The Effect of Negative Pressure Wound Therapy on Abdominal Deep Incisional Surgical Site Infections in High-Risk Patients
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Surgical site infections (SSIs) significantly increase morbidity, mortality, and hospital stay, especially among high-risk patients with comorbidities such as diabetes or malignancy. Negative pressure wound therapy (NPWT) has improved outcomes in complex wounds, but its cost limits routine use in resource-constrained settings. Objective. To evaluate the effectiveness of NPWT on infection resolution, morbidity, and mortality in high-risk and emergency surgery patients with abdominal deep incisional SSI. Materials and Methods. High-risk patients (American Society of Anesthesiologistsphysical status class ≥III) who received NPWT for abdominal deep incisional SSI between February 1, 2015, and February 1, 2024, were analyzed. All cases met United States Centers for Disease Control and PreventionNational Healthcare Safety Network criteria for deep SSI. NPWT was applied following infection control and surgical debridement. Dressings were changed on postoperative day 3 and every 5 days thereafter. Mean patient follow-up was 1 year. Results. Sixty-five patients (36 females, 29 males; mean [standard deviation] age, 58.7 [13.4] years) were included. Emergency surgery was the initial surgical procedure in 56.9% of cases, and malignancy was the primary surgical indication in 50.8% of all 65 cases. Diabetes was the most common comorbidity (47.7%). The overall mortality rate was 4.6%. No SSI recurrence was observed during mean 1-year follow-up. Conclusion. NPWT is a safe and effective therapy for managing abdominal deep incisional SSIs in high-risk patients. In low-resource settings, its selective use in high-risk populations may optimize outcomes and cost-effectiveness.
Surgical site infections (SSIs) remain among the most frequent and challenging postoperative complications, leading to significant increases in morbidity, mortality, and hospital length of stay—particularly in patients with diabetes, malignancy, or multiple comorbidities.1 The development of SSI is multifactorial and is influenced by a combination of patient-related factors (eg, malnutrition, diabetes, immunosuppression, malignancy), surgical variables (including emergency procedures, prolonged surgical duration, and suboptimal aseptic technique), and hospital conditions (eg, inadequate ventilation, inappropriate antimicrobial prophylaxis).1,2
The incidence of SSI is especially high after abdominal surgery, with a rate of up to 8.3% in the general population and 25% to 30% among high-risk groups.3,4 Compared with patients without infection, those who develop an SSI have more than twice the mortality rate, a 60% higher likelihood of intensive care unit (ICU) admission, and a 5-fold increased risk of readmission.5 SSIs develop when the fragile balance between host immune defense and microbial burden shifts in favor of the pathogen. The most common sources of these pathogens are the patient’s own skin, mucous membranes, and endogenous intestinal flora. Multicenter surveillance and cohort data in Turkey have also highlighted a substantial SSI burden, supporting the need to contextualize advanced wound management strategies within the local health care setting.6,7
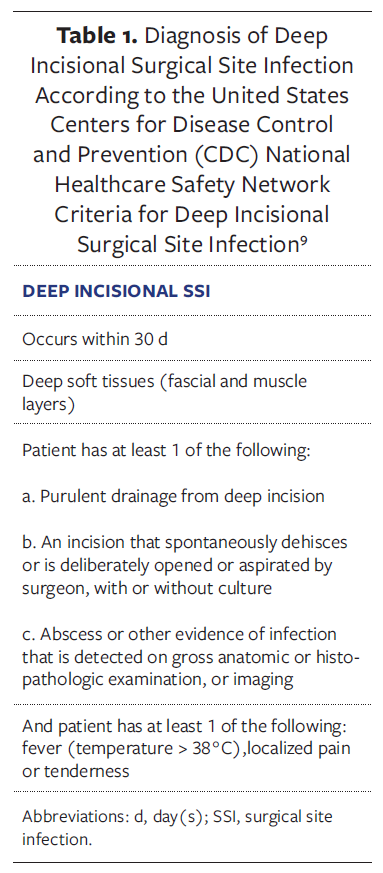
According to United States Centers for Disease Control and Prevention (CDC) National Healthcare Safety Network (NHSN) criteria, SSIs are categorized as superficial incisional, deep incisional, or organ/space infections.8 Dehiscence, defined as a mechanical separation of wound edges, is not classified as an SSI unless it is accompanied by clinical or microbiological signs of infection, such as purulent discharge, positive culture, fever with localized tenderness, or radiologic/histopathologic evidence. While superficial SSIs are usually managed with appropriate wound care and antibiotics, deep incisional infections, particularly in the abdominal region, often require surgical debridement and advanced wound management.9 In the absence of implants, deep SSIs typically manifest within 30 days postoperatively, most often around day 10, whereas in procedures involving implants, infection can appear months later, affecting deeper layers such as fascia and muscle.8 Experimental and clinical studies have demonstrated that negative pressure wound therapy (NPWT) can reduce bacterial load, control tissue edema, and stimulate granulation tissue formation by improving local perfusion and promoting mechanically induced cellular proliferation.10,11 NPWT has reshaped modern wound management, providing a major advance in the treatment of complex, hard-to-heal wounds.
Increasing evidence supports the role of NPWT in the management of complex or infected/dehisced abdominal wounds and, in selected settings, as an adjunct to reduce wound complications in high-risk or contaminated procedures; however, evidence derived from prophylactic incisional NPWT should not be directly extrapolated to the treatment of established deep incisional SSIs.12-15
NPWT is a costly intervention, however. Expenditures vary depending on device type, duration of use, and required consumables. In resource-limited health care settings, this cost may restrict widespread adoption, underscoring the need to apply NPWT selectively in patients who are most likely to benefit from its use.
Previous studies have identified the American Society of Anesthesiologists (ASA) Physical Status Classification Systemas a strong predictor of SSI development, with markedly higher infection rates among patients with comorbidities undergoing major general surgery compared with healthier individuals.16,17
The present retrospective study aimed to evaluate the effectof NPWT on infection resolution, morbidity, and mortality in high-risk or comorbid patients and in emergency cases in which deep abdominal incisional SSI developed following surgery.
