What Is This Persistent Scaly Plaque in the Axilla?
Case Report
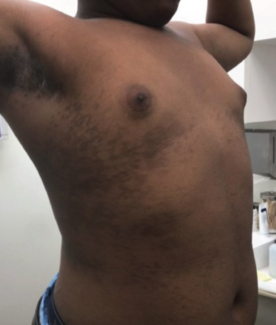
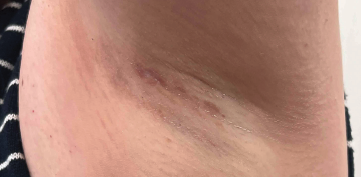
right axilla.
A 55-year-old woman with a history of hypertension and nonalcoholic fatty liver disease presented with a pruritic rash localized to her right axilla for 2 months. She reported similar relapsing eruptions in both axillae over the past 6 years, which resolved with pimecrolimus and triamcinolone but recurred upon cessation. She also reported using fragrance-free body wash and moisturizers and switching her laundry detergents periodically, although the rash has persisted. She denied new hygiene products, recent infections, or systemic symptoms. She only takes amlodipine and has no known allergies. Physical examination revealed a 3-cmx 2-cm sharply demarcated, reddish-violaceous, eroded plaque with fibrinous scaling and surrounding erythema in the right axilla (Figure 1). A shave biopsy was performed.
What is your diagnosis?
Scroll below to find out!
Diagnosis
Granular Parakeratosis
Granular parakeratosis (GP) is a rare, chronic disorder of keratinization characterized by pruritic, hyperkeratotic plaques that predominantly affect intertriginous areas, such as the axillae, inframammary folds, and inguinal region. Lesions often exhibit peripheral scaling with central hyperpigmentation and may be pruritic.1 First described by Northcutt et al. in 1991, GP has since been identified as a reaction pattern rather than a distinct dermatosis, with multiple proposed etiologies, including irritant contact dermatitis (ICD), microbial involvement, and surfactant-induced epidermal dysfunction.1-3 Recent reports have implicated benzalkonium chloride, a quaternary ammonium compound commonly found in personal care products and disinfectants, as a frequent trigger for GP.4 The condition has also been linked to sodium lauryl sulfate, an anionic surfactant found in cleansing agents.5 Additional triggers include friction, occlusion, and excessive sweating, which can compromise the skin barrier, allowing irritants or microbes to induce epidermal hyperplasia and dyskeratosis.6
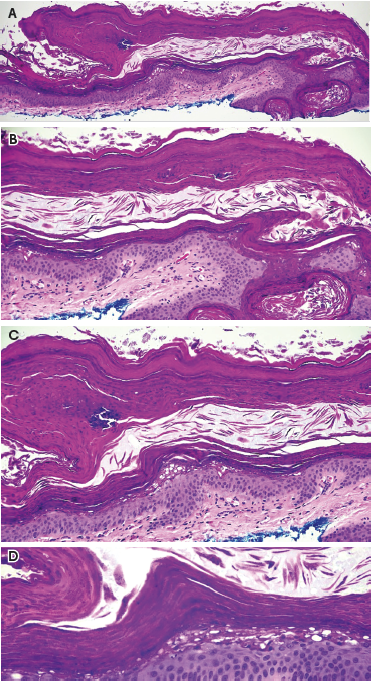
Histologically, GP is distinguished by parakeratosis, retention of keratohyalin granules within the stratum corneum, hypergranulosis, and acanthosis (Figure 2). These characteristics, along with the presence of elongated rete ridges and dermal inflammation, differentiate it from other hyperkeratotic disorders and intertriginous dermatoses.7,8
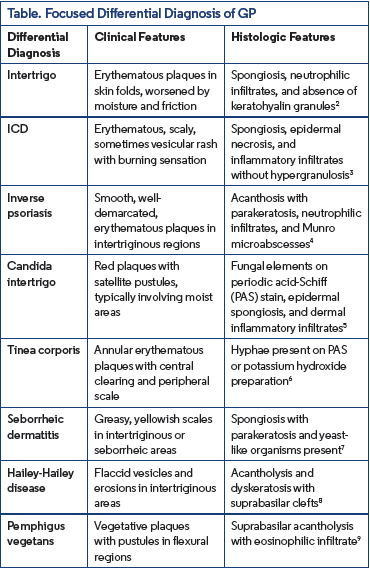
Differential Diagnosis
The diagnosis of GP can be challenging due to its variable clinical presentation and histopathologic overlap with other dermatologic conditions. Clinically, GP may resemble various inflammatory, infectious, or keratinization disorders, leading to potential misdiagnosis.Given these challenges, it is essential to maintain a broad differential diagnosis when evaluating persistent, hyperkeratotic,or intertriginous lesions. Key clinical and histopathologic mimickers of GP include intertrigo, ICD, inverse psoriasis, candida intertrigo, tinea corporis, seborrheic dermatitis, Hailey-Hailey disease, and pemphigus vegetans2-9 (Table).
Management and Treatment
The management of GP centers on reducing inflammation, restoring the epidermal barrier, and eliminating potential irritants, with multiple treatment modalities available depending on disease severity and patient response.
Topical corticosteroids remain the cornerstone of therapy, particularly moderate-to-high potency agents such as triamcinolone 0.1%, which are effective in controlling inflammation and pruritus.10 They provide symptomatic relief and are often the first-line treatment in active disease. Adjunctive keratolytics, including ammonium lactate, urea, and salicylic acid, facilitate epidermal turnover by softening and reducing hyperkeratosis, which can contribute to disease persistence.11 In cases where corticosteroids prove insufficient, topical vitamin D analogues such as calcipotriol have been reported to improve lesions, suggesting a role in modulating epidermal differentiation.12
Infections, whether bacterial or fungal, can complicate or exacerbate GP. Topical and systemic antibiotics may be beneficial in cases where bacterial colonization is suspected. Reports of GP associated with Klebsiella pneumoniae have demonstrated a positive response to oral amoxicillin-clavulanic acid, underscoring the importance of targeted antimicrobial therapy when indicated.13 Likewise, in patients with suspected secondary fungal involvement, topical or systemic antifungals can be useful in preventing persistent inflammation and superinfection.14
For refractory cases, alternative treatment modalities have been explored. Botulinum toxin injections have shown promise, particularly in patients with GP affecting intertriginous areas, where excessive sweating and friction exacerbate symptoms. By reducing local perspiration and irritation, botulinum toxin may improve disease control in these challenging cases.15 Additionally, avoidance of irritants is critical; patients should discontinue the use of benzalkonium chloride-containing detergents, surfactants, and antiseptics, as these compounds have been implicated in triggering or worsening the condition.4,5
More advanced treatment options include retinoids, which play a role in epidermal differentiation and reducing hyperkeratosis. Both topical agents and oral retinoids have been successfully used in certain cases, particularly those with significant hyperkeratosis or recalcitrant disease.16 In patients who fail conventional therapies, cryotherapy and laser therapy have also been explored. These modalities may help in persistent, treatment-resistant cases, particularly where scarring or thickened plaques contribute to ongoing symptoms.17
Ultimately, the management of GP requires a multifaceted approach tailored to individual patient presentations, incorporating anti-inflammatory agents, keratolytics, infection control, and adjunctive treatments for refractory disease. Further research is needed to optimize long-term treatment strategies and improve patient outcomes.
Our Patient
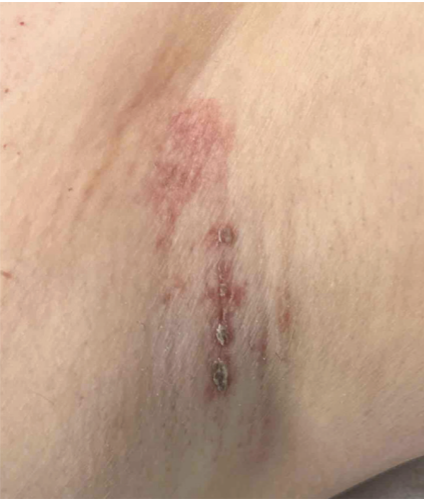
Diagnosis of GP was confirmed through biopsy and histopathologic examination. The patient was counseled to avoid triggers, especially fragrances and benzalkonium chloride, and use gentle emollients such as petroleum jelly. Topical gentamicin 0.1% ointment twice a day was also prescribed for a 2- to 3-week course to prevent superinfection. Follow up after 3 weeks showed significant improvement (Figure 3).
Conclusion
Granular parakeratosis is a unique but underrecognized disorder of keratinization with a complex etiology, often associated with allergic contact dermatitis, particularly in response to benzalkonium chloride. This case underscores the importance of identifying environmental and chemical triggers in the management of GP. The strong correlation between GP and ICD necessitates a multifaceted approach, including discontinuation of suspected irritants, barrier repair, and targeted therapies such as corticosteroids and keratolytics.
Given the chronic and relapsing nature of GP, clinicians should maintain a high index of suspicion in patients presenting with persistent intertriginous dermatoses. The increasing evidence implicating benzalkonium chloride and other surfactants highlights the need for heightened awareness and patient education regarding product selection. Further research is needed to elucidate the precise pathogenesis of GP and establish standardized treatment protocols. By recognizing GP as a reactive pattern rather than a distinct entity, we can improve diagnostic accuracy and optimize patient outcomes through tailored management strategies. As our understanding of GP evolves, future investigations into genetic predisposition, immune dysregulation, and improved therapeutic modalities will be critical in advancing patient outcomes.
