Bimekizumab Demonstrates Sustained 3-Year Efficacy Across Joint, Skin, and Nail Domains in Psoriatic Arthritis
Long-term results from 2 phase 3 studies indicate that bimekizumab provides sustained efficacy across multiple disease domains in patients with active psoriatic arthritis (PsA) and coexisting psoriasis, with a consistent safety profile over 3 years.
“Patients with PsA and coexisting psoriasis often experience more severe disease manifestations and reduced treatment durability,” the investigators noted. Bimekizumab, a monoclonal antibody targeting both IL-17A and IL-17F, was evaluated for its ability to deliver “sustained, long-term control across multiple disease domains.”
This post hoc analysis included patients from the BE OPTIMAL (biologic-naïve) and BE COMPLETE (TNF inhibitor-inadequate responder [TNFi-IR]) phase 3 trials with ≥3% body surface area psoriasis at baseline. Patients received bimekizumab 160 mg every 4 weeks, with outcomes assessed through 3 years using modified non-responder imputation and observed case analyses.
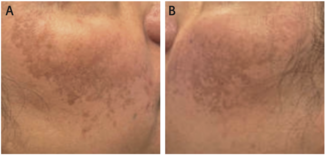
Efficacy outcomes remained robust over time. High proportions of patients achieved Psoriasis Area and Severity Index 100 (PASI100), American College of Rheumatology 50 (ACR50), resolution of nail psoriasis (modified Nail Psoriasis Severity Index=0), and swollen joint count of 0 through week 160/156. In biologic-naïve patients, PASI100 responses reached approximately 74% at year 3, while ACR50 responses approached 70%. Similar sustained responses were observed in TNFi-IR patients, indicating efficacy regardless of prior biologic exposure.
Safety outcomes were consistent with prior reports. Over 3 years, most treatment-emergent adverse events were non-serious, with low rates of serious infections, malignancies, and major adverse cardiovascular events. No new safety signals were identified during long-term follow-up.
“Bimekizumab resulted in sustained clinical efficacy up to 3 years in patients with PsA who also had baseline psoriasis, with high proportions… achieving efficacy outcomes across the domains of joints, skin, and nails,” the authors concluded. “Bimekizumab was well tolerated to 3 years… with no new safety signals identified.”
These findings support bimekizumab as a durable, multi-domain treatment option for PsA patients with concomitant psoriasis, including those with prior biologic exposure.
Reference
Lebwohl M, Asahina A, Gottlieb AB, et al. Bimekizumab safety and sustained long-term efficacy in patients with active psoriatic arthritis and baseline psoriasis: up to 3-year results from two phase 3 studies. Presented at: American Academy of Dermatology Annual Meeting; March 27–31, 2026; Denver, Colorado.