Materials and Methods
Ethical standards
This clinical study was approved by the Institutional Review Board of Prof Dr Cemil Taşcıoğlu City Hospital, Istanbul, Turkey(Approval No.05.02.2019/1122) and was conducted in accordance with the principles outlined in the Declaration of Helsinki. All patients were evaluated by an experienced surgical team and received detailed verbal and written information about the procedure. Written informed consent was obtained prior to the initiation of treatment. Representative wound photographs were included with each patient’s permission and are presented solely for educational and illustrative purposes.
Study design
This retrospective study evaluated the efficacy of NPWT in treating abdominal deep incisional SSIs in high-risk patients and examined its effect on mortality and morbidity. The analysis was based on a prospectively maintained clinical database. Patients who underwent NPWT for abdominal deep incisional SSI between February 1, 2015, and February 1, 2024, at the Department of General Surgery at the University of Health Sciences, Istanbul, Turkey, were included.
The diagnosis of deep incisional SSI was made according to the CDC and NHSN definitions (Table 1).8 High-risk patients were identified by the presence of major comorbidities, malignancy, emergency surgery, or high-energy trauma. Only patients with an ASA physical status greater than or equal to III were eligible for inclusion. Patients with allergy to dressing components, silver sensitivity, acrylic adhesive intolerance, necrotizing fasciitis, pregnancy, or age younger than 18 years were excluded, as were those who had previously received open wound packing or vacuum-assisted therapy at the index operation.
Case ascertainment according to CDCNHSN criteria
CDCNHSN definitions were strictly followed.8 A case was considered a deep incisional SSI when, within the surveillance period for the index operation, any of the following conditions were met: the presence of purulent drainage from the deep incision; spontaneous or surgically opened dehiscence associated with fever (>38°C), localized pain, or tenderness with a positive culture; evidence of infection detected during reoperation, histopathologic examination, or imaging; or when the attending surgeon or physician diagnosed deep incisional SSI based on clinical judgment.Cases showing wound separation without culture positivity were classified as mechanical dehiscence. Using these definitions, deep incisional infections were carefully differentiated from superficial and organ/space SSIs, and only abdominal deep incisional SSIs were included in the main analysis.
Data collection
Comprehensive demographic and clinical data were collected, including comorbidities, surgical details, immunosuppressive status and HIV status, chemotherapy or corticosteroid use, diabetes, ASA classification, smoking history, body mass index, and laboratory parameters such as preoperative hemoglobin (Hb), albumin, fasting glucose, and HbA1c levels. Additional information regarding perioperative findings, hospital stay, morbidity, and mortality was also recorded.
In patients with poorly controlled diabetes (HbA1c >7%), postoperative blood glucose levels were closely monitored 4 times daily, and insulin adjustments were made according to institutional and guideline-based protocols to maintain glucose between 110 mg/dL and 180 mg/dL.
NPWT procedure
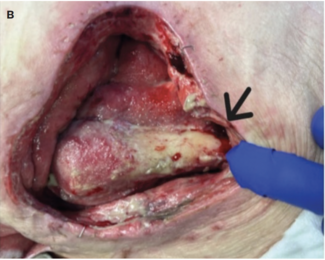
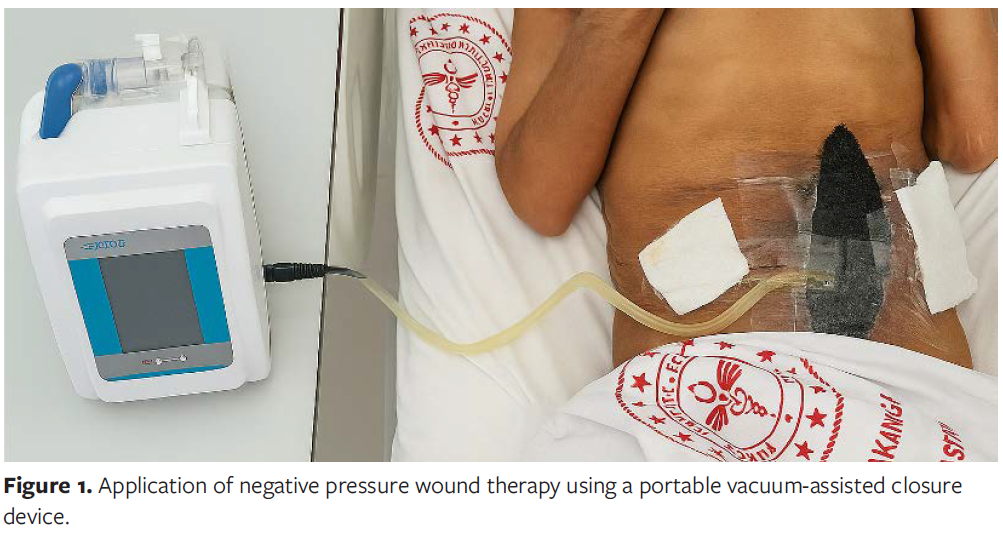
NPWT was applied following thorough surgical debridement and wound bed preparation. All wounds were irrigated with sterile saline until viable tissue margins were achieved. Therapy was initiated only after local infection control was established, in line with manufacturer recommendations and clinical wound care protocols. NPWT was not applied in cases of uncontrolled or spreading infection.The Comfort NPWT system (Konfort Özel Sağlık Hizmetleri Tıbbi Malz San ve Tic Ltd Şti) was used with continuous negative pressure of −75 mm Hg. The first dressing change was performed on postoperative day 3, followed by changes every 5 days. Figures 2A and 2B illustrate the application process in accordance with the institutional protocol.
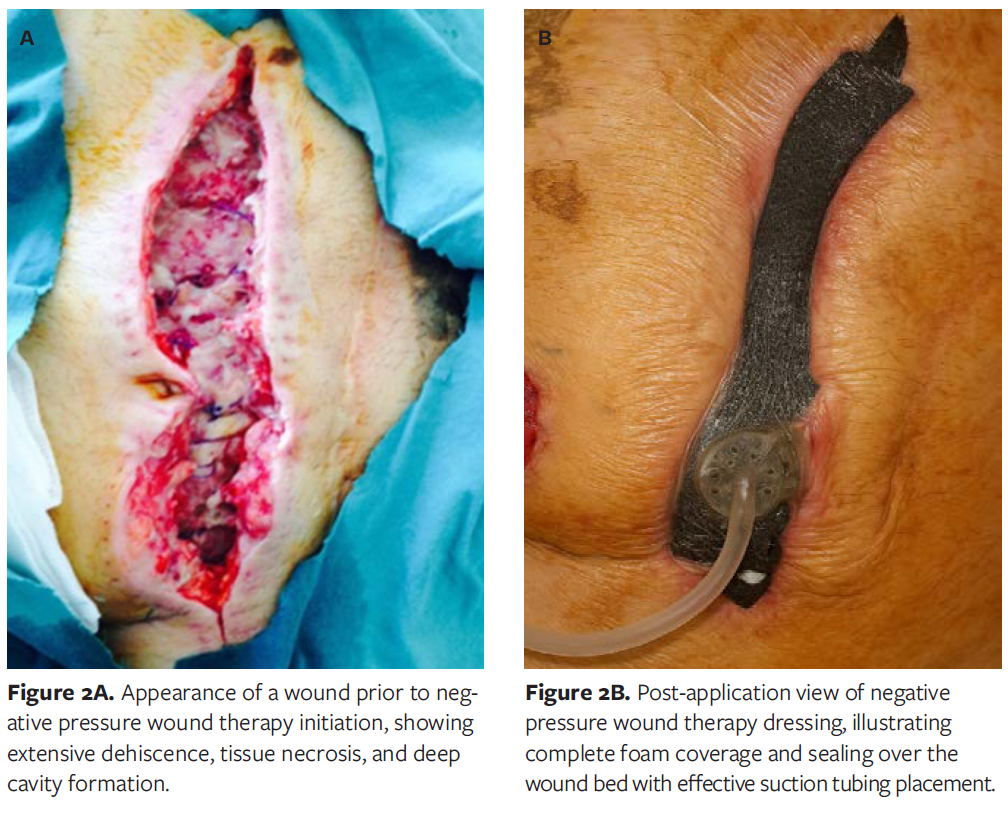
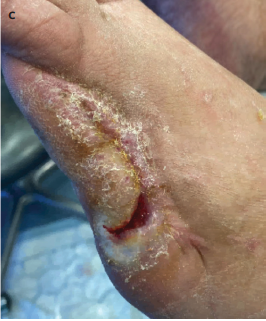
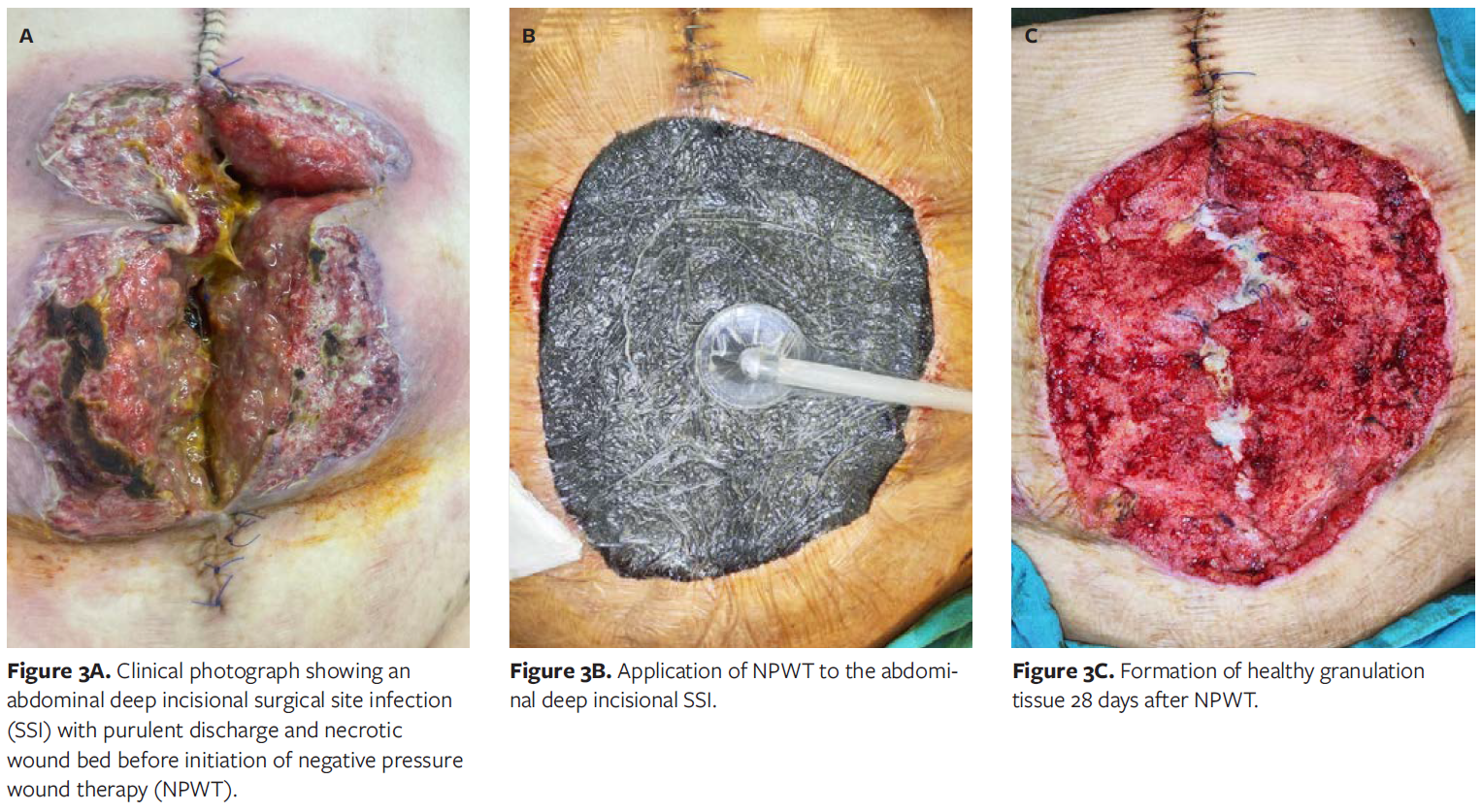
Throughout treatment, NPWT facilitated progressive wound contraction, reduced exudate and necrotic debris, and promoted the formation of healthy granulation tissue. No device-related complications such as bleeding, pain, or allergic reactions were observed. Representative pre- and posttreatment photographs demonstrate substantial improvement in wound bed appearance(Figures 3A-3C).
Microbiological evaluation
Wound cultures were obtained before initiating NPWT, and systemic antimicrobial therapy was tailored according to culture and sensitivity results, following the Infectious Diseases Society of America guidelines.18
Empirical broad-spectrum antibiotics were started immediately after sample collection and later modified based on susceptibility profiles. Treatment was continued during the initial NPWT phase until clinical improvement was evident, typically lasting 7 days to 10 days depending on wound severity and microbiological findings. Culture specimens were collected from the deepest part of the wound after debridement and saline cleansing using sterile swabs and then promptly transported to the microbiology laboratory for aerobic and anaerobic culture. Microbial identification and antibiotic susceptibility testing were performed using standard and automated techniques. Patients were reevaluated at the outpatient clinic on postoperative day 7 after discharge and then received telemedicine-based monitoring until complete recovery.
Statistical analysis
Data were analyzed using SPSS version 25.0 (IBM Corporation). Descriptive statistics were presented as numbers and percentages for categorical variables, and as mean (SD), minimum, and maximum values for continuous variables. Normally distributed continuous data were compared using the t test. Normality of continuous variables was assessed using the Shapiro-Wilk test. Differences in hospital stay were evaluated between patients with and without diabetes, as well as between those with and without malignancy. A P value less than .05 was considered statistically significant.
Results
A total of 65 patients were included in the study, with 36 females (55.4%) and 29 males (44.6%) with a mean (SD) age of 58.7 (13.4) years (range, 24 years-91 years). The initial surgical procedure was performed under emergency conditions in 37 patients (56.9%) and electively in 28 patients (43.1%). Malignancy was the primary surgical indication in 33 cases (50.8%).
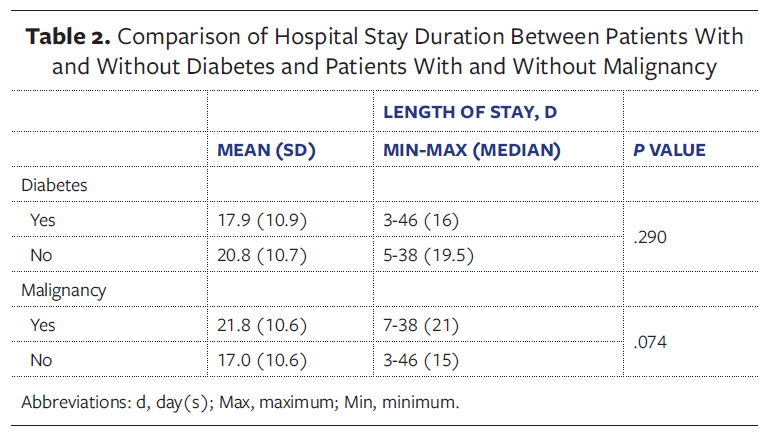
The overall mortality rate was 4.6% (n = 3). The mean hospital length of stay was 19.4 days (range, 8 days-46 days), which was relatively long because the study population consisted of high-risk patients with multiple comorbidities and malignancies. No statistically significant difference in hospital stay was observed between patients with and without diabetes or between those with and without malignancy (P > .05 for both comparisons) (Table 2). A notable finding was that 58 patients (89.2%) had undergone their primary operation at outside hospitals and were subsequently referred to the authors’ center for SSI management. The remaining 7 patients (10.8%) underwent emergency surgery at the authors’ institution, where NPWT could not be applied during the index procedure.
Surgical procedures and comorbidities
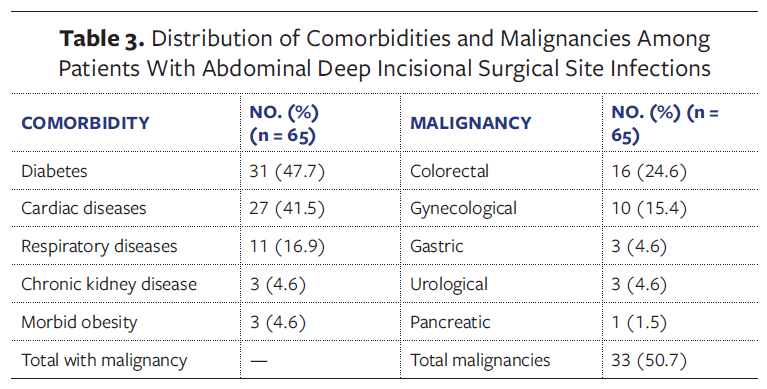
The most frequent surgical procedure was abdominal hernia repair (n = 22 [33.8%]), nearlyhalf of which were incisional hernias (n = 10). Diabetes was the most common comorbidity, affecting 31 patients (47.7%), with a mean (SD) preoperative HbA1c value of 8.1%(1.0%). Among malignancies, colorectal cancer was most prevalent (n = 16 [24.6%]). The most frequent oncologic operations were debulking procedures and colectomy/anterior resection, each performed in 8 patients (12.3%).
Comorbidity analysis revealed severe cardiac disease in 27 patients, significant respiratory disease in 11, chronic kidney disease requiring hemodialysis in 3, and morbid obesity in 3. The detailed distribution of comorbidities and malignancies is shown in Table 3. Six patients (9.2%) had metastatic cancer, 4 had multiple primary malignancies, and 1 had peritoneal carcinomatosis.
