Percutaneous Closure of a Giant Coronary Artery Aneurysm: A Case Summary
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
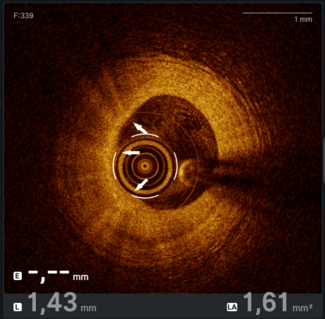
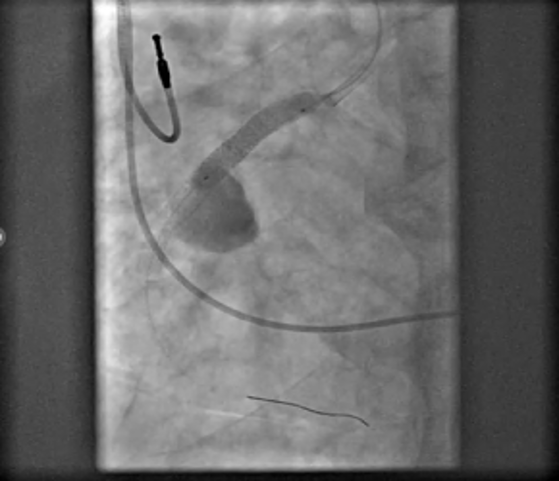
Coronary artery aneurysms (CAAs) are rare anomalies defined as coronary dilations greater than 1.5 times the normal diameter. Giant CAAs are even less common and often pose significant management challenges. We present the successful percutaneous exclusion of a giant right coronary artery (RCA) aneurysm in an older, high-risk patient (Figure 1).
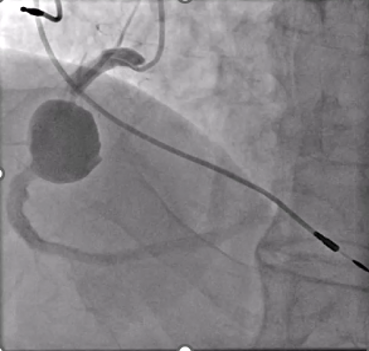
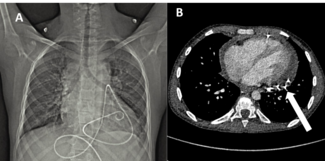
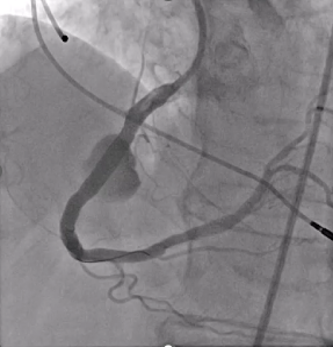
An 83-year-old man with hypertension, dyslipidemia, and permanent atrial fibrillation presented with New York Heart Association (NYHA) Class III symptoms. He had a prior RCA aneurysm identified in 2009, along with ectatic coronary vessels and a dual-chamber pacemaker for bradyarrhythmia (Figure 2). The aneurysm may have caused heart failure symptoms because of reduced coronary flow in the RCA. Another possible explanation was that the gigantic aneurysm caused a mild compression of the right atrium and a tricuspid valve regurgitation.
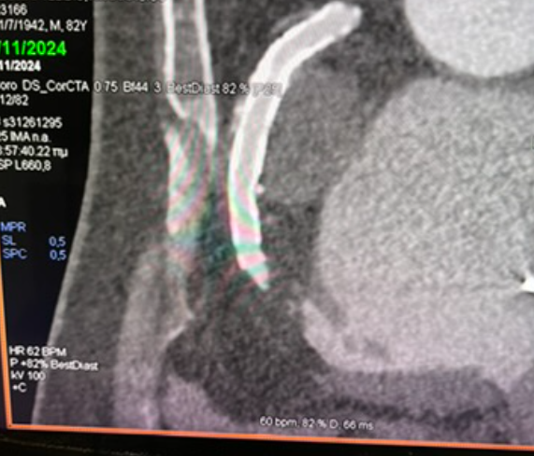
Computed tomography (CT) imaging demonstrated mild compression of the right atrium (Figure 3). Transesophageal echocardiogram (Video 1) confirmed tricuspid regurgitation, which was significantly reduced after closure of the aneurysm (Video 2). No ischemia test was performed, as the aneurysm had shown a considerable increase compared with the previous left heart catheterization, suggesting progressive expansion; therefore, it was considered necessary to proceed with closure without delay.
