System Matters: How Structure and Access Shape CLTI Outcomes
Abstract
Chronic limb-threatening ischemia (CLTI) is characterized by ischemic rest pain, tissue loss, and a high risk of major amputation and death. Limb threat is commonly described using both the Rutherford classification and the Wound, Ischemia, and Foot Infection (WIfI) framework; while WIfI provides granular prognostic stratification, Rutherford remains the most widely adopted and clinically intuitive classification in referral pathways and real-world practice. Despite major advances in endovascular and surgical techniques, outcomes in CLTI remain poor. Increasing evidence suggests that these outcomes are determined not only by procedural expertise and device technology but by the organization and timeliness of care.
Delays occur at multiple stages of the CLTI pathway, including disease recognition, referral to specialist services, access to diagnostic testing and revascularization, and post-intervention surveillance. Observational studies consistently associate prolonged time to treatment with higher risks of major amputation, impaired wound healing, and mortality with paradoxically longer delays affecting the most clinically vulnerable patients. High-performing models—including multidisciplinary limb preservation programs, hub-and-spoke referral networks, and formally defined CLTI teams—compress time to revascularization, reduce major amputation rates, and mitigate socioeconomic disparities, even among patients with severe disease and high comorbidity burdens. However, substantial system-level gaps persist, including under-recognition in primary care, fragmented referral pathways, diagnostic bottlenecks, misaligned reimbursement, and inconsistent longitudinal surveillance.
Taken together, these findings support reframing CLTI as a systems disease. Improving outcomes requires not only technical skills but deliberate pathway design with rapid triage, guaranteed access to revascularization, integrated wound and perfusion surveillance, and accountability for longitudinal limb salvage.
J CRIT LIMB ISCHEM 2026:6(1):E1-E9. doi: 10.25270/jcli/CLIG-2500010
Key words: chronic limb-threatening ischemia, amputation, time to treatment, multidisciplinary care surveillance.
Chronic limb-threatening ischemia (CLTI)—the end stage of peripheral arterial disease—is characterized by ischemic rest pain, tissue loss, infection, and imminent risk of major amputation. The Wound, Ischemia, and Foot Infection (WIfI) framework and the PLAN (Patient, Limb severity, ANatomic complexity)/GLASS (GLobal Anatomic Staging System) approach are endorsed to standardize revascularization decisions and study design.1
Historically, vascular programs have prioritized technical optimization (open bypass, multilevel endovascular recanalization, advanced crossing/debulking strategies, and drug-eluting or bioabsorbable technologies), yet mounting evidence indicates that for many patients the decisive determinant of outcome is not how we revascularize but whether and when revascularization occurs.2
Along real-world care pathways, patients experience serial failures: delayed recognition, prolonged referral processes, limited access to diagnostics or revascularization, and inconsistent post-procedural surveillance, each missed step driving preventable amputation, readmission, and death. In a systematic review of 32 studies including approximately 71,310 patients, median times from symptom onset to specialist assessment ranged from 15 to 126 days, and from assessment to treatment from 1 to 91 days; only 4 studies defined “delay”, and definitions were inconsistent, highlighting the need for explicit time standards.2 UK service guidance (PAD-QIF) therefore proposes operational targets from referral to revascularization (≤5 days for nonelective pathways and ≤14 days for outpatient pathways), underscoring that delay thresholds should be operational, not aspirational.2
The burden of CLTI is rising with aging, diabetes, and multimorbidity. Those affected face high risks of mortality, major adverse cardiovascular events, and amputation, with marked disparities by access, literacy, minority status, and socioeconomic deprivation.3 Waiting times beyond approximately7 days correlate with increased postoperative amputation and in-hospital mortality, while paradoxically the sickest patients often wait the longest.2 Early intervention is not only clinically advantageous but economically favorable: early endovascular treatment at the rest-pain stage provides an Incremental Cost-Effectiveness Ratio approximately equal to €12,438 ($14,764)/Quality-Adjusted Life Year (QALY), increases 5-year QALYs (from 2.0 to 2.5), and reduces major amputations by 6% compared with delayed intervention.4
This review examines CLTI as a systems disease, focusing on failure points in recognition, referral, and access; why delays translate into limb loss and death; organizational models that mitigate delay; and the often-neglected role of longitudinal surveillance.
Failure Points in the CLTI Pathway: Recognition, Referral, and Access
Patients with rest pain and tissue loss may spend months in primary care or wound-care pathways without vascular specialist assessment, and once referred, may still wait weeks for revascularization. The Global Vascular Guidelines emphasize urgent referral to vascular specialists, objective hemodynamic assessement, WIfI staging, and PLAN/GLASS-guided revascularization, highlighting standardization as foundational to equity and quality of care.1
The reasons for this delay are various and are reported in Figure 1.