Microbiological findings
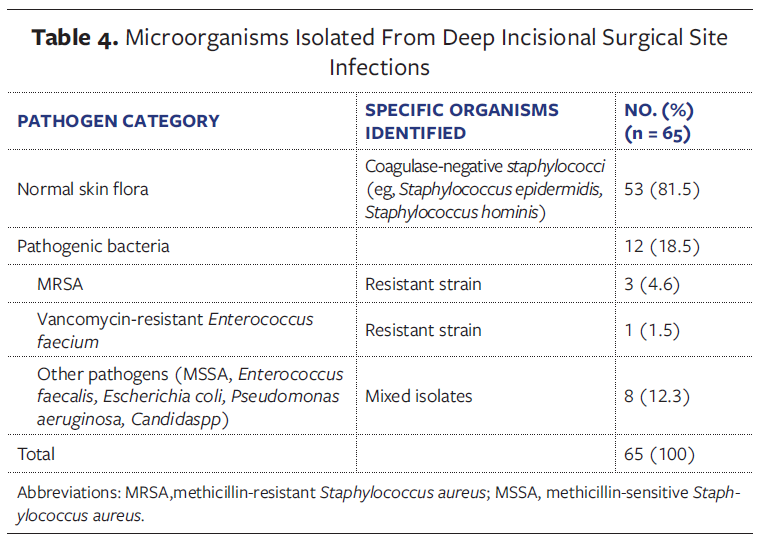
Wound cultures revealed normal skin flora in 53 patients (81.5%) and pathogenic microorganisms in 12 patients (18.5%). Methicillin-resistant Staphylococcus aureus (MRSA) was isolated in 3 cases and vancomycin-resistant enterococci(VRE) in 1. Normal flora consisted predominantly of coagulase-negative Staphylococcus species, mainly S epidermidis and S hominis. Although typically commensal, these organisms can act as opportunistic pathogens in high-risk surgical wounds, particularly when host defenses are compromised or wound healing is delayed.
Among pathogenic isolates, MRSA and VRE represented the most clinically relevant resistant strains. Additional isolates included methicillin-sensitive S aureus, Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa, and Candida species. Overall, gram-positive cocci predominated, suggesting that most infections originated from endogenous skin flora. Gram-negative and fungal isolates were less frequent and primarily occurred in polymicrobial infections. The detailed microbiological distribution is summarized in Table 4.
Mortality and follow-up
Of the 3 deaths, 2 occurred in patients with advanced, multimetastatic cancer, and 1 occurred in an 81-year-old patient with uncontrolled diabetes, ischemic heart disease, and hypertension. Patients were followed for an average of 1 year, during which no recurrence of SSI was observed. Two patients with metastatic malignancies—1 with metastatic colon cancer and 1 with metastatic cervical cancer—died 3 and 4 months after discharge, respectively.
Other comorbid conditions
Additional conditions, each observed in a single patient, included severe ulcerative colitis, gunshot wound, COVID-19, HIV infection, toxic hepatitis, liver cirrhosis, Behçet disease, and sarcoidosis.
Discussion
SSIs remain among the most frequent health care–associated infections, yet their global health effect continues to be underestimated. They contribute substantially to postoperative morbidity and mortality, prolonged hospital stays, increased ICU admission rates, and significant health care expenditures.19 The development of SSIs is multifactorial, influenced by both patient-related and procedural risk factors.20 Common predisposing factors include diabetes, obesity, malnutrition, advanced age, smoking, immunosuppressive therapy, and remote infections. The risk is particularly elevated in emergency surgeries, where contamination rates are inherently higher.21 Furthermore, prolonged surgical time, intraoperative contamination, and inadequate antibiotic prophylaxis also play major roles in infection pathogenesis.20 Therefore, interventions that target modifiable factors can help reduce postoperative morbidity and mortality in these vulnerable groups.
In addition to global reports, Turkey-
specific surveillance and multicenter data indicate that SSI rates in Turkey can be substantial and, for several procedure types, have been reported to be higher than CDCNHSN rates and comparable to international consortium estimates.6 Moreover, a multicenter cohort study from Turkey including abdominal procedures confirmed that SSI remains frequent and is strongly associated with emergency surgery and increased contamination risk.7
The World Health Organization (WHO) recommends considering prophylactic NPWT (pNPWT) as a preventive measure for SSIs in high-risk surgical patients, especially in those with poor tissue perfusion, soft tissue injury, hematoma, dead space, or intraoperative contamination.13 However, the WHO also notes that in
resource-limited settings, pNPWT may not always be prioritized over other essential infection prevention strategies due to cost and availability constraints.14
Importantly, the present study does not evaluate pNPWT; rather, it focuses on therapeutic, open wound NPWT applied to established deep abdominal incisional SSIs with wound dehiscence/complexity. This distinction is clinically important because evidence for prophylactic incisional NPWT cannot be directly extrapolated to the treatment of already infected, dehisced wounds. The present study focused on a high-risk surgical cohort with multiple comorbidities and malignancies, many of whom were predicted to have poor survival outcomes. In this context, this cohort reflects a real-world, high-risk surgical population commonly encountered in tertiary referral centers in Turkey, where emergency surgery, malignancy, and multiple comorbidities are prevalent. Despite these challenges, NPWT achieved a 95% clinical success rate, with only 3 deaths—2 in patients with advanced metastatic disease and 1 in an elderly patient with uncontrolled diabetes. Clinical success was defined as durable infection control without the need for reoperation, reintervention, or readmission due to recurrent SSI during the follow-up period. Notably, no recurrence of SSI was observed during 1-year follow-up, underscoring the durability and effectiveness of NPWT in this complex population.
Given the limited availability of robust Turkey-specific outcome datasets for therapeutic NPWT in established deep incisional SSI, the findings in the present study provide clinically relevant “real-
world” evidence from a high-risk cohort treated in Turkey.
