Deep Vein Arterialization for Acute Limb Ischemia: Initial Experience in Three Patients
Abstract
Background: Deep vein arterialization (DVA) can be an effective strategy for either surgically or minimally invasive non-reconstructible vessels in “no-option” patients with chronic limb-threatening ischemia, but its application in acute limb ischemia (ALI) remains undefined. Methods: The authors present a retrospective case series of 3 patients with ALI who underwent DVA due to persistent, unresolvable limb-threatening tibiopedal occlusive disease after failed endovascular or surgical procedures between March and April 2025. All patients underwent percutaneous DVA using “off-the-shelf” devices. Results: Three patients (mean age 68.3; 2 female, 1 male) underwent DVA. All patients had multiple failed prior revascularizations, including catheter-directed thrombolysis, and underwent DVA within 48 hours of ALI onset. At a median follow-up of 121 days (range 114 to 133 days), 1 patient required reintervention with DVA revision and eventually underwent a below-knee amputation; 1 patient underwent above-knee amputation; and 1 patient required transmetatarsal amputation only. