Ventricular Septal Rupture After Anterior Wall Myocardial Infarction
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
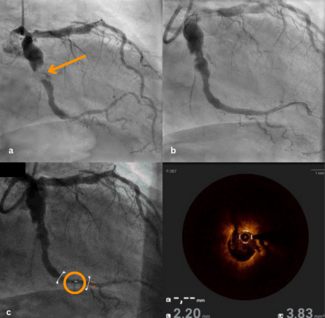
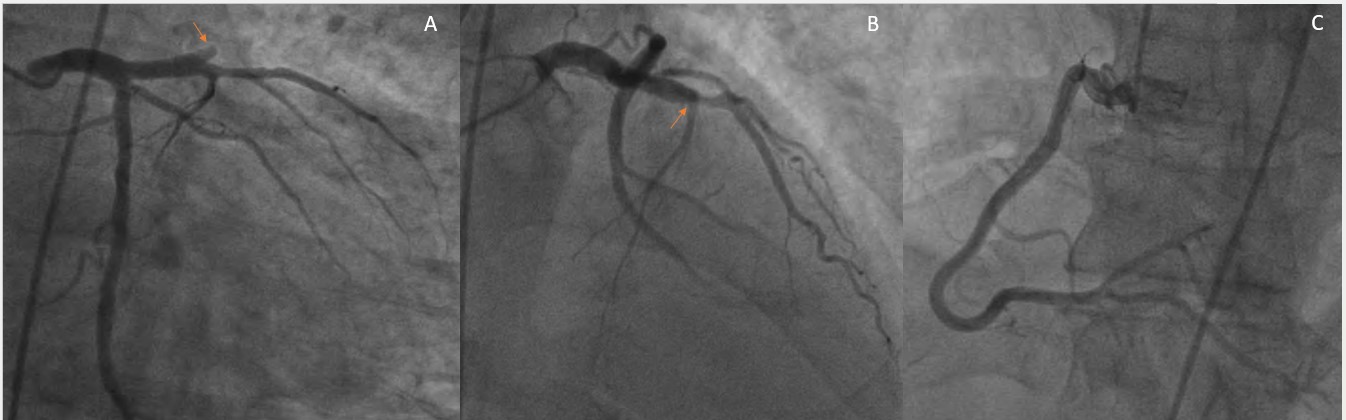
A 52-year-old man with a history of smoking and diabetes presented to the emergency department with chest pain and shortness of breath for the past 2 days. At presentation, his blood pressure was 110/70 mm Hg, and his pulse was 110 beats per minute. On auscultation, fine bilateral basilar crepitations could be heard; no murmurs or added heart sounds were heard. Electrocardiography was suggestive of anterior wall myocardial infarction (MI), with ST elevation in leads V1 to V5 and reciprocal ST depression in the inferior leads. After loading with antiplatelets and a statin, he was taken for coronary angiography, which revealed complete occlusion of the mid-left anterior descending artery (LAD) (Figure 1).
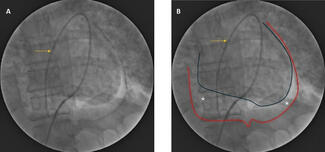
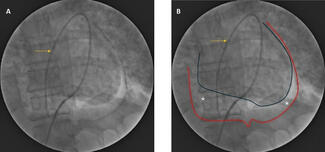
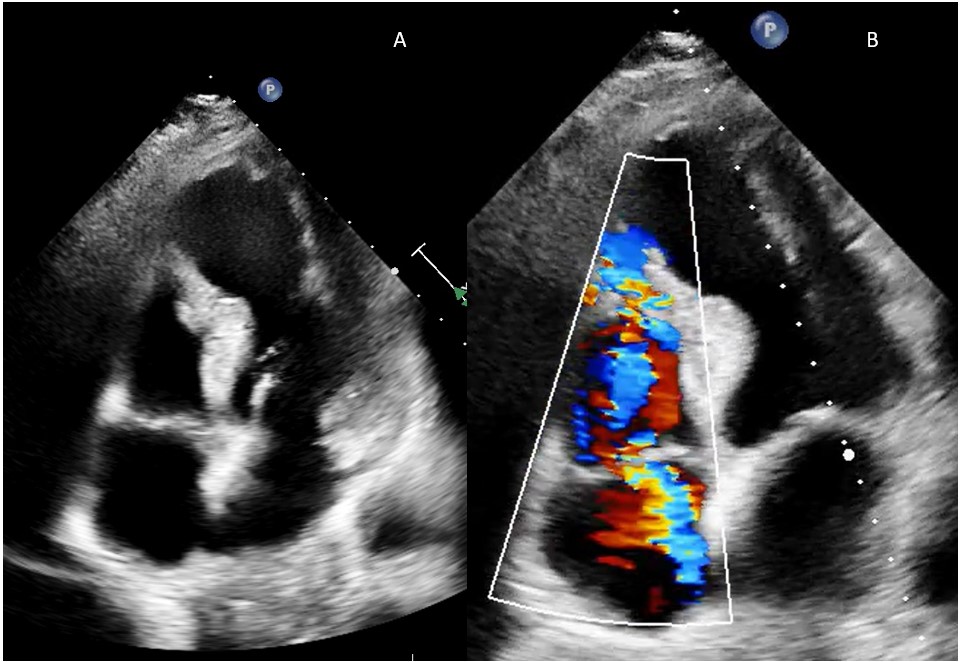
Shortly after completion of angiography, patient developed worsening shortness of breath and new onset hypotension in the catheterization laboratory. Auscultation revealed a new onset systolic murmur in the lower left sternal border and bilateral crepitations. There was no obvious cause to explain the hypotension—there was no radial site complication or new ischemic ST-T changes. Urgent echocardiography was performed, which revealed an aneurysmal and thinned-out apical territory, supplied by the LAD (Figure 2A). It also showed a left-to-right shunt at the apical region due to ventricular septal rupture (VSR) (Figure 2B; Videos 1 and 2). Tricuspid regurgitation was also noted.
The incidence of VSR after MI is about 0.3%.1 It is more frequent in the apical region because of anterior MI when compared with inferobasal VSR, which is due to inferior MI. The presentation of VSR is bimodal; it occurs either within the first 24 hours of MI or between 3 to 5 days afterwards. The characteristic clinical findings include hypotension, worsening chest pain and shortness of breath, and holosystolic murmur along left sternal border.2 The mortality remains 50% despite advances in treatment.1 Other common mechanical complications after MI include acute mitral regurgitation, which is more common in inferior MI because of complete blood supply of posteromedial papillary muscle by the right coronary artery. Free wall rupture, which occurs rarely, is catastrophic and associated with fast-accumulating pericardial effusion.
