the data from our cohort demonstrate encouraging results, further and longer-term data are required, particularly given that previous analyses of ELCA for stent failure have shown high TVR rates.26 This previous observation is likely a result of the complexity of stent-failure cases where ELCA is required (including multiple stent layers and often the use of ELCA as a last resort for stent-failure cases).
While there are some data to support the use of combination therapies, there are limited randomized data so far showing outcome advantages with such a strategy. It is possible that, in the future, a combination therapy may show improved results over a single device; however, the routine use of combination therapies has significant cost implications and, as such, should be reserved for cases where a single device provides insufficient modification to achieve an optimal procedural result. The authors would therefore recommend a single-device strategy where possible, with combination therapies reserved for cases where a single device does not achieve satisfactory results.
Limitations
There are a number of limitations worth considering when interpreting this analysis. The first and most important factor is that this is a single-center, retrospective analysis and, therefore, has all the limitations associated with this study design, particularly the risk of selection bias. Second, a significant proportion of the data used in this analysis was recorded as part of the BCIS database; while this is contemporaneously recorded, there is no independent cross-referencing to ensure accuracy. Third, this is a small cohort, which is unsurprising given the niche potential role of ELCA-combination therapies. Finally, our center often receives referral from hospitals outside our catchment area for the management of restenosis because of our ELCA program. If these patients re-presented to their base hospital with TVR and this was not communicated to our center, our analysis would not record that TVR. It is, therefore, possible that the TVR/TVMI frequency in this analysis is an underestimate.
Conclusions
The combination of ELCA and IVL is an effective and safe option for managing coronary calcification for which other atherectomy devices are less well suited, particularly for stent underexpansion. The medium-term follow-up data suggest good outcomes for these patients; however, further data are required to confirm the safety, efficacy, and durability of these strategies.
Affiliations and Disclosures
Jonathan Hinton, MD; William Carr, MBBS; Natasha Khullar, MBBS; Trisha Singh, PhD; Vivek Kodoth, MD; Jehangir Din, MD; Peter O’Kane, MD
From the Dorset Heart Centre, Royal Bournemouth Hospital, Castle Lane East, Bournemouth, United Kingdom.
Disclosures: Dr Hinton has received support to attend educational events from MedAlliance, Terumo, Shockwave Medical, Amarin, Abbott Vascular, and Vascular Perspectives; has received speaker fees from Shockwave Medical, Amarin, Boston Scientific, Medtronic, and Cordis; and has research relationships, including financial, with Abbott Vascular and Beckman Coulter. Dr O’Kane has received speaker fees from Abbott Vascular, Boston Scientific, Philips, Medtronic, Shockwave Medical, and Terumo; and has a research relationship with Abbott Vascular. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Jonathan Hinton, MD, Dorset Heart Centre, Royal Bournemouth Hospital, Castle Lane East, Bournemouth BH77DW, United Kingdom. Email: jonathan.hinton1@nhs.net
References
- Hinton J, O’Kane P. Combination tools for calcium modification from RASER to Orbitalshock. Interv Cardiol. 2024;19:e18. doi:10.15420/icr.2024.24
- Honton B, Monsegu J. Best practice in intravascular lithotripsy. Interv Cardiol. 2022;17:e02. doi:10.15420/icr.2021.14
- Saito S, Yamazaki S, Takahashi A, et al; Disrupt CAD IV Investigators. Intravascular lithotripsy for vessel preparation in calcified coronary arteries prior to stent placement: Japanese Disrupt CAD IV study 1-year results. Circ Rep. 2022;4(9):399-404. doi:10.1253/circrep.CR-22-0068
- Hill JM, Kereiakes DJ, Shlofmitz RA, et al; Disrupt CAD III Investigators. Intravascular lithotripsy for treatment of severely calcified coronary artery disease. J Am Coll Cardiol. 2020;76(22):2635-2646. doi:10.1016/j.jacc.2020.09.603
- Ali ZA, Nef H, Escaned J, et al. Safety and effectiveness of coronary intravascular lithotripsy for treatment of severely calcified coronary stenoses: the Disrupt CAD II study. Circ Cardiovasc Interv. 2019;12(10):e008434. doi:10.1161/CIRCINTERVENTIONS.119.008434