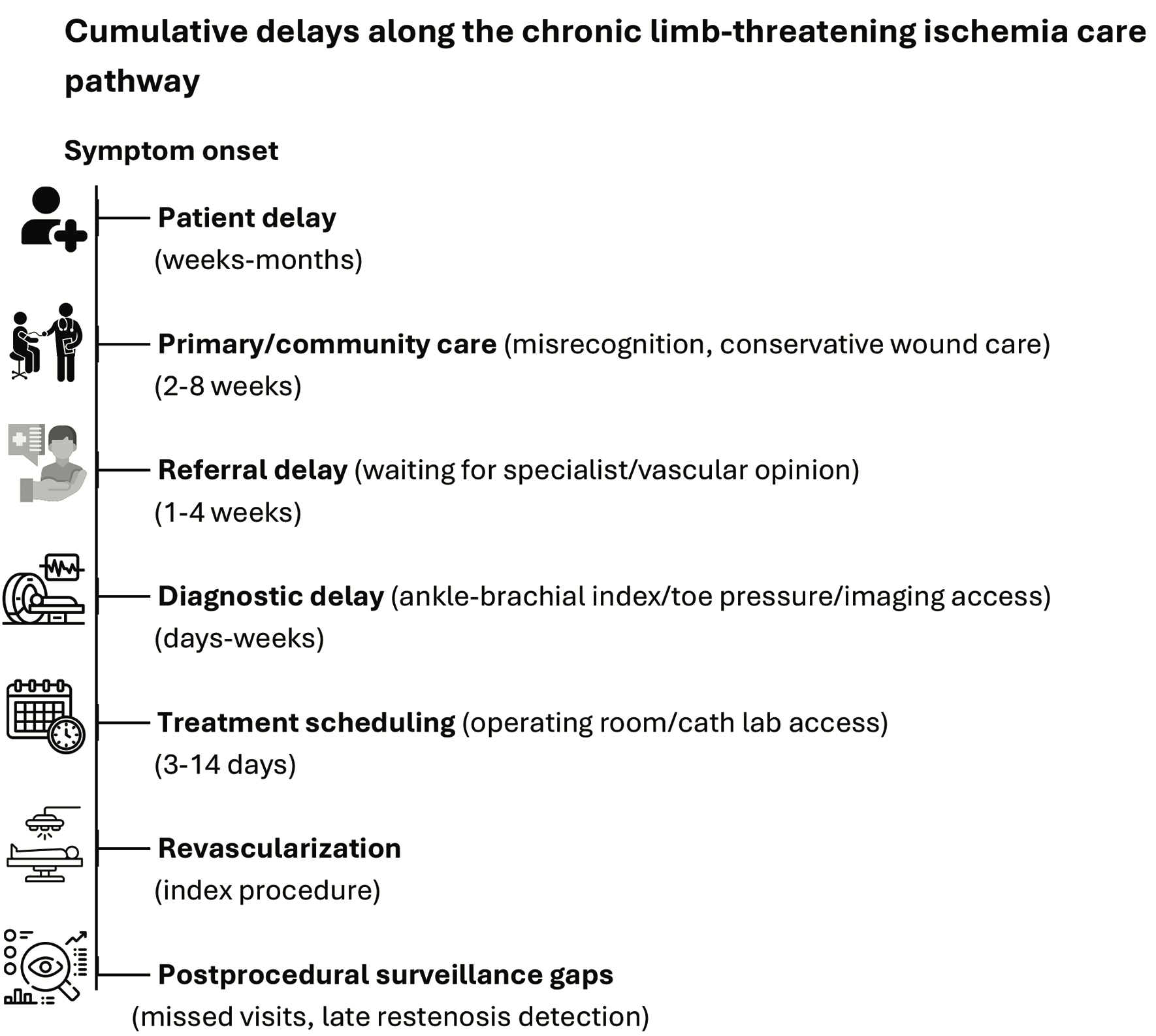
Patient-level factors
Patients frequently normalize ischemic rest pain or under-recognize tissue loss, while neuropathy or retinopathy blunts symptom perception.5 Interview studies consistently report limited understanding of CLTI and uncertainty about when specialist care is required.6
Infection is often managed locally without timely escalation, and inadequate education regarding diabetic foot ulcers (DFUs) repeatedly emerges as a driver of delay.7 The absence of clear public messaging—“limb pain plus ulcer equals a vascular emergency”—further delays help-seeking.
Non-specialist clinician factors
Recognition and referral outside vascular services remain inconsistent. General practitioners, community nurses, diabetologists, and podiatrists report uncertainty regarding CLTI presentation, limited training or equipment for vascular examination, and difficulty interpreting ischemic rest pain.2,8 Misdiagnosis or under-recognition is common, and some clinicians report apprehension about referral.2,8
These challenges are compounded by increasing primary-care workload without commensurate staffing or funding, aging and frail patient populations, and suboptimal digital tools, which impede thorough assessment and smooth referral.9 Limited awareness of guidelines and locally agreed pathways further influences referral behaviour.10,11 Some clinicians delay referral out of concern about “over-referring” or “bothering the specialist”, or from a perception that “nothing can be done”. Inaccurate grading of severity and urgency is a recognized cause of delay,6,7,12,13 and each additional professional encountered before specialist assessment markedly increases time to treatment.14
Notably, 17% of podiatrists report seeking vascular input only if a DFU fails to heal after 42 days—an interval likely to compromise limb salvage.11
The Rutherford classification remains the most widely used and clinically intuitive framework for describing CLTI, particularly in referral pathways and real-world practice. Rutherford categories 4 through 6 (ischemic rest pain to tissue loss) continue to define CLTI in most clinical trials, registries, and health systems worldwide and are frequently used by non-vascular clinicians to determine referral urgency.1 By contrast, WIfI classification provides a more granular assessment of limb threat and amputation risk but is more complex and variably adopted outside of specialized centers.15
Failure to recognize Rutherford 5 and 6 disease as a vascular emergency is a recurrent contributor to delayed referral. In this context, Rutherford staging remains highly relevant for front-end recognition and triage, while WIfI may be most valuable once patients reach specialist multidisciplinary teams, where it can guide revascularization strategy and surveillance intensity.
