Coronary Intravascular Lithotripsy for Acute Stent Underexpansion: A Case Series
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
Introduction
Stent underexpansion due to heavy calcification increases the risk of acute stent thrombosis and in-stent restenosis.1 Plaque modification using atherectomy or intravascular lithotripsy (IVL) may not result in full balloon expansion in all cases. In such cases, post-stent implantation atherectomy may increase the risk of burr entrapment, stent avulsion, coronary dissection, and perforation.1 The use of IVL immediately following stent deployment was not studied in the DISRUPT-CAD studies.2 Here, we present a case series with off-label use of Shockwave IVL (Shockwave Medical) in acutely underexpanded stents.
Methods
The study was conducted at a large, high-volume academic medical center located in suburban Chicago, Illinois. Patients treated with IVL post-stent deployment for acute stent underexpansion from July 2024 to December 2024 were included in this case series. The research was performed in accordance with the appropriate ethical guidelines, and informed consent was waived because of the retrospective nature of the study. No generative artificial intelligence (AI) or AI-assisted technologies were used.
Data acquisition
Clinical, angiographic, and intravascular ultrasound (IVUS) imaging data were collected retrospectively. Offline quantitative coronary analysis (QCA) was performed using the Mac-Lab system (GE). Measurements included minimum lumen diameter (MLD) pre- and post-IVL. IVUS analysis was also performed offline using the AVVIGO + Multi-Modality Guidance System (Boston Scientific). Pre- and post-IVL pullbacks were matched by identifying branches, vessel ostium, and stent borders to obtain the minimum cross-sectional stent area (MSA).
Endpoints definition
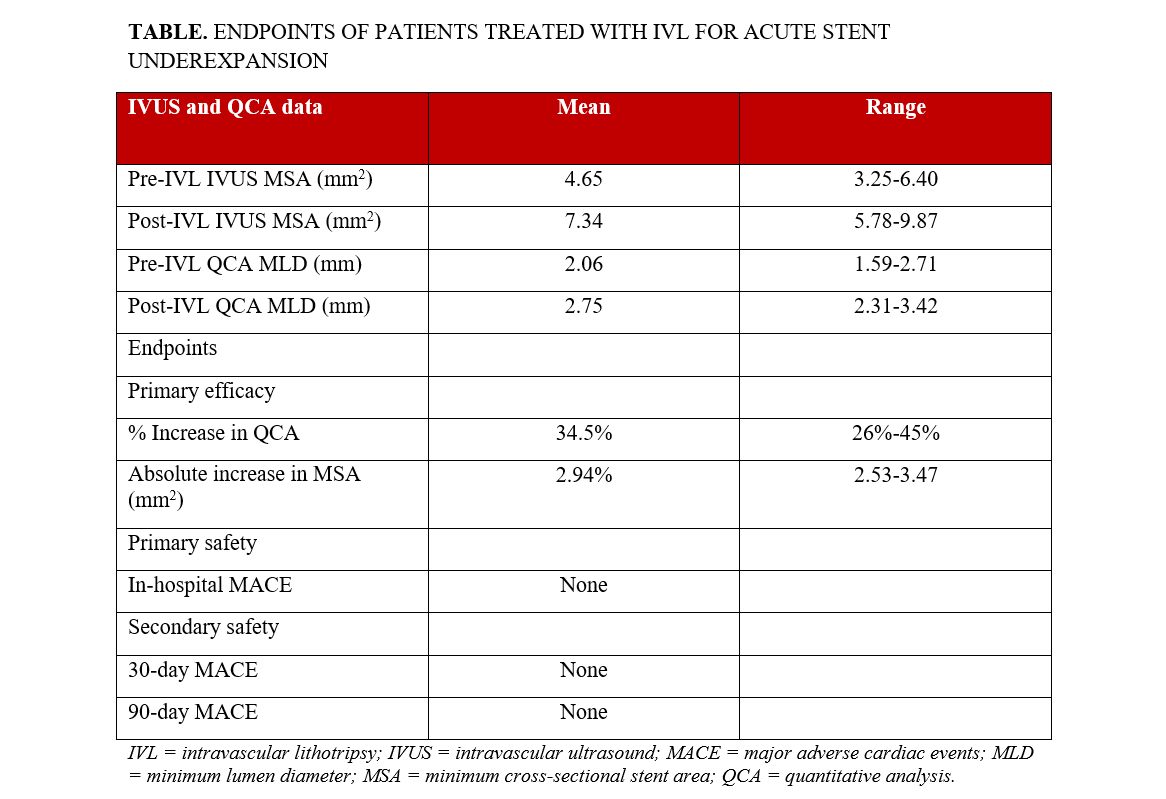
The primary safety endpoint was in-hospital major adverse cardiac events (MACE), and secondary safety endpoints were 30- and 90-day MACE, all defined as a composite of cardiac death, non-fatal myocardial infarction, or target lesion revascularization (TLR). The primary efficacy endpoint was device success as defined by an increase in QCA MLD by 20% and/or an increase in IVUS MSA by 2 mm2.
