Multipurpose Radiofrequency Wire System Improves Procedural Workflow Compared With Mechanical Needle for Left Atrial Appendage Closure
© 2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.
Abstract
Percutaneous left atrial appendage closure (LAAC) is a rapidly emerging technology with numerous devices now CE marked. This procedure is currently performed worldwide as an alternative to anticoagulation for atrial fibrillation patients who are not candidates for long-term anticoagulation. Transseptal puncture is a necessary part of LAAC. The VersaCross Connect (VCC) system (Boston Scientific) is a novel technology that includes the VersaCross radiofrequency wire for transseptal puncture and a shapeable dilator that fits within the WATCHMAN delivery sheath (Boston Scientific). The authors sought to compare procedural time and adverse events between the use of mechanical Brockenbrough needle (Medtronic) vs VCC for WATCHMAN implantation.
Introduction
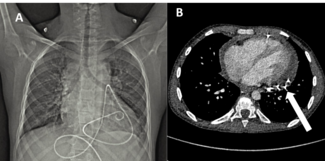
Stroke prevention with oral anticoagulants is one of the main pillars in atrial fibrillation (AF) therapy. However, as a significant proportion of patients have contraindications to their usage, left atrial appendage closure (LAAC) is an established alternative in patients with nonvalvular AF.1 Nonetheless, the procedural workflow for closure device delivery to the left atrial appendage (LAA) is rather complex, including several guidewire and sheath exchanges. Safe puncture of the interatrial septum is critical, and most centers use mechanical puncture with the Brockenbrough (BRK) needle (Medtronic) as their standard of care. There is a novel multipurpose radiofrequency wire system that includes a radiofrequency wire for transseptal puncture and a shapeable dilator that fits within the closure device delivery sheath. Consequently, transseptal puncture and closure device deployment can be performed within the same sheath, and both wire and dilator are removed after left atrial access is achieved (Figure A and B).
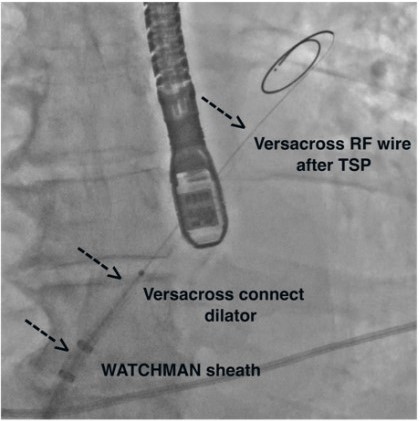
As this multipurpose radiofrequency wire system is highly promising for simplifying procedural steps,2 it has been compared to the BRK needle in previous trials. However, these trials were either conducted in AF ablation cohorts,3 early case series,2 or retrospective analyses.4 Therefore, we sought to compare procedural time and adverse events between the use of mechanical BRK needle vs the multipurpose radiofrequency wire system specifically for LAAC prospectively, with 360-day follow-up including cardiac computed tomography angiography (CTA) at 3 months.
Methods
All patients undergoing LAAC at Vancouver General Hospital were enrolled in our prospective observational registry. Institutional research ethics board approval was obtained, and all patients gave informed consent. We compared the procedural workflow and outcomes in 20 consecutive patients who underwent transseptal puncture with VersaCross Connect (VCC) (Boston Scientific), which was the multipurpose radiofrequency wire system used vs BRK mechanical needle for implantation of the WATCHMAN FLX device (Boston Scientific). Both transseptal systems are routinely used in our laboratory, and we sequentially alternated the use of the radiofrequency wire system with BRK needle. Patients underwent LAAC using the WATCHMAN FLX device under general anesthesia and transesophageal echocardiography guidance. We assessed transseptal puncture and device implant success, procedural times, and procedural complications. The total procedural time was defined as time from femoral access to femoral closure. During 360 days of follow-up, we assessed for major adverse events including stroke, transient ischemic attack, myocardial infarction, and both cardiovascular and non-cardiovascular death.
Results
These 20 cases were performed between June 2023 to January 2024. The patient median age was 74.5 years, 20.0% were women, and 90.0% had a previous major bleeding such as an intracranial hemorrhage or gastrointestinal bleed. Patients had either permanent (n = 11, 55.0%) or paroxysmal (n = 9, 45.0%) nonvalvular AF. Their median (interquartile range [IQR]) CHA₂DS₂-VASc and HASBLED Scores were 3.5 (3.0-5.0) and 3.0 (3.0-4.0), respectively. Most common comorbidities of this cohort included hypertension (n = 16, 80.0%), dyslipidemia (n = 10, 50.0%), and renal insufficiency (n = 9, 45.0%). Three patients (15.0%) had a patent foramen ovale and 5 patients (25.0%) had a lipomatous interatrial septum. All assessed baseline and anatomical characteristics of the interatrial septum were comparable between patients in the radiofrequency wire system and BRK needle (all P > .05) groups.
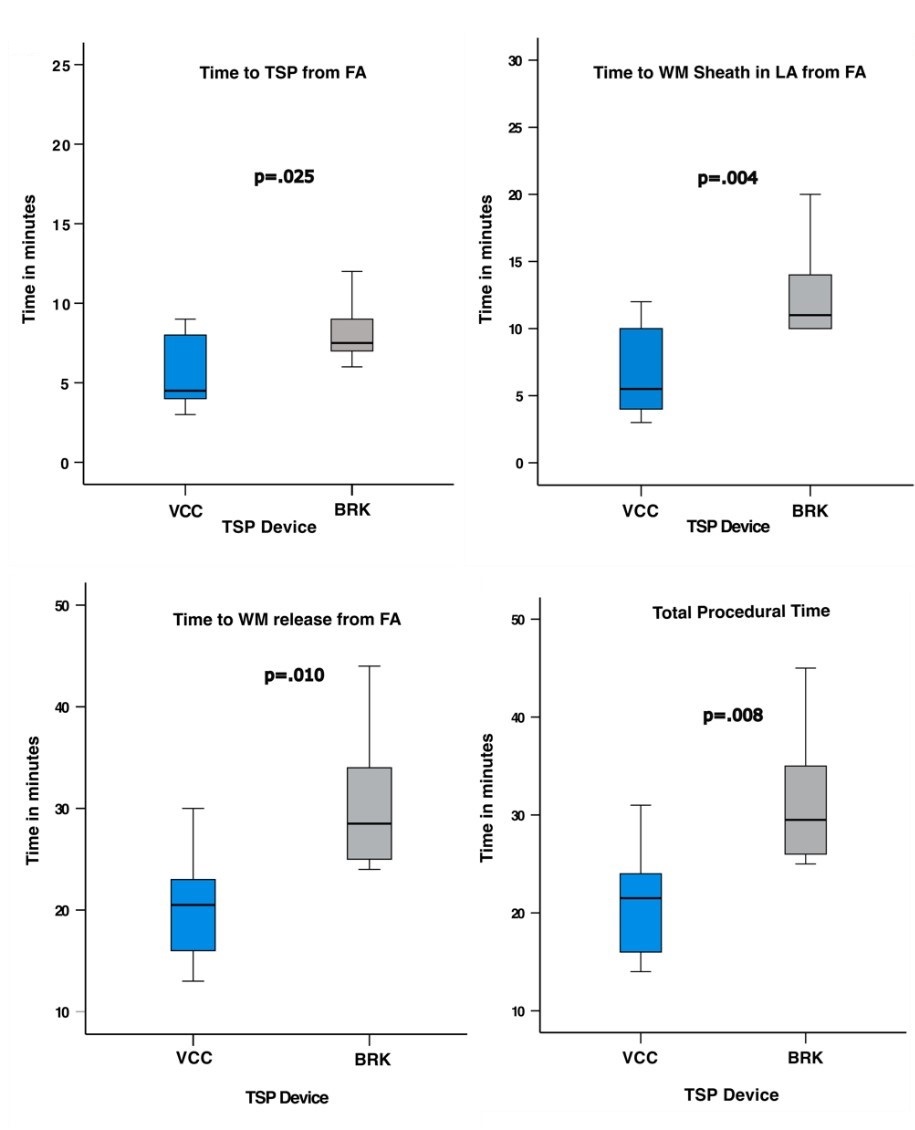
The transseptal puncture was successful in all 20 cases using the chosen access equipment. Time to transseptal puncture from femoral vein access was 40.0% faster in the radiofrequency wire system group compared with the BRK group (median [IQR] time in minutes: 4.5 [4.0, 7.5] vs 7.5 [7.0, 9.0], respectively; P = .025). Time to enter the closure device delivery sheath into the left atrium (median [IQR] time in minutes: 5.5 [4.2, 9.2] vs 11.0 [10.0, 13.5]; P = .004) and the time to closure device release from femoral access (median [IQR] time in minutes: 20.5 [16.2, 23.0] vs 28.5 [25.0, 33.5]; P = .010) were both significantly faster in the radiofrequency wire system group compared with the BRK group. The reduction in the total procedural time was 27.1% when using the radiofrequency wire system compared with the BRK (median [IQR] time in minutes: 21.5 [16.5, 23.8] vs 29.5 [26.0, 34.5]; P =.008) (Figure C). In addition, the total fluoroscopy time (median [IQR] time in minutes: 6.5 [5.1, 8.1] vs 9.3 [8.4, 11.0]; P = .034) and contrast dose (median [IQR] dose: 50.0 mL [42.0, 67.5] vs 72.5 mL [63.5, 85.2]; P = .019) were significantly lower in the radiofrequency wire system group. No procedural complication occurred, and all patients were discharged either same-day or 1-day post-procedure. During 360 days of follow-up, 1 ischemic stroke in the radiofrequency wire system group 3 months after the procedure was the only major adverse event, which occurred after ipsilateral carotid artery intervention. Cardiac CTA at 3 months after procedure showed no device thrombus or embolization in all cases.