Structural access barriers
Even when CLTI is suspected, system frictions delay treatment. Geography and capacity constraints (distance to an arterial hub, limited specialist clinic availability, and restricted operating-room or angiography-suite access) extend timelines. Centralization may further impede access for patients with diabetes and PAD in settings constrained by funding, staffing, or transport. Ambiguity regarding referral points, urgency, and pre-referral imaging contributes to hesitation and circular referrals, while outpatient access to ankle-brachial index (ABI), toe pressure testing, duplex ultrasound, and computed tomography angiography (CTA) remains variable.16
Service access and communication failures compound these barriers. Specialist teams report difficulties engaging diabetic-foot services owing to funding, staffing, and centralization,7 and communication gaps within multidisciplinary teams are a recognized root cause of delay. Across Europe, primary-care surveys show substantial variation in multidisciplinary team meeting (MDT) use, referral thresholds, and awareness of specialist services.17
Within hub-and-spoke networks, these issues translate into measurable time costs: approximately 45% of outpatients exceed the 14-day target from specialist assessment to revascularization.18 Patients presenting with tissue loss experience longer delays, likely reflecting greater comorbidity or frailty and the need for medical optimization before intervention.18 Moreover, patients first assessed at non-arterial spoke hospitals wait substantially longer than patients assessed at arterial hubs, with more than a twofold difference for emergency pathways.18
Communication failures and equity
Limited feedback to referrers perpetuates diagnostic insecurity and hesitancy. The absence of shared platforms for documenting wound status and ischemia leads to fragmented, asynchronous care. The cumulative effect is inequitable access: patients with diabetes, dialysis dependence, lower health literacy, and socioeconomic deprivation consistently wait longer and present later, compounding their risk of adverse outcomes.18
Taken together, patient-, clinician-, and system-level barriers interact to create compounded delay, such that patients with the highest limb threat are paradoxically the least likely to receive timely revascularization.
Why Delays Matter
Time to treatment is survival-relevant. Across pathways, delays beyond approximately 7 days are associated with higher postoperative major amputation and in-hospital mortality.18 Among emergency admissions, very early revascularization (<3 days) is associated with higher in-hospital mortality—likely confounding by severity (sickest, most ischemic patients triaged fastest)—but the lowest risk window typically falls between days 3 and 7, after which risk rises progressively.18
A Finnish study demonstrated that a delay exceeding 2 weeks from primary-care assessment to revascularization independently predicted major amputation in patients with diabetes and CLTI (odds ratio 3.1, 95% confidence interval [CI] 1.4–6.9).18 Early treatment is also economically dominant or highly cost-effective, increasing QALYs and reducing amputations compared with delaying intervention.4
The effect of delay is not uniform. Older age, high comorbidity burden, tissue loss, and infection are consistently associated with longer, not shorter, delays to revascularization—meaning that those at greatest risk often wait the longest.18 This inverted triage logic allows stable claudication to enter vascular pathways faster than septic tissue loss in a frail, dialysis-dependent diabetic foot. Inpatients face additional delays related to bed flow and operating room/angiography suite access, or the need to coordinate multiple teams, while outpatients are limited by referral mechanics and scheduling.18
The BASIL-2 and BASIL-3 trials further contextualize these findings. BASIL-2 compared vein bypass with endovascular therapy for infrapopliteal disease,19 while BASIL-3 evaluated different endovascular strategies in severe limb ischemia.20 Although neither trial explicitly studied treatment delay, both highlight that advanced CLTI frequently requires complex, resource-intensive interventions delivered in specialized centers. Late presentation narrows therapeutic options and may partially explain why real-world outcomes often fall short of trial results, reinforcing the importance of timely escalation.
What High-Performing Systems Do Differently
If delays cost limbs, the next question is: How can systems prevent them?
Limb preservation programs
Limb preservation programs (LPPs) co-locate vascular surgery, podiatry, wound care, and allied specialties within a dedicated pathway. In a mixed-methods cohort (2014–2023; 983 limbs), LPP patients were more likely to be non-White, diabetic, and dialysis dependent, with higher WIfI stage and socioeconomically deprived; yet they achieved similar freedom from death, major adverse limb events (MALE), and patency loss compared to healthier, less-deprived patients in traditional clinics. Among the most deprived, LPP patients experienced later patency loss than traditional clinic patients.3
Qualitative interviews identified transportation barriers, visit burden, disease understanding, distrust, and racial discordance as key obstacles partially mitigated by co-location and consistent teams.3 Core components of LPPs include direct referral access (no “gatekeeper”), rapid hemodynamic assessment (ABI, toe-brachial index [TBI], duplex), integrated wound and infection management, access to urgent revascularization (endovascular, open, or hybrid), and joint decision-making.
Hub-and-spoke networks
In hub-and-spoke models:
- The hub is a high-volume vascular center with hybrid operating room, endovascular capability, bailout open surgical options, and a defined MDT.
- The spokes are community/secondary hospitals and outpatient providers (including general practitioners, diabetologists, and wound nurses) trained to identify suspected CLTI and trigger a direct referral pathway.
- Routine follow-up and wound care can be decentralized back to the spoke, potentially via shared telemedicine and image-sharing, while complex decision-making and urgent revascularization are centralized at the hub.
Evidence from England suggests that hub-based patients receive revascularization substantially faster with corresponding reductions in major amputation and in-hospital death:
- Hub inpatients: median 5 days from diagnosis to revascularization vs 12 days for spoke inpatients.
- Hub outpatients: median 13 days vs 26 days for spoke outpatients.18
Insights from BEST-CLI
The BEST-CLI trial was not designed to evaluate delays, but it provides critical insights into how system maturity and team structure with CLTI care teams influence outcomes in CLTI. BEST-CLI demonstrated that, in patients with an adequate single-segment great saphenous vein, surgical bypass resulted in fewer MALE or death than endovascular therapy, while outcomes were similar between strategies when a suitable vein was unavailable.21 These findings underscore that appropriate patient selection and access to multiple revascularization options are essential components of high-quality CLTI care.
Subsequent analyses have shown that outcomes varied substantially by center characteristics.22 Only 31% of centers reported formal multidisciplinary CLTI teams, yet these centers, with higher procedural volume and access to both open and endovascular techniques, treated the majority of enrolled patients (83%, n = 1520), and achieved significantly lower major amputation rates (hazard ratio, 0.60; 95% CI 0.42–0.86; P=.005). These findings underscore that organizational structure and access to comprehensive expertise, rather than device choice alone, are decisive determinants of outcome.