Dr Hussain is a dermatologist at Galaria Plastic Surgery & Dermatology in Chantilly, VA. Umayr R. Shaikh is an MS4 at Georgetown University School of Medicine in Washington, DC. Kabir Al-Tariq is a PGY1 resident at Garnet Health Medical Center in Middletown, NY. Dr Terrano is a dermatopathologist at Bethesda Dermatopathology Lab in Bethesda, MD.
Disclosure: The authors report no relevant financial relationships.
References
1. Northcutt AD, Nelson DM, Tschen JA. Axillary granular parakeratosis. J Am Acad Dermatol. 1991;24(4):541-544. doi:10.1016/0190-9622(91)70078-g
2. Ding CY, Liu H, Khachemoune A. Granular parakeratosis: a comprehensive review and a critical reappraisal. Am J Clin Dermatol. 2015;16(6):495-500. doi:10.1007/s40257-015-0148-2
3. Scheinfeld NS, Mones J. Granular parakeratosis: pathologic and clinical correlation of 18 cases of granular parakeratosis. J Am Acad Dermatol. 2005;52(5):863-867.doi:10.1016/j.jaad.2004.12.031
4. Dear K, Gan D, Stavrakoglou A, Ronaldson C, Nixon RL. Hyperkeratotic flexural erythema (more commonly known as granular parakeratosis) with use of laundry sanitizers containing benzalkonium chloride. Clin Exp Dermatol. 2022;47(12):2196-2200. doi:10.1111/ced.15358
5. Segal R, Eskin-Schwartz M, Trattner A, et al. Recurrent flexural pellagroid dermatitis: an unusual variant of irritant contact dermatitis. Acta Derm Venereol. 2015;95(1):116-117. doi:10.2340/00015555-1862
6. Resnik KS, Kantor GR, DiLeonardo M. Dermatophyte-related granular parakeratosis. Am J Dermatopathol. 2004;26(1):70-71. doi:10.1097/00000372-200402000-00011
7. Pock L, Čermáková A, Zipfelová J, Hercogová J. Incidental granular parakeratosis associated with molluscum contagiosum. Am J Dermatopathol. 2006;28(1):45-47. doi:10.1097/01.dad.0000157448.54281.d9
8. Compton AK, Jackson JM. Isotretinoin as a treatment for axillary granular parakeratosis. Cutis. 2007;80(1):55-56.
9. Ravitskiy L, Heymann WR. Botulinum toxin-induced resolution of axillary granular parakeratosis. Skinmed. 2005;4(2):118-120. doi:10.1111/j.1540-9740.2005.03700.x
10. Contreras ME, Gottfried LC, Bang RH, Palmer CH. Axillary intertriginous granular parakeratosis responsive to topical calcipotriene and ammonium lactate. Int J Dermatol. 2003;42(5):382-383. doi:10.1046/j.1365-4362.2003.01722.x
11. Mehregan DA, Vandersteen P, Sikorski L, Mehregan DR. Axillary granular parakeratosis. J Am Acad Dermatol. 1995;33(2 Pt 2):373-375. doi:10.1016/0190-9622(95)91437-4
12. Wallace CA, Pichardo RO, Yosipovitch G, Hancox J, Sangueza OP. Granular parakeratosis: a case report and literature review. J Cutan Pathol. 2003;30(5):332-335. doi:10.1034/j.1600-0560.2003.00066.x
13. Herat A, Gonzalez Matheus G, Kumarasinghe SP. Hyperkeratotic flexural erythema/granular parakeratosis responding to doxycycline. Australas J Dermatol.2022;63(3):368-371. doi:10.1111/ajd.13868
14. Kumarasinghe SPW, Chandran V, Raby E, Wood B. Hyperkeratotic flexural erythema responding to amoxicillin-clavulanic acid therapy: report of four cases. Australas J Dermatol. 2019;60(4):311-314. doi:10.1111/ajd.13069
15. Lee G, Fischer G. Support for a microbial basis for hyperkeratotic flexural erythema. Australas J Dermatol. 2020;61(4):377-378. doi:10.1111/ajd.13303
16. Brown SK, Heilman ER. Granular parakeratosis: resolution with topical tretinoin. J Am Acad Dermatol. 2002;47(5 Suppl):S279-S280. doi:10.1067/ mjd.2002.109252
17. Laimer M, Emberger M, Brunasso AM, et al. Laser for the treatment of granular parakeratosis. Dermatol Surg. 2009;35(2):297-300. doi:10.1111/ j.1524-4725.2008.01052.x