Discussion
This prospective study showed significant improvement of procedural workflow when using the radiofrequency wire system vs the mechanical needle for LAAC, given a reduced total procedural time of about 30%. In addition, the total fluoroscopy time as well as contrast dose were significantly lower in the radiofrequency wire system group.
Several procedural factors can impact the safety of LAAC, including anatomical features, transseptal puncture location, and operator experience. Simplifying procedural steps by reducing exchanges of sheaths/equipment can shorten procedural duration and also have promising potential to reduce complications like air or thromboembolism.5 Notably, in contrast to other radiofrequency technologies (eg, Baylis), the radiofrequency wire system used in this study permits pressure monitoring and contrast injection only after the dilator tip has entered the left atrium. However, in the context of structural interventions, this has only limited practical relevance, as these procedures are performed under continuous imaging guidance.
In our series, the median procedural time for LAAC was only 21.5 minutes when using the radiofrequency wire system, with no associated periprocedural complications and a comparable outcome to BRK regarding adverse events during 360 days of follow-up, as well as cardiac CTA results.
Conclusions
The radiofrequency wire system appears to be a highly efficient alternative to the mechanical needle approach. Larger and randomized trials will be needed to further investigate the clinical impact on periprocedural complications and long-term outcome.
Affiliations and Disclosures
Desiree Wussler, MD1,2; Sophie Offen, MBBS, PhD1; Julius Jelisejevas, MD1; Hassan Ogran, MD1; Jacqueline Saw, MD1
From the 1Department of Cardiology, Vancouver General Hospital, University of British Columbia, Canada; 2Department of Cardiology and Cardiovascular Research Institute Basel (CRIB), University Hospital Basel, University of Basel, Switzerland.
Disclosures: Dr Wussler reports research grants from the Swiss National Science Foundation (Grant Reference P500PM_225285), the Swiss Heart Foundation (Grant Reference FF22112), the University Hospital Basel, and the German Heart Foundation (Grant Reference K22/13), as well as speaker honoraria from PHC outside the submitted work. Dr Saw has received unrestricted research grant support from the Canadian Institutes of Health Research, the Heart & Stroke Foundation of Canada, the National Institutes of Health, the University of British Columbia Division of Cardiology, AstraZeneca, Abbott, St Jude Medical, Boston Scientific, and Servier; salary support from the Michael Smith Foundation of Health Research; speaker honoraria from AstraZeneca, Abbott, Boston Scientific, and Sunovion; consultancy and advisory board honoraria from AstraZeneca, St Jude Medical, Abbott, Boston Scientific, Baylis, Gore, and FEops); and proctorship honoraria from Abbott, St Jude Medical, and Boston Scientific. The remaining authors report no financial relationships or conflicts of interest regarding the content herein.
Address for correspondence: Jacqueline Saw, MD, Department of Cardiology, Vancouver General Hospital, University of British Columbia, 2775 Laurel Street, 9th Floor, Vancouver, British Columbia V5Z 1M9, Canada. Email: jsaw@mail.ubc.ca
References
1. Saw J, Inohara T, Gilhofer T, et al. The Canadian WATCHMAN registry for percutaneous left atrial appendage closure. CJC Open. 2023;5(7):522-529. doi:10.1016/j.cjco.2023.03.016
2. Perrin N, McAlister C, Tsang M, Mondésert B, Ibrahim R, Saw J. Procedural simplification of left atrial appendage occlusion using the VersaCross connect system: first in-human experience. Catheter Cardiovasc Interv. 2023;101(1):227-230. doi:10.1002/ccd.30503
3. Dewland TA, Gerstenfeld EP, Moss JD, et al. Randomized comparison of a radiofrequency wire versus a radiofrequency needle system for transseptal puncture. JACC Clin Electrophysiol. 2023;9(5):611-619. doi:10.1016/j.jacep.2022.10.017
4. Asfour IK, Elchouemi M, Gianni C, et al. Novel streamlined technique for left atrial appendage closure using a radiofrequency wire-based large access system. J Cardiovasc Electrophysiol. 2023;34(8):1690-1697. doi:10.1111/jce.15995
5. Deneke T, Nentwich K, Schmitt R, et al. Exchanging catheters over a single transseptal sheath during left atrial ablation is associated with a higher risk for silent cerebral events. Indian Pacing Electrophysiol J. 2014;14(5):240-249. doi:10.1016/s0972-6292(16)30795-1