Infrastructure, Incentives, and the Economics of Delay
Even in well-structured teams, 2 system frictions repeatedly undermine care:
Diagnostic bottlenecks: Toe pressure measurement, duplex arterial mapping, and point-of-care CTA runoff (defined as rapid, protocolized CTA performed at or near the time of clinical assessment to guide revascularization) are not uniformly available. Access to magnetic resonance angiography (MRA) and contrast-enhanced MRA is likewise variable. Underuse of basic hemodynamic testing creates diagnostic ambiguity, which fuels referral delay.23
Misaligned reimbursement: Reimbursement models often do not reflect the reality of CLTI work. Long, multivessel tibial/pedal endovascular procedures in frail, septic patients are not reimbursed proportionally to complexity or procedural time. The result is perverse: the sickest limbs are the least “attractive” to treat in systems driven by procedure billing, and centers under economic pressure may undertreat, stage excessively, delay, or even elude treatment.23 That is how “undertreatment” happens: not because clinicians do not know what to do, but because the system makes optimal care irrational.
Medical Optimization: An Essential but Inconsistently Delivered Pillar
Comprehensive CLTI management extends beyond revascularization and must include rigorous infection control, metabolic management, smoking cessation, and evidence-based pharmacological therapy.1,12 These elements are integral to limb salvage, wound healing, and survival.
Infection control requires early recognition, antimicrobial therapy, timely debridement, and coordinated wound care. Poor glycemic control impairs wound healing and increases infection risk, while continued smoking is associated with graft failure, restenosis, amputation, and mortality. 24,25 Despite this, structured risk-factor modification remains inconsistently embedded.
Variation in pharmacological therapy is striking. Antiplatelets, statins, and renin–angiotensin inhibitors remain underprescribed.26 The COMPASS trial demonstrated that rivaroxaban plus aspirin reduced major adverse cardiovascular and limb events,27 and VOYAGER PAD showed similar benefit after revascularization.28 Uptake remains variable due to bleeding concerns, fragmented responsibility, and poor care transitions.
As a result, many patients with CLTI leave the hospital after revascularization without optimized secondary prevention, exposing them to preventable cardiovascular and limb events.
Follow-up and Surveillance: The Hidden Determinant of Limb Salvage
One of the most striking weaknesses in contemporary CLTI care is what happens after the index revascularization. While procedural success is often emphasized, post-intervention surveillance is a critical determinant of limb salvage, durability, and equity of outcomes. The Global Vascular Guidelines identify structured surveillance as a core component of evidence-based CLTI management, recommending structured, longitudinal follow-up that integrates objective hemodynamic assessment (preferably toe pressures in diabetic or neuropathic patients), duplex ultrasound of the treated arterial segment,29 and standardized wound documentation, embedded within a WIfI- and PLAN/GLASS-informed framework to trigger timely reintervention when failure is detected.1 However, real-world adherence to such surveillance strategies remains inconsistent, and patients at highest risk of adverse outcomes are often those least likely to benefit from organized follow-up.30,31
Risk stratification and surveillance intensity
The need for surveillance is not uniform across all patients with CLTI. Multiple studies demonstrate that patients with advanced disease (Rutherford 5-6), high WIfI stage,32 diabetes,33,34 renal failure, tissue loss, or infrapopliteal revascularization have a substantially higher risk of restenosis, nonhealing wounds, and major amputation.35 In these populations, early failure is common and often clinically silent until wounds deteriorate or infection occurs. Accordingly, intensified surveillance during the first 3 to 6 months after revascularization is particularly critical, as this period captures the highest hazard for loss of patency and limb-threatening recurrence.
Observational data consistently show that structured surveillance—combining duplex imaging with clinical and hemodynamic assessment—is associated with earlier detection of restenosis and higher rates of secondary patency and limb salvage, particularly after infrainguinal bypass and complex tibial interventions.36,37 By contrast, unstructured or symptom-driven follow-up frequently identifies failure late, when options for minor revision are limited and the likelihood of major amputation is higher.3
Optimal surveillance intervals
Although randomized trials defining optimal surveillance intervals in CLTI are lacking, consensus recommendations and registry-based evidence support early and frequent follow-up, particularly in high-risk patients.38 Most guidelines and expert pathways advocate:
- An initial post-revascularization assessment within 7 to 14 days, focused on wound status, infection control, and early hemodynamic response.
- Reassessment at 4 to 6 weeks, incorporating objective perfusion testing and duplex ultrasound of the treated segment or graft.
- Continued surveillance at 1- to 3-month intervals during the first 6 months, followed by individualized spacing thereafter based on durability, wound trajectory, and patient risk profile.
Failure to attend early post-intervention visits has been shown to carry measurable clinical consequences. In an outpatient CLTI cohort, missing the first scheduled wound-clinic visit was independently associated with significantly lower 6-month wound healing rates, with disproportionate impact among socioeconomically disadvantaged and non-White patients, highlighting surveillance as both a clinical and equity-sensitive intervention.30
Noninvasive and remote monitoring approaches
Noninvasive monitoring technologies offer an important opportunity to strengthen surveillance while reducing access barriers. Toe pressures and TBI measurements are more reliable than ABI in patients with diabetes or medial arterial calcification and provide actionable information on perfusion recovery and decline.39-41 Serial toe pressure decline has been associated with wound deterioration and risk of amputation, often preceding overt clinical failure.
Beyond traditional testing, emerging remote and point-of-care monitoring technologies may further reduce surveillance gaps. These include:
- Portable or point-of-care hemodynamic devices for toe pressure or photoplethysmography assessment in outpatient or community settings.
- Transcutaneous oxygen pressure monitoring to assess microcirculatory perfusion and predict wound healing.
- Digital wound imaging and telemedicine platforms, enabling standardized wound photography, remote review, and earlier escalation when deterioration is detected.
While high-quality outcome data for remote perfusion monitoring in CLTI remain limited, early studies suggest that telemedicine-supported wound surveillance improves attendance, accelerates reassessment, and may reduce unplanned readmissions and amputations, particularly for patients living far from arterial hubs.42 Importantly, such approaches align with hub-and-spoke models by allowing intensive follow-up to occur locally while maintaining specialist oversight.