The patient was stabilized with an intra-aortic balloon pump and underwent successful surgical VSR repair combined with left internal mammary artery-to-LAD bypass. He was doing well at a 4-month follow-up.
This report emphasizes the importance of echocardiography in diagnosing mechanical complications after MI. In combination with physical examination findings, echocardiography can help differentiate among the different mechanical complications. Prompt diagnosis, stabilization with mechanical circulatory support, and surgical repair improve the prognosis and outcomes in these patients.
Affiliations and Disclosures
Mohan Prasad Akkineni, MD1; Krishna Prasad Akkineni, MD, DM2; Maithili Charan Gattu, MD3; Devesh Kumar, MD, DM4; Avishkar Agrawal, MD, DM5
From the 1Department of Cardiology, Postgraduate Institute of Medical Education and Research, Chandigarh, India; 2Department of Cardiology, Apollo Hospitals, Jubilee Hills, Hyderabad, India; 3Department of Emergency Medicine, All India Institute of Medical Sciences, Delhi, India; 4Department of Cardiology, Safdarjung Hospital, New Delhi, India; 5Department of Cardiology, All India Institute of Medical Sciences, New Delhi, India.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Consent statement: The authors confirm that informed consent was obtained from the patient for the intervention described in the manuscript and for the publication thereof, including photographs.
Address for correspondence: Krishna Prasad Akkineni, Room number 1, Ground floor, Apollo Hospitals, Jubilee Hills, Hyderabad 500033, India. Email: ramuchinnu92@gmail.com; Instagram: @krishnaakkineni
References
- Damluji AA, van Diepen S, Katz JN, et al; American Heart Association Council on Clinical Cardiology; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Surgery and Anesthesia; and Council on Cardiovascular and Stroke Nursing. Mechanical complications of acute myocardial infarction: a scientific statement from the American Heart Association. Circulation. 2021;144(2):e16-e35. doi:10.1161/CIR.0000000000000985.
- Birnbaum Y, Fishbein MC, Blanche C, Siegel RJ. Ventricular septal rupture after acute myocardial infarction. N Engl J Med. 2002;347(18):1426-32. doi:10.1056/NEJMra020228