- Rawlins J, Din JN, Talwar S, O’Kane P. Coronary intervention with the excimer laser: review of the technology and outcome data. Interv Cardiol. 2016;11(1):27-32. doi:10.15420/icr.2016:2:2
- Sardella G, Stefanini G, Leone PP, et al. Coronary lithotripsy as elective or bail-out strategy after rotational atherectomy in the Rota-Shock registry. Am J Cardiol. 2023;198:1-8. doi:10.1016/j.amjcard.2023.04.032
- Wong B, Kam KKH, So CY, et al. Synergistic coronary artery calcium modification with combined atherectomy and intravascular lithotripsy. J Invasive Cardiol. 2023;35(3):E128-E135. doi:10.25270/jic/22.00272
- Gonzálvez-García A, Jiménez-Valero S, Galeote G, Moreno R, López de Sá E, Jurado-Román A. “RotaTripsy”: combination of rotational atherectomy and intravascular lithotripsy in heavily calcified coronary lesions: a case series. Cardiovasc Revasc Med. 2022;35:179-184. doi:10.1016/j.carrev.2021.04.011
- Hinton J, Varma R, Din J, Kodoth V, Talwar S, O’Kane P. ‘RotaShock’—a revolution in calcium modification: long-term follow-up from a single high-volume centre. Interv Cardiol. 2025;20:e08. doi:10.15420/icr.2024.34
- Vera-Vera S, Garcia A, Jimenez-Valero S, Galeote G, Moreno R, Jurado-Roman A. Excimer laser coronary atherectomy to treat calcified lesions. Interv Cardiol. 2022;17:e14. doi:10.15420/icr.2022.10
- Hinton J, Mariathas M, Chan E, et al. Novel application of intravascular lithotripsy in stent under-expansion: a single-center experience. Catheter Cardiovasc Interv. 2023;101(2):243-249.doi:10.1002/ccd.30516
- Tovar Forero MN, Sardella G, et al. Coronary lithotripsy for the treatment of underexpanded stents: the international multicentre CRUNCH registry. EuroIntervention. 2022;18(7):574-581. doi:10.4244/EIJ-D-21-00545
- Jurado-Román A, García A, Moreno R. ELCA-Tripsy: combination of laser and lithotripsy for severely calcified lesions. J Invasive Cardiol. 2021;33(9):E754-E755. doi:10.25270/jic/21.00207
- Protty MB, Gallagher S, Farooq V, et al. Combined use of rotational and excimer LASER coronary atherectomy (RASER) during complex coronary angioplasty—an analysis of cases (2006–2016) from the British Cardiovascular Intervention Society database. Catheter Cardiovasc Interv. 2021;97(7):E911-E918. doi:10.1002/ccd.29377
- Egred M. RASER angioplasty. Catheter Cardiovasc Interv. 2012;79(6):1009-1012. doi:10.1002/ccd.23174
- Takahashi T, Menegus M, Choi H, et al. Complementary utility of intravascular lithotripsy with atherectomy for severely calcified coronary stenoses in contemporary practice. J Invasive Cardiol. 2023;35(1):E46-E54. doi:10.25270/jic/22.00310
- Jurado-Román A, Gómez-Menchero A, Rivero-Santana B, et al. Rotational atherectomy, lithotripsy, or laser for calcified coronary stenosis: the ROLLER COASTR-EPIC22 trial. JACC Cardiovasc Interv. 2025;18(5):606-618. doi:10.1016/j.jcin.2024.11.012
- Sharma SK, Mehran R, Vogel B, et al. Rotational atherectomy combined with cutting balloon to optimise stent expansion in calcified lesions: the ROTA-CUT randomised trial. EuroIntervention. 2024;20(1):75-84. doi:10.4244/EIJ-D-23-00811
- Allali A, Toelg R, Abdel-Wahab M, et al. Combined rotational atherectomy and cutting balloon angioplasty prior to drug-eluting stent implantation in severely calcified coronary lesions: the PREPARE-CALC-COMBO study. Catheter Cardiovasc Interv. 2022;100(6):979-989. doi:10.1002/ccd.30423
- Vrints C, Andreotti F, Koskinas KC, et al; ESC Scientific Document Group. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur Heart J. 2024;45(36):3415-3537. doi:10.1093/eurheartj/ehae177
- Gao XF, Ge Z, Kong XQ, et al; ULTIMATE Investigators. 3-year outcomes of the ULTIMATE trial comparing intravascular ultrasound versus angiography-guided drug-eluting stent implantation. JACC Cardiovasc Interv. 2021;14(3):247-257. doi:10.1016/j.jcin.2020.10.001
- Lee JM, Choi KH, Song YB, et al; RENOVATE-COMPLEX-PCI Investigators. Intravascular imaging-guided or angiography-guided complex PCI. N Engl J Med. 2023;388(18):1668-1679. doi:10.1056/NEJMoa2216607
- Egred M, Brilakis ES. Excimer laser coronary angioplasty (ELCA): fundamentals, mechanism of action, and clinical applications. J Invasive Cardiol. 2020;32(2):E27-E35. doi:10.25270/jic/19.00325
- Nan J, Joseph TA, Bell MR, Singh M, Sandoval Y, Gulati R. Outcomes of excimer laser-contrast angioplasty for stent underexpansion. EuroIntervention. 2021;17(1):78-80. doi:10.4244/EIJ-D-19-01074
- Hinton J, Tuffs C, Varma R, et al. An analysis of long-term clinical outcome following the use of excimer laser coronary atherectomy in a large UK PCI center. Catheter Cardiovasc Interv. 2024;104(1):27-33. doi:10.1002/ccd.31080