Surveillance as a system-level intervention
These observations align with the broader delay literature: once patients leave the operating room or angiography suite, system friction often re-emerges in the form of limited access to toe-pressure testing, duplex capacity, wound photography, or clear pathways for urgent reassessment. The absence of predefined hemodynamic or imaging thresholds for reintervention further contributes to delayed response to failure. Surveillance should therefore be viewed not as an optional adjunct but as a system-level intervention designed to detect failure early, permit low-morbidity reintervention, and prevent progression to major amputation.2
In this context, effective CLTI programs treat surveillance as “inevitable rather than optional”, embedding it into standard work with pre-scheduled visits, shared documentation across disciplines, and explicit escalation criteria.
What “Good” Should Look Like
High-performing CLTI services report waiting times, make access frictionless, and treat follow-up as inevitable. The Global Vascular Guidelines provide the clinical scaffold, but these elements should be embedded as standard work rather than left to individual discretion, as illustrated in Figure 2.1
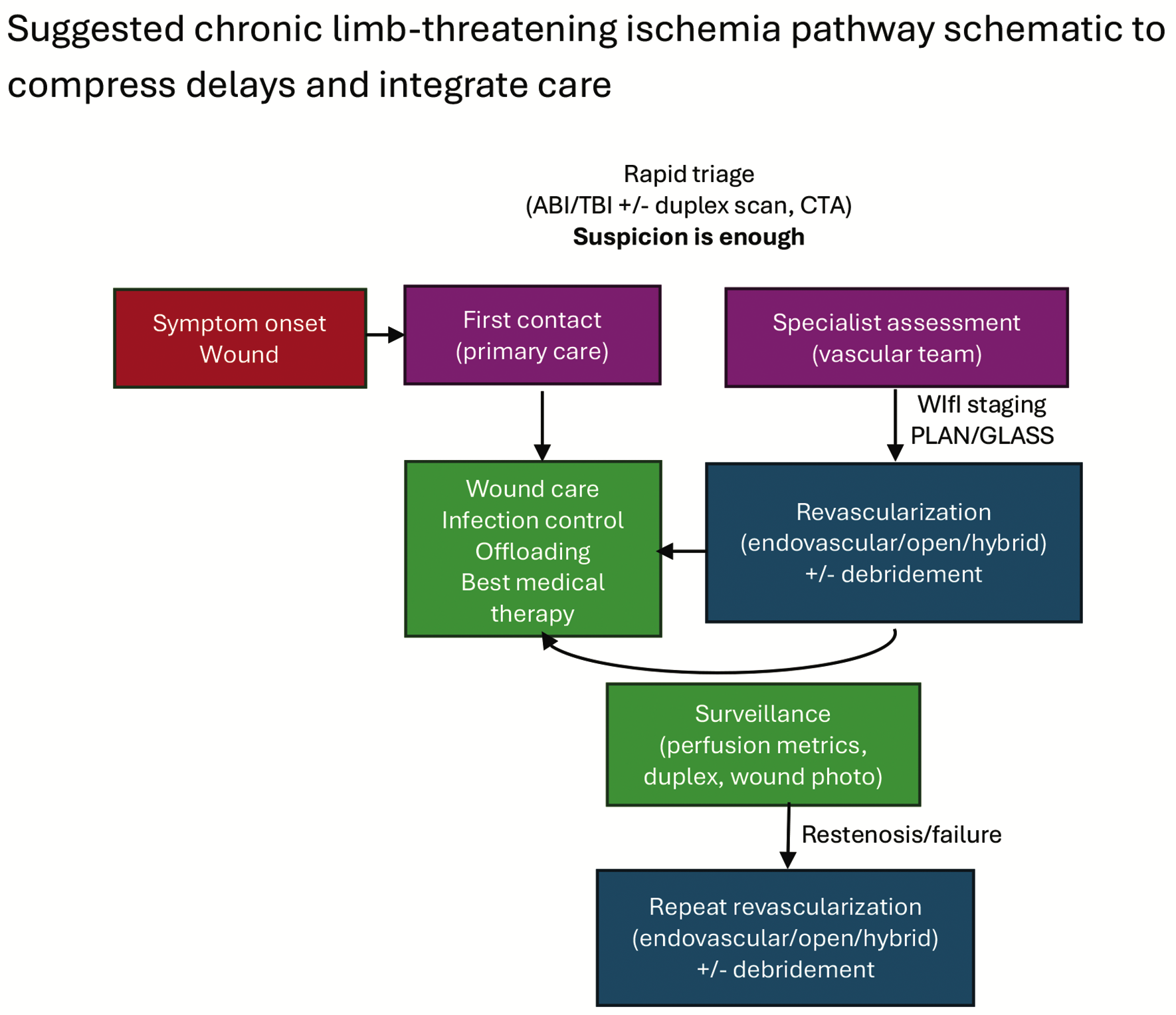
Abbreviations: ABI, ankle-brachial index; TBI, toe-brachial index; CTA, computed tomography angiography; WIfI, Wound, Ischemia, and Foot Infection; PLAN/GLASS, Patient, Limb severity, ANatomic complexity/GLobal Anatomic Staging System.
At the front end, all first-line professionals (general practice, diabetology, community nursing, podiatry, and wound care) should be trained to treat rest pain, tissue loss, and infection in a vasculopathic limb as a vascular emergency, with same-week vascular assessment for suspected CLTI and 72-hour or less assessment when tissue loss or infection is present (Table).2 Single-point referral (telephone plus e-referral) with immediate acknowledgement and feedback to referrers should be implemented so that uncertainty about “who to call” or “how urgent this is” no longer generates avoidable waiting. Where services are regionalized, local access to wound care and scheduled follow-up should be maintained at spoke sites and supported by telemedicine to reduce travel burden and inequity for patients living far from hubs, since centralization without reliable local touchpoints and transfer capacity has been identified as a structural cause of delay.2 Short, consistent patient-facing messages should be embedded across all entry points—“ulcer + pain or infection = vascular emergency”—because missed or delayed recognition is a recurrent driver of late presentation.