Follow-up
All patients were followed up until discharge. Clinical events were collected retrospectively from the electronic medical record at 30 and 90 days.
Results
One hundred twenty-seven patients underwent IVL during the study period. Four patients underwent IVL for acute stent underexpansion and were included in this analysis. The mean age was 67.7 +/-9.7 years, and all were male. Two presented with acute coronary syndrome. Three underwent atherectomy for balloon-uncrossable lesions prior to stenting. Two underwent IVL prior to stenting. All patients were treated with an appropriately sized noncompliant (NC) balloon pre- and post-stenting. The mean post-dilation inflation pressure was 16 mm Hg (range, 14-20 mm Hg). Three patients underwent IVL immediately post-stent implantation. One patient underwent IVL 2 days later in addition to OPN NC balloon (Sis Medical AG) angioplasty. The mean number of IVL pulses was 60 (range, 40-120 pulses). IVUS was performed post-stenting in all patients. Final IVUS post-IVL could not be obtained in 1 patient because of technical reasons. Device success was achieved in all patients after IVL. No IVL-related procedural complications or MACE were observed while in-hospital or at 30 and 90 days post-procedure. A representative case is shown along with QCA, IVUS data and endpoints (Figure; Table).
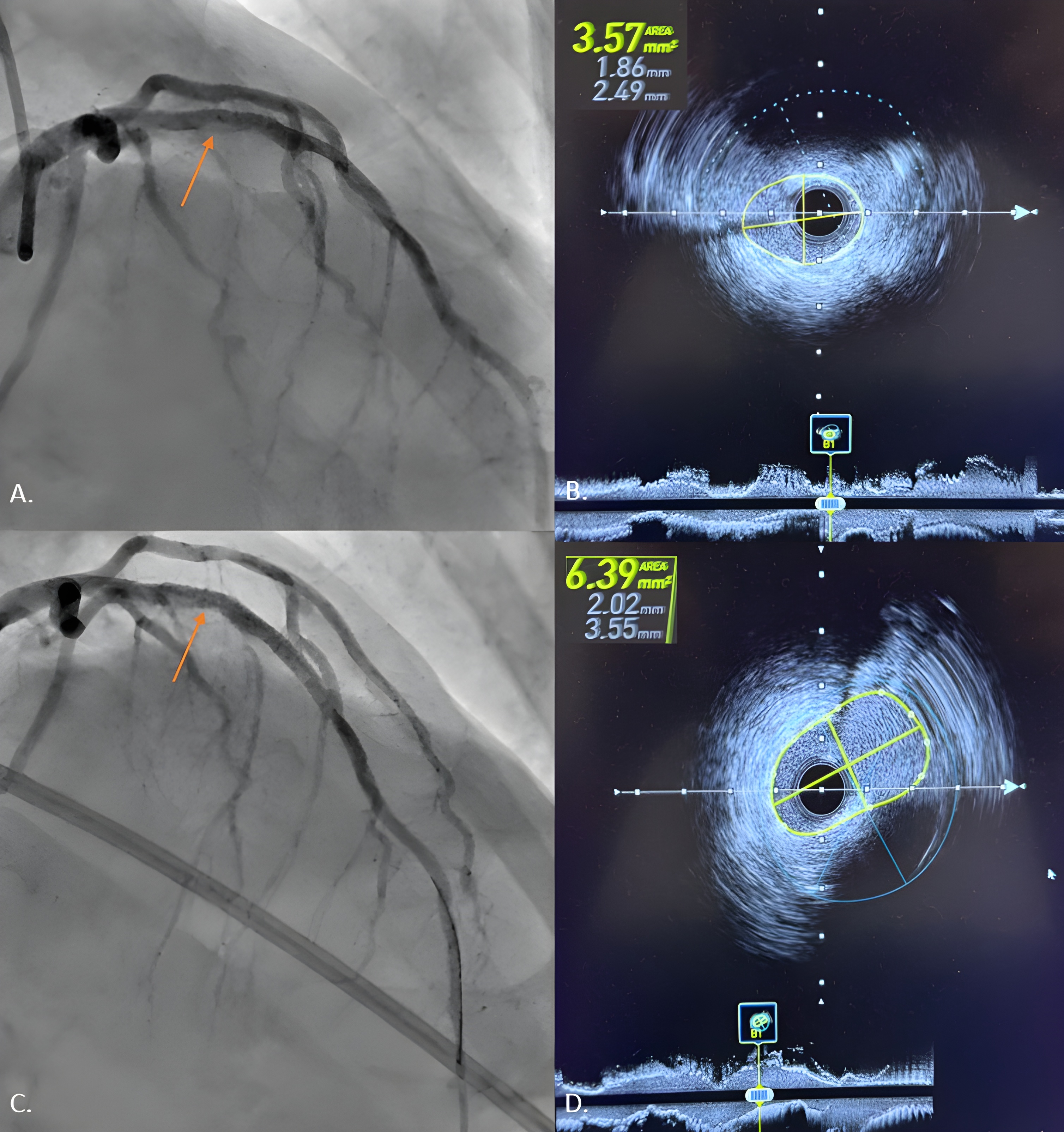
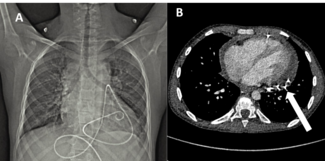
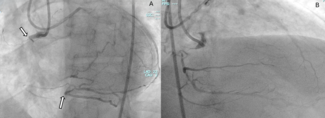
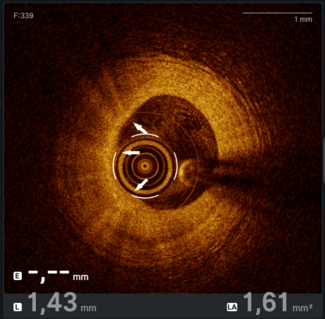
Figure. (A) Mid-left anterior descending artery acute underexpansion with (B) corresponding intravascular ultrasound (IVUS) minimum cross-sectional stent area (MSA) of 3.57 mm2. (C) Post-intravascular lithotripsy angiography showed improvement in stent expansion with (D) corresponding IVUS MSA of 6.39 mm2.