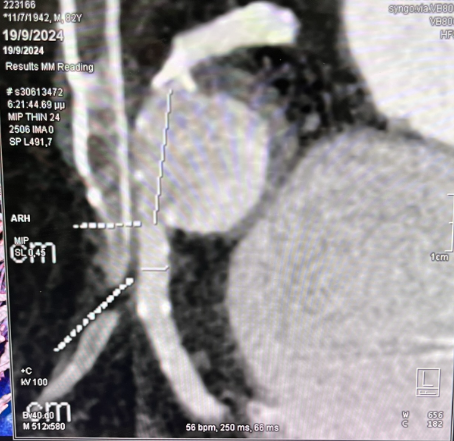
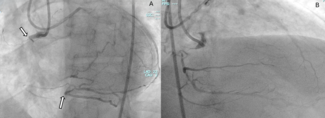
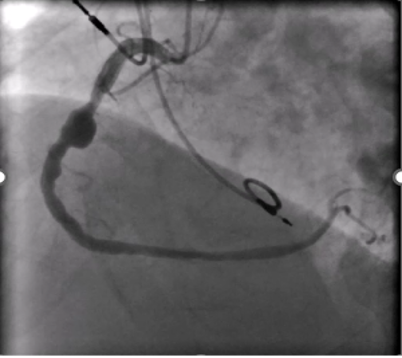
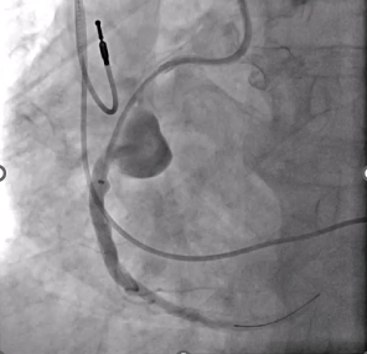
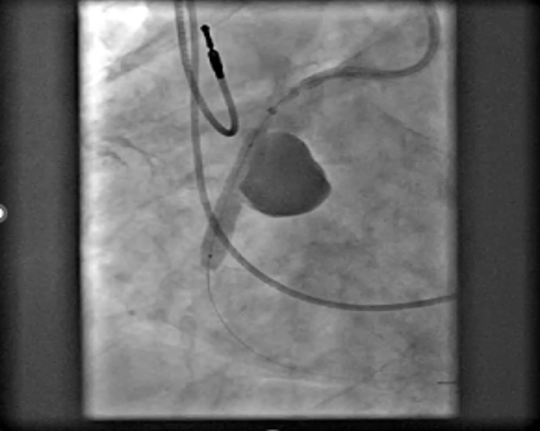
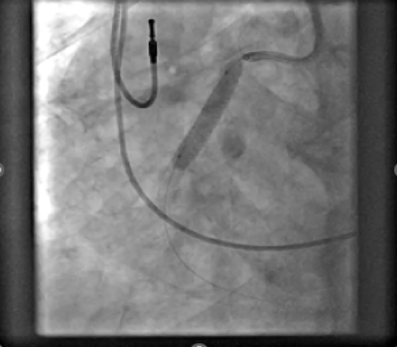
Recent imaging showed a significant progression of the RCA aneurysm (2.9 cm), with thrombus and mild compression of the right atrium (Figure 3). Given his surgical risk, a percutaneous approach was selected. Via femoral access, a 7F Amplatz Left 1 guide catheter was used for RCA cannulation. After crossing the lesion with a Sion Blue wire (Asahi) and exchanging for an IRON MAN wire (Abbott) using a microcatheter, a 7F guide-extension catheter facilitated deep vessel engagement and accurate aneurysm mapping (Figure 4). Three PK Papyrus covered stents (Biotronik) were sequentially deployed as follows:
- Distal to the aneurysm (4.5 × 26 mm)
- Proximal to the aneurysm (5.0 × 26 mm)
- Proximal sealing stent to fully exclude the sac (5.0 × 26 mm)
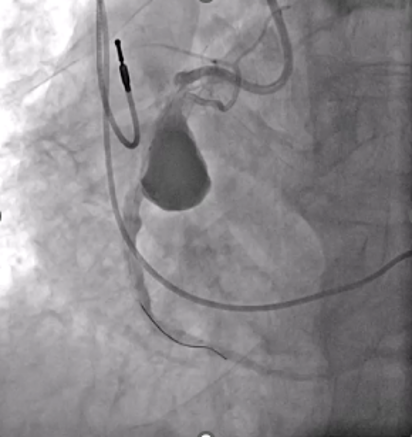
Postdilation with a 5.0 × 12-mm noncompliant balloon ensured full stent apposition (Figures 5-8). Final angiography confirmed complete exclusion of the aneurysm with no contrast leak (Figure 9). At the 1-month follow-up, CT imaging (Figure 10) showed total thrombosis of the aneurysm with no residual flow. The patient reported clinical improvement (NYHA Class I-II) and remained under evaluation for 6 months for tricuspid valve intervention.
This case supports the feasibility and effectiveness of percutaneous aneurysm closure in selected high-risk patients using new generation covered stents. Precise imaging, wire support, and thoughtful technique are key for successful outcomes.
Affiliations and Disclosures
Konstantinos A. Manousopoulos, MD, PhD1; Panagiotis Varelas, MD1; Petros Dardas, MD2; Nikolaos Mezilis, MD2; Ioannis Tsiafoutis, MD, PhD1
From the 1Hemodynamic Laboratory, General Hospital GHA Korgialeneio Mpenakeio-Hellenic Red Cross, Athens, Greece; 2Hemodynamic Laboratory, St Luke’s Hospital, Thessaloniki, Greece.
Disclosures: The authors report no financial relationships or conflicts of interest regarding the content herein.
Consent statement: The authors confirm that informed consent was obtained from the patient(s) for the intervention(s) described in the manuscript and for the publication thereof, including any and all images.
Address for correspondence: Konstantinos A. Manousopoulos, MD, PhD, Hemodynamic Department, General Hospital GHA Korgialeneio Mpenakeio-Hellenic Red Cross, Athanasaki 2 Street, Athens 11526, Greece. Email: konman7777777@gmail.com