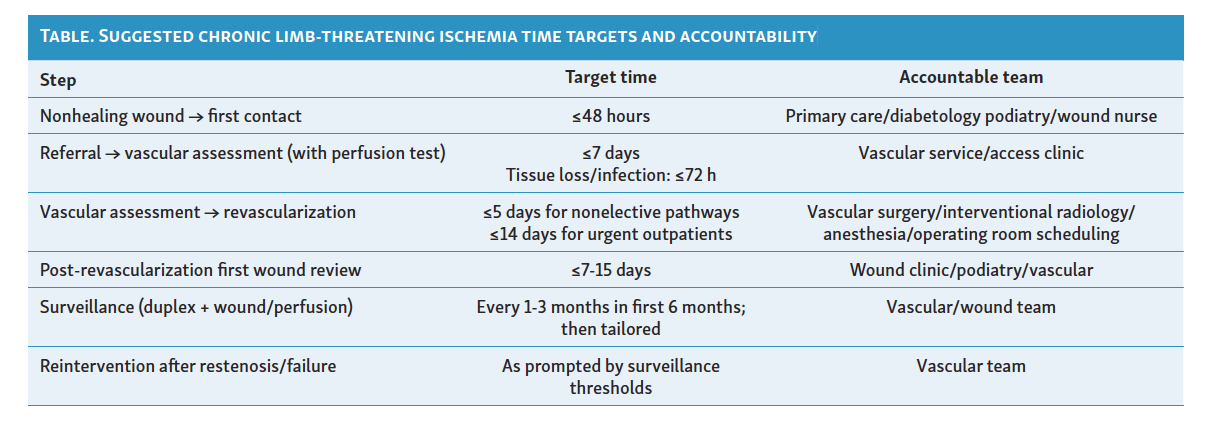
Longitudinal surveillance must be structured, codified, and easy for patients to attend. After revascularization, programs should schedule protocolized follow-up that combines duplex of the treated segment or graft, wound review with standardized photography and documentation, and objective perfusion reassessment, with pre-agreed thresholds that trigger expedited reassessment or re-intervention.1 Surveillance should use shared documentation across inpatient, outpatient, and community caregivers so deterioration is visible to the entire team. Early engagement with wound services is particularly consequential,30 so systems must proactively prevent “no-shows” and rapidly rebook when they occur.
Aligned incentives and protected capacity are required so that doing the right thing is the path of least resistance. Because earlier treatment of appropriate candidates is not only clinically beneficial but cost-effective,4 payers and hospitals should recognize the resource intensity of limb salvage and fund the work accordingly. That means reimbursement that reflects the complexity and duration of multilevel tibial/pedal work in frail, infected patients; protected cath lab/hybrid suite/operating room time for CLTI (not only for elective aneurysm or carotid lists); and financed, nurse-led wound surveillance with same-week hemodynamic testing so patients can actually meet time targets in routine workflow. Where initial revascularization is deferred in carefully selected cases (mostly low WIfI stages), conservative pathways should still adhere to the Society for Vascular Surgery's Objective Performance Goal logic (disciplined surveillance and predefined escalation) because acceptable survival, amputation-free survival, and major amputation rates under wound care alone have been achieved only in programs that supervise closely and escalate promptly when wounds stall.43
In summary, “good” CLTI care should be designed rather than improvised: universal front-end recognition and rapid triage, local access supported by spokes and telemedicine, structured post-revascularization surveillance with shared documentation and explicit triggers, and incentives plus protected capacity that make timely revascularization and follow-up feasible every week—not only on good days.
Limitations and Generalizability
Most proposed time-to-care thresholds are derived from observational studies, registry analyses, and expert consensus rather than randomized controlled trials.2,18,22 As such, these benchmarks should be viewed as system-level guides rather than universal standards.
The feasibility of multidisciplinary programs varies across health systems, particularly in rural or resource-limited settings. Workforce shortages, reimbursement misalignment, logistical constraints, and fragmented electronic records remain major barriers.9,23 Future studies should evaluate system-level interventions across diverse contexts rather than solely focus on individual devices or techniques.
Conclusion
CLTI outcomes are profoundly system dependent. Limbs are lost not only because lesions are complex or revascularization fails but because patients were not recognized early, not triaged rapidly, not revascularized in time, not followed systematically, and not escalated when restenosis or infection reappeared.
The current data tell a consistent story:
- Delays to specialist assessment and to revascularization are common, measurable, and strongly associated with amputation and mortality.
- These delays are worst in the very populations most at risk: older, multimorbid, socioeconomically deprived, diabetic, dialysis-dependent patients with active tissue loss.
- Structured systems—multidisciplinary limb preservation programs, hub-and-spoke referral networks, defined CLTI teams—can compress time to treatment and narrow outcome disparities, even in the highest-risk groups.
CLTI, therefore, is not just a disease that can be managed by a procedure; it should be managed by a well-functioning system. For any vascular program or health system serious about limb salvage, the question is no longer only “Which device?” but “Which pathway, at which speed, under what longitudinal surveillance?” Designing and measuring these pathways should be a core competency of modern vascular programs.
Affiliations and Disclosures
From the Department of Vascular and Endovascular Surgery, CHU de Bordeaux, Université de Bordeaux, France.
The authors report no financial relationships or conflicts of interest regarding the content herein.
Manuscript accepted January 19, 2026.