SSIs remain a leading cause of postoperative morbidity in abdominal surgery despite adherence to modern preventive protocols. Diabetes is a well-established independent risk factor, particularly in high-risk patients. Poor glycemic control, reflected by elevated HbA1c levels, is known to impair wound healing and increase infection susceptibility.22,23 In the present study, although hospital stay and postoperative outcomes did not differ significantly between patients with diabetes and those without diabetes, elevated HbA1c levels appeared to be associated with an increased tendency toward SSI and delayed wound healing. These findings suggest that glycemic control status may be an important contributing factor to postoperative recovery in high-risk surgical patients. The microbiological results were consistent with recent studies on abdominal SSIs, with normal skin flora—predominantly coagulase-negative Staphylococcus species—representing the most common isolates. This finding suggests that most infections originated from endogenous rather than exogenous sources. Resistant organisms such as MRSA and VRE were rare but clinically significant, consistent with previous reports highlighting the low yet relevant prevalence of multidrug-resistant pathogens in gastrointestinal surgery.7 These results emphasize that effective SSI prevention depends not only on surgical technique but also on optimizing host factors, such as glycemic control, together with continuous microbiological surveillance and rational antibiotic use.
The findings of the present study also support previous large-scale studies that identified ASA physical status III or higher as a strong predictor of SSI.16,17 All patients in the cohort in the present study had ASA III or higher, confirming the close association between systemic disease burden and postoperative infection risk.
Acute abdominal wall hernias were the most frequent surgical indication in this study, accounting for one-third of all operations. Emergency hernia repairs often involve bowel obstruction, ischemia, or strangulation, conditions that inherently increase contamination and infection risk.24,25 These findings are in line with earlier reports identifying obesity, diabetes, and smoking as major contributors to SSI development in hernia surgery—factors that are rarely modifiable in emergency settings.
The integration of NPWT into multimodal SSI prevention bundles has been associated with significantly reduced infection rates in gastrointestinal surgery.26 Previous studies demonstrated that pNPWT can be especially beneficial and cost-effective in high-risk subgroups, including in patients aged 65 years or older, those receiving chronic corticosteroid therapy, or those with multiple comorbidities.26 In the present series, patients with colorectal malignancy or those on long-term immunosuppressive therapy (eg, patients with ulcerative colitis on corticosteroids) experienced favorable outcomes with NPWT despite prolonged hospitalization.
Because the intervention in the present study was therapeutic NPWT applied to established deep incisional SSI, the key clinical implication is not “routine prophylaxis” but rather “timely escalation to advanced wound therapy” in high-risk patients when deep SSI or wound breakdown occurs. Recent high-quality evidence also suggests that prophylactic incisional NPWT should not be used universally after emergency laparotomy, reinforcing the importance of selective, indication-driven use rather than routine application.15
Dressing change frequency remains an important practical consideration. Although manufacturers recommend changing dressings every 48 hours to 72 hours, clinical evidence supports extending this interval without compromising outcomes.27 In the institution of the authors of the present study, a 5-day dressing change protocol proved both effective and resource-efficient, reducing material and labor costs while maintaining clinical efficacy.
The effect of SSIs extends beyond wound healing alone. In oncology patients, infections can delay adjuvant chemotherapy, potentially affecting recurrence-free and overall survival.28 In the cohort in the present study, 55% of patients had malignancies, and successful SSI management enabled timely continuation of chemotherapy in all surviving patients, highlighting the importance of infection control in preserving continuity of oncologic care.
While NPWT has demonstrated clear benefits in SSI management, its prophylactic use remains controversial. Some studies have shown no significant reduction in SSI incidence or hospital stay with pNPWT following colorectal surgery.29 Given the economic constraints of health care systems, routine universal use of pNPWT may be premature until robust cost-effectiveness data become available.
In Turkey, available health economic estimates indicate that SSIs impose a substantial national cost burden, primarily driven by prolonged hospitalization, reinterventions, and increased antibiotic use, underscoring the importance of strategies that reduce downstream resource utilization.30 Accordingly, “selective use” should be guided by patient risk, wound status (infected/dehisced vs clean closed incision), and expected clinical benefit rather than routine adoption. Consistent with recent meta-analytic findings, a selective, indication-driven approach to NPWT appears more appropriate than routine application across all surgical settings.31
Beyond infection control, NPWT has been associated with accelerated wound healing, reduced pain, fewer dressing changes, and improved patient mobility.32 Earlier studies comparing NPWT with standard wound care (SWC) yielded inconsistent findings regarding quality of life (QoL). Some favored NPWT, while others reported higher anxiety levels associated with device use, thereby favoring SWC.33,34 Factors such as restricted mobility, device noise, and unfamiliarity with the technology may contribute to these discrepancies. However, comprehensive patient education and the use of portable NPWT systems have been shown to enhance comfort, adherence, and overall satisfaction.
In the institution of the authors of the present study, trained wound care specialists and structured patient counseling improved compliance and satisfaction, indicating that when appropriately applied, NPWT can foster not only physical recovery but also psychosocial well-being. Furthermore, because the present study exclusively targeted high-risk surgical patients, the benefits of NPWT were found to be more pronounced in this subgroup than in the general surgical population.
Recent research has also demonstrated that combining NPWT with tailored physiotherapy techniques can further enhance recovery in patients with posttraumatic or postoperative wounds by promoting active mobilization and social interaction during rehabilitation.35 Active rehabilitation accelerates physical healing, reinforces emotional reintegration, and improves overall QoL.35 Additionally, NPWT has been linked to lower rates of hospital readmission and reoperation compared with SWC, yielding both clinical and systemic advantages.36
Limitations
This study has several limitations. The most notable is the absence of an internal control group composed of patients with comparable clinical risk profiles who did not receive NPWT. This was an intentional methodological choice, guided by ethical concerns, because withholding NPWT from high-risk patients might have led to unacceptable increases in morbidity or mortality. Although this limits the ability to perform direct statistical comparisons, it represents a clinically and ethically justified decision within this high-risk population.