Discussion
Achieving optimal stent expansion is a central tenet of coronary stent implantation. Heavily calcified lesions often necessitate plaque modification, typically with intravascular lithotripsy (IVL) or atherectomy, to facilitate adequate expansion. Nevertheless, acute stent underexpansion can still occur in clinical practice, often requiring additional intervention. Several registries and meta-analyses have been published for off-label use in in-stent restenosis and acute stent underexpansion.3,4 While some therapies, such as contrast laser atherectomy, may pose a risk of damaging the drug-eluting stent polymer, IVL may not be associated with such risks.5 Our case series highlights the potential safety and favorable complication rates of IVL in the management of acute stent underexpansion.
Conclusions
Our case series indicates that bailout IVL for acute stent underexpansion is a safe and effective approach through 90 days of follow-up. Larger prospective studies are warranted to validate these findings.
Affiliations and Disclosures
Pooja Mishra, MD; Jonathan Rosenberg, MD; Verghese Mathew, MD; Justin Levisay, MD; Mark J. Ricciardi, MD; Arman Qamar, MD, MPH
From the Endeavor Center for Cardiovascular Intervention Outcomes Research and Evaluation (ECCORE) at Endeavor Health (NorthShore) Cardiovascular Institute, Glenview, Illinois.
The abstract has been presented as a poster at the 2025 Society for Cardiovascular Angiography & Interventions (SCAI) Scientific Sessions and published in JSCAI: https://doi.org/10.1016/j.jscai.2025.103219
Disclosures: Dr Qamar has received a research grant from Novartis, Novo Nordisk, Idorsia Pharmaceuticals, Siemens, Apple, Imperative Care, and Penumbra; and has received personal fees from Penumbra and Chiesi Pharmaceuticals. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Artificial intelligence (AI) statement: During the preparation of this research letter the author(s) did not use generative AI and AI-assisted technologies in the writing process.
Address for correspondence: Arman Qamar, MD, MPH, 2100 Pfingsten Rd, Glenbrook Hospital, Endeavor Health Cardiovascular Institute, Glenview, IL 60026, USA. Email: AQamar@northshore.org, aqamar@alumni.harvard.edu; X: @AqamarMD
References
1. Ng P, Maehara A, Kirtane AJ, et al. Management of coronary stent underexpansion. J Am Coll Cardiol. 2025;85(6):625-644. doi:10.1016/j.jacc.2024.12.009
2. Hill JM, Kereiakes DJ, Shlofmitz RA, et al; Disrupt CAD III Investigators. Intravascular lithotripsy for treatment of severely calcified coronary artery disease. J Am Coll Cardiol. 2020;76(22):2635-2646. doi:10.1016/j.jacc.2020.09.603
3. Tovar Forero MN, Sardella G, Salvi N, et al. Coronary lithotripsy for the treatment of underexpanded stents: the international & multicentre CRUNCH registry. EuroIntervention. 2022;18(7):574-581. doi:10.4244/EIJ-D-21-00545
4. Caminiti R, Vetta G, Parlavecchio A, et al. A systematic review and meta-analysis including 354 patients from 13 studies of intravascular lithotripsy for the treatment of underexpanded coronary stents. Am J Cardiol. 2023;205:223-230. doi:10.1016/j.amjcard.2023.07.144
5. Rivero-Santana B, Galán C, Pérez-Martínez C, et al. ELLIS study: comparative analysis of excimer laser coronary angioplasty and intravascular lithotripsy on drug-eluting stent as assessed by scanning electron microscopy. Circ Cardiovasc Interv. 2024;17(11):e014505. doi:10.1161/CIRCINTERVENTIONS.124.014505