Address for correspondence: Caroline Caradu, MD, PhD, Department of Vascular and Endovascular Surgery, CHU de Bordeaux, Hôpital Pellegrin, 33000 Bordeaux, France. Email: caroline.caradu@chu-bordeaux.fr
References
1. Conte MS, Bradbury AW, Kolh P, et al; GVG Writing Group for the Joint Guidelines of the Society for Vascular Surgery (SVS); European Society for Vascular Surgery (ESVS); World Federation of Vascular Societies (WFVS). Global vascular guidelines on the management of chronic limb-threatening ischemia. Eur J Vasc Endovasc Surg. 2019;58(1S):S1-S109.e33. doi:10.1016/j.ejvs.2019.05.006
2. Nickinson ATO, Bridgwood B, Houghton JSM, et al. A systematic review investigating the identification, causes, and outcomes of delays in the management of chronic limb-threatening ischemia and diabetic foot ulceration. J Vasc Surg. 2020;71(2):669-681.e2. doi:10.1016/j.jvs.2019.08.229
3. Campbell DB, Gutta G, Sobol CG, et al. How multidisciplinary clinics may mitigate socioeconomic barriers to care for chronic limb-threatening ischemia. J Vasc Surg. 2024;80(4):1226-1237.e2. doi:10.1016/j.jvs.2024.05.033
4. Vadia R, Malyar N, Stargardt T. Cost-utility analysis of early versus delayed endovascular intervention in critical limb-threatening ischemia patients with rest pain. J Vasc Surg. 2023;77(1):299-308.e2. doi:10.1016/j.jvs.2022.07.007
5. Macfarlane RM, Jeffcoate WJ. Factors contributing to the presentation of diabetic foot ulcers. Diabet Med. 1997;14(10):867-870. doi:10.1002/(SICI)1096-9136(199710)14:10<867:AID-DIA475>3.0.CO;2-L
6. Feinglass J, Shively VP, Martin GJ, et al. How ‘preventable’ are lower extremity amputations? A qualitative study of patient perceptions of precipitating factors. Disabil Rehabil. 2012;34(25):2158-2165. doi:10.3109/09638288.2012.677936
7. Pankhurst CJW, Edmonds ME. Barriers to foot care in patients with diabetes as identified by healthcare professionals. Diabet Med. 2018;35(8):1072-1077. doi:10.1111/dme.13653
8. Atkins E, Birmpili P, Kellar I, et al. Understanding delays in chronic limb-threatening ischaemia care: application of the theoretical domains framework to identify factors affecting primary care clinicians’ referral behaviours. J Foot Ankle Res. 2024;17(2):e12015. doi:10.1002/jfa2.12015
9. Atkins E, Kellar I, Birmpili P, et al. Hospital clinicians’ perceptions and experiences of care pathways for chronic limb-threatening ischaemia: a qualitative study. J Foot Ankle Res. 2023;16(1):62. doi:10.1186/s13047-023-00664-6
10. Lecouturier J, Scott J, Rousseau N, Stansby G, Sims A, Allen J. Peripheral arterial disease diagnosis and management in primary care: a qualitative study. BJGP Open. 2019;3(3):bjgpopen19X101659. doi:10.3399/bjgpopen19X101659
11. Normahani P, Mustafa C, Standfield NJ, Duguid C, Fox M, Jaffer U. Management of peripheral arterial disease in diabetes: a national survey of podiatry practice in the United Kingdom. J Foot Ankle Res. 2018;11:29. doi:10.1186/s13047-018-0270-5
12. Mills JL, Beckett WC, Taylor SM. The diabetic foot: consequences of delayed treatment and referral. South Med J. 1991;84(8):970-974.
13. Bailey CMH, Saha S, Magee TR, Galland RB. A 1 year prospective study of management and outcome of patients presenting with critical lower limb ischaemia. Eur J Vasc Endovasc Surg. 2003;25(2):131-134. doi:10.1053/ejvs.2002.1817
14. Sanders AP, Stoeldraaijers LGMC, Pero MWM, Hermkes PJ, Carolina RCA, Elders PJM. Patient and professional delay in the referral trajectory of patients with diabetic foot ulcers. Diabetes Res Clin Pract. 2013;102(2):105-111. doi:10.1016/j.diabres.2013.09.016
15. Mills Sr JL, Conte MS, Armstrong DG, et al. The Society for Vascular Surgery Lower Extremity Threatened Limb Classification System: risk stratification based on wound, ischemia, and foot infection (WIfI). J Vasc Surg. 2014;59(1):220-234.e1-2. doi:10.1016/j.jvs.2013.08.003
16. Noronen K, Saarinen E, Albäck A, Venermo M. Analysis of the elective treatment process for critical limb ischaemia with tissue loss: diabetic patients require rapid revascularisation. Eur J Vasc Endovasc Surg. 2017;53(2):206-213. doi:10.1016/j.ejvs.2016.10.023
17. Manu C, Lacopi E, Bouillet B, et al. Delayed referral of patients with diabetic foot ulcers across Europe: patterns between primary care and specialised units. J Wound Care. 2018;27(3):186-192. doi:10.12968/jowc.2018.27.3.186
18. Li Q, Birmpili P, Johal AS, et al. Delays to revascularization for patients with chronic limb-threatening ischaemia. Br J Surg. 2022;109(8):717-726. doi:10.1093/bjs/znac109
19. Bradbury AW, Moakes CA, Popplewell M, et al; BASIL-2 Investigators. A vein bypass first versus a best endovascular treatment first revascularisation strategy for patients with chronic limb threatening ischaemia who required an infra-popliteal, with or without an additional more proximal infra-inguinal revascularisation procedure to restore limb perfusion (BASIL-2): an open-label, randomised, multicentre, phase 3 trial. Lancet. 2023;401(10390):1798-1809. doi:10.1016/S0140-6736(23)00462-2
20. Bradbury AW, Hall JA, Popplewell MA, et al; BASIL-3 Investigators. Plain versus drug balloon and stenting in severe ischaemia of the leg (BASIL-3): open label, three arm, randomised, multicentre, phase 3 trial. BMJ. 2025;388:e080881. doi:10.1136/bmj-2024-080881.