In addition, the retrospective nature of the study and the relatively small sample size may restrict the generalizability of the results. Nevertheless, all patients were consecutively included and managed using a standardized institutional protocol, thereby minimizing selection bias. Another limitation is the lack of a formal cost-
effectiveness analysis. This is particularly relevant given the high expense associated with NPWT devices and consumables. However, considering the substantial economic burden of SSIs reported in Turkey, the potential downstream cost implications of therapeutic NPWT, such as reduced complications, reinterventions, and prolonged hospitalization, remain clinically relevant.30 In this context, by extending the dressing change interval to approximately every 5 days, the authors’ institutional protocol effectively reduced overall material and labor costs without compromising treatment efficacy or patient outcomes.
Future prospective, multicenter trials with larger sample sizes and integrated economic analyses are warranted to confirm these findings, refine patient selection criteria, and establish standardized NPWT application protocols that balance clinical benefit with resource utilization. Such studies would be particularly valuable in generating Turkey-specific comparative outcome and health economic data for therapeutic NPWT in established deep incisional SSIs.
Conclusions
NPWT represents a safe, effective, and clinically valuable treatment modality for managing established abdominal deep incisional SSIs, particularly among high-risk patients such as those with malignancies or diabetes, or those undergoing emergency surgical procedures. In this challenging population, NPWT facilitated durable infection control, promoted wound healing, and was associated with low recurrence rates and acceptable mortality, even in patients with significant systemic disease burden.
Unlike prophylactic incisional NPWT applied to closed wounds, therapeutic open wound NPWT was shown in the present study to be beneficial in infected or dehisced abdominal incisions, addressing an important clinical scenario that is underrepresented in the current literature. This distinction is crucial, because evidence supporting routine prophylactic use cannot be directly extrapolated to the treatment of established infections.
Considering the substantial cost of NPWT systems, selective application in carefully identified high-risk patients who are most likely to benefit appears to be the most rational strategy, particularly in resource-limited health care settings such as Turkey. Broader implementation should be supported by robust cost-effectiveness evaluations and well-designed, prospective multicenter randomized controlled trials focusing on therapeutic NPWT, with the aim of optimizing treatment parameters, improving patient-centered outcomes, and strengthening evidence-based wound management practices across abdominal and general surgery.
Author and Public Information
Authors: Semih Hot, MD and Seracettin Eğin, MD
Affiliation: Department of General Surgery, Hamidiye Faculty of Medicine, University of Health Sciences, Istanbul, Turkey
Disclosure: The authors declare no financial or other conflicts of interest.
Ethical Approval: This study was approved by the Institutional Review Board of Prof Dr Cemil Taşcıoğlu City Hospital, Istanbul, Turkey (Approval No.05.02.2019/1122).
Correspondence: Semih Hot, MD; Selimiye, Atölyeler Cd. No. 7, 34668 Üsküdar/Istanbul, Turkey; semihhot@hotmail.com
Manuscript Accepted: January 5, 2026
References
- Coccolini F, Improta M, Cicuttin E, et al. Surgical site infection prevention and management in immunocompromised patients: a systematic review of the literature. World J Emerg Surg. 2021;16(1):33. doi:10.1186/s13017-021-00375-y
- Joseph N, Vishnoi M, Garg P, HedgeA. Determinants of delayed wound healing in postoperative patients: a multihospital-based prospective study performed in South India. Wounds. 2025;37(9):340-349. doi:10.25270/wnds/24216
- Calderwood MS, Kleinman K, Bruce CB, et al. National validation of the Centers for Medicare & Medicaid Services strategy for identifying potential surgical-site infections following colon surgery and abdominal hysterectomy. Infect Control Hosp Epidemiol. 2024;45(2):167-173. doi:10.1017/ice.2023.193
- Liu B, Ye J, Sun W, Yang Y, Zhu J, Zhao S. Role of wound protectors in preventing surgical site infection in patients undergoing abdominal surgery: a meta-analysis of randomized controlled trials. BMC Surg. 2025;25(1):72. doi:10.1186/s12893-025-02809-9
- Savio L, Simeone P, Baron S, et al. Surgical site infection in severe trauma patients in intensive care: epidemiology and risk factors. Ann Intensive Care. 2024;14(1):136. doi:10.1186/s13613-024-01370-7
- Leblebicioglu H, Erben N, Rosenthal VD, et al. Surgical site infection rates in 16 cities in Turkey: findings of the International Nosocomial Infection Control Consortium (INICC). Am J Infect Control. 2015;43(1):48-52. doi:10.1016/j.ajic.2014.09.017
- Aktaş A, Güner A, Güneş O, et al. A detailed analysis of surgical site infections and risk factors: a multicentric cohort study in Türkiye. Journal of Experimental and Clinical Medicine. 2024;41(2):361-369.
- Alkaaki A, Al-Radi OO, Khoja A, et al. Surgical site infection following abdominal surgery: a prospective cohort study. Can J Surg. 2019;62(2):111-117.doi:10.1503/cjs.004818
- Rezaei AR, Zienkiewicz D, Rezaei AR. Surgical site infections: a comprehensive review. J Trauma Inj. 2025;38(2):71-81. doi:10.20408/jti.2025.0019
- Normandin S, Safran T, Winocour S, et al. Negative pressure wound therapy: mechanism of action and clinical applications. Semin Plast Surg. 2021;35(3):164-170. doi:10.1055/s-0041-1731792
- Tuuli MG, Liu J, Tita ATN, et al. Effect of prophylactic negative pressure wound therapy vs standard wound dressing on surgical-site infection in obese women after cesarean delivery: a randomized clinical trial. JAMA. 2020;324(12):1180-1189. doi:10.1001/jama.2020.13361
- Li PY, Yang D, Liu D, Sun SJ, Zhang LY. Reducing surgical site infection with negative-pressure wound therapy after open abdominal surgery: a prospective randomized controlled study. Scand J Surg. 2017;106(3):189-195. doi:10.1177/1457496916668681
- Allegranzi B, Zayed B, Bischoff P, et al. New WHO recommendations on intraoperative and postoperative measures for surgical site infection prevention: an evidence-based global perspective. Lancet Infect Dis. 2016;16(12):e288-e303. doi:10.1016/S1473-3099(16)30402-9
- GlobalSurg Collaborative. Surgical site infection after gastrointestinal surgery in high-income, middle-income, and low-income countries: a prospective, international, multicentre cohort study. Lancet Infect Dis. 2018;18(5):516-525. doi:10.1016/S1473-3099(18)30101-4
- Atherton K, Brown J, Clouston H, et al; SUNRRISE Trial Study Group. Negative pressure dressings to prevent surgical site infection after emergency laparotomy: the SUNRRISE randomized clinical trial. JAMA. 2025;333(10):853-863. doi:10.1001/jama.2024.24764
- Khan M, Rooh-ul-Muqim, Zarin M, Khalil J, Salman M. Influence of ASA score and Charlson comorbidity index on the surgical site infection rates. J Coll Physicians Surg Pak. 2010;20(8):506-509.