21. Farber A, Menard MT, Conte MS, et al; BEST-CLI Investigators. Surgery or endovascular therapy for chronic limb-threatening ischemia. N Engl J Med. 2022;387(25):2305-2316. doi:10.1056/NEJMoa2207899
22. Jones DW, Farber A, Armstrong DG, et al. Characteristics of multidisciplinary limb preservation teams and their impact on outcomes in the BEST-CLI trial. J Vasc Surg. 2026;83(2):528-538. doi:10.1016/j.jvs.2025.08.028
23. Hammad TA, Shishehbor MH. Advances in chronic limb-threatening ischemia. Vasc Med. 2021;26(2):126-130. doi:10.1177/1358863X21998436
24. Lane KL, Abusamaan MS, Voss BF, et al. Glycemic control and diabetic foot ulcer outcomes: a systematic review and meta-analysis of observational studies. J Diabetes Complications. 2020;34(10):107638. doi:10.1016/j.jdiacomp.2020.107638
25. Willigendael EM, Teijink JAW, Bartelink M, Peters RJG, Büller HR, Prins MH. Smoking and the patency of lower extremity bypass grafts: a meta-analysis. J Vasc Surg. 2005;42(1):67-74. doi:10.1016/j.jvs.2005.03.024
26. Hirsch AT, Haskal ZJ, Hertzer NR, et al; American Association for Vascular Surgery/Society for Vascular Surgery; Society for Cardiovascular Angiography and Interventions; Society for Vascular Medicine and Biology; Society of Interventional Radiology; ACC/AHA Task Force on Practice Guidelines. ACC/AHA Guidelines for the Management of Patients with Peripheral Arterial Disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Associations for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (writing committee to develop guidelines for the management of patients with peripheral arterial disease)—summary of recommendations. J Vasc Interv Radiol. 2006;17(9):1383-1397; quiz 1398. doi:10.1097/01.RVI.0000240426.53079.46
27. Eikelboom JW, Connolly SJ, Bosch J, et al; COMPASS Investigators. Rivaroxaban with or without aspirin in stable cardiovascular disease. N Engl J Med. 2017;377(14):1319-1930. doi:10.1056/NEJMoa1709118
28. Bonaca MP, Bauersachs RM, Anand SS, et al. Rivaroxaban in peripheral artery disease after revascularization. N Engl J Med. 2020;382(21):1994-2004. doi:10.1056/NEJMoa2000052
29. Stocco F, Dhital K, Laloo R, et al. Benefits of duplex ultrasound surveillance of infrainguinal bypass grafts and institutional costs of graft failure: a retrospective single-center study. Ann Vasc Surg. 2025;113:244-253. doi:10.1016/j.avsg.2025.01.025.
30. Browder SE, Yohann A, Filipowicz TR, et al. Differential impact of missed initial wound clinic visit on 6-month wound healing by race/ethnicity among patients with chronic limb-threatening ischemia. Wound Repair Regen. 2023;31(5):647-654. doi:10.1111/wrr.13116
31. Kim TI, Lee SR, Gorecka J, et al. Intervention to optimize follow-up after lower extremity revascularization. JVS-Vascular Insights: An Open Access Publication from the Society for Vascular Surgery. 2023;1:100005.
32. Causey MW, Ahmed A, Wu B, et al. Society for Vascular Surgery limb stage and patient risk correlate with outcomes in an amputation prevention program. J Vasc Surg. 2016;63(6):1563-1573.e2. doi:10.1016/j.jvs.2016.01.011
33. Awad S, Karkos CD, Serrachino-Inglott F, et al. The impact of diabetes on current revascularisation practice and clinical outcome in patients with critical lower limb ischaemia. Eur J Vasc Endovasc Surg. 2006;32(1):51-59. doi:10.1016/j.ejvs.2005.12.019
34. Freisinger E, Malyar NM, Reinecke H, Lawall H. Impact of diabetes on outcome in critical limb ischemia with tissue loss: a large-scaled routine data analysis. Cardiovasc Diabetol. 2017;16(1):41. doi:10.1186/s12933-017-0524-8
35. Almasri J, Adusumalli J, Asi N, et al. A systematic review and meta-analysis of revascularization outcomes of infrainguinal chronic limb-threatening ischemia. Eur J Vasc Endovasc Surg. 2019;58(1S):S110-S119. doi:10.1016/j.ejvs.2019.04.013
36. Kiwan G, Mohamedali A, Kim T, et al. The impact of clinical follow-up after revascularization on the outcomes of patients with chronic limb threatening ischemia. Ann Vasc Surg. 2022;86:286-294. doi:10.1016/j.avsg.2022.05.043
37. McKenna M, Elghazaly H, Bergman H, et al. Meta-analysis of duplex surveillance following lower limb endovascular intervention. J Endovasc Ther. 2025;32(5):1351-1359. doi:10.1177/15266028231215215
38. Godoy MR, Brochado-Neto FC, Martins Cury MV, et al. Randomized clinical trial of duplex ultrasound surveillance after endovascular therapy for chronic limb-threatening ischemia. J Vasc Surg. 2025;82(6):2181-2191.e2. doi:10.1016/j.jvs.2025.07.043
39. Álvaro-Afonso FJ, García-Álvarez Y, García-Morales EA, et al. Determining the best noninvasive test for peripheral arterial disease diagnosis to predict diabetic foot ulcer healing in patients following endovascular revascularization. Healthcare (Basel). 2024;12(16):1664. doi:10.3390/healthcare12161664
40. Bordonado-Murcia A, Marco-Lledó J, Nieto-Gil P, et al. The prognostic value of transcutaneous oxygen pressure (TcPO2) in diabetic foot ulcer healing: a protocol for a systematic review. Diagnostics (Basel). 2025;15(7):909. doi:10.3390/diagnostics15070909
41. Sun H, Wu Y, Sung L, et al. Use of consecutive transcutaneous oxygen measurement when assessing the need for revascularization and association with the outcomes of ischemic diabetic ulcers. Int Wound J. 2024;21(1):e14635. doi:10.1111/iwj.14635