- Bhat RA, Isaac NV, Joy J, Chandran D, Jacob KJ, Lobo S. The effect of American Society of Anesthesiologists score and operative time on surgical site infection rates in major abdominal surgeries. Cureus. 2024;16(2):e55138. doi:10.7759/cureus.55138
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59(2):e10-e52. doi:10.1093/cid/ciu296
- De Simone B, Sartelli M, Coccolini F, et al. Intraoperative surgical site infection control and prevention: a position paper and future addendum to WSES intra-abdominal infections guidelines. World J Emerg Surg. 2020;15(1):10. doi:10.1186/s13017-020-0288-4
- Calu V, Piriianu C, Miron A, Grigorean VT. Surgical site infections in colorectal cancer surgeries: a systematic review and meta-analysis of the impact of surgical approach and associated risk factors. Life (Basel). 2024;14(7):850. doi:10.3390/life14070850
- Papadopoulos A, Machairas N, Tsourouflis G, et al. Risk factors for surgical site infections in patients undergoing emergency surgery: a single-centre experience. In Vivo. 2021;35(6):3569-3574. doi:10.21873/invivo.12660
- Martin ET, Kaye KS, Knott C, et al. Diabetes and risk of surgical site infection: a systematic review and meta-analysis. Infect Control Hosp Epidemiol. 2016;37(1):88-99. doi:10.1017/ice.2015.249
- BR DS, Karim HMR, Agrawal N, Kumar M. The relation of preoperative HbA1c level with intraoperative and postoperative complications in type-2 diabetic patients: an observational study. Cureus. 2024;16(7):e64487. doi:10.7759/cureus.64487
- Tan DJH, Yaow CYL, Mok HT, et al. The influence of diabetes on postoperative complications following colorectal surgery. Tech Coloproctol. 2021;25(3):267-278. doi:10.1007/s10151-020-02373-9
- Proctor VK, O’Connor OM, Burns FA, et al. Surgical site infections after emergency hernia repair: substudy from the management of acutely symptomatic hernia (MASH) study. BJS Open. 2023;7(1):zrac155. doi:10.1093/bjsopen/zrac155
- Pellino G, Sciaudone G, Selvaggi F, Canonico S. Prophylactic negative pressure wound therapy in colorectal surgery: effects on surgical site events—current status and call to action. Updates Surg. 2015;67(3):235-245. doi:10.1007/s13304-015-0298-z
- Wagner JC, Wetz A, Wiegering A, et al. Successful surgical closure of infected abdominal wounds following preconditioning with negative pressure wound therapy. Langenbecks Arch Surg. 2021;406(7):2479-2487. doi:10.1007/s00423-021-02221-w
- de Moraes FCA, Kelly FA, Souza MEC, Burbano RMR. Impact of adjuvant chemotherapy on survival after pathological complete response in rectal cancer: a meta-analysis of 31,558 patients. Int J Colorectal Dis. 2024;39(1):96. doi:10.1007/s00384-024-04668-x
- Murphy PB, Knowles S, Chadi SA, et al. Negative pressure wound therapy use to decrease surgical nosocomial events in colorectal resections (NEPTUNE): a randomized controlled trial. Ann Surg. 2019;270(1):38-42. doi:10.1097/SLA.0000000000003111
- Bosut MP, İspir P, Baysal G, Baysu F. Economic burden of surgical site infections in Turkey. Value in Health. 2020;23(suppl 2):S569. doi:10.1016/j.jval.2020.08.996
- Groenen H, Jalalzadeh H, Buis DR, et al. Incisional negative pressure wound therapy for the prevention of surgical site infection: an up-do-date meta-analysis and trial sequential analysis. EClinicalMedicine. 2023;62:102105. doi:10.1016/j.eclinm.2023.102105
- Guo Q. Comparison and evaluation of negative pressure wound therapy versus standard wound care in the treatment of diabetic foot ulcers. BMC Surg. 2025;25(1):208. doi:10.1186/s12893-025-02885-x
- Upton D, Stephens D, Andrews A. Patients’ experiences of negative pressure wound therapy for the treatment of wounds: a review. J Wound Care. 2013;22(1):34-39. doi:10.12968/jowc.2013.22.1.34
- Ousey KJ, Milne J, Cook L, Stephenson J, Gillibrand W. A pilot study exploring quality of life experienced by patients undergoing negative-pressure wound therapy as part of their wound care treatment compared to patients receiving standard wound care. Int Wound J. 2014;11(4):357-365. doi:10.1111/j.1742-481X.2012.01098.x
- Stanciu CT, Vermesan D, Pop DL, Hogea B, Vlad SV. Functional and clinical outcomes in acute wound management: measuring the impact of negative pressure wound therapy and specialized physical therapy. Life (Basel). 2025;15(4):511. doi:10.3390/life15040511
- Nicolazzo D, Rusin E, Varese A, Galassi M. Negative pressure wound therapy and traditional dressing: an Italian health technology assessment evaluation. Int J Environ Res Public Health. 2023;20(3):2400. doi:10.3390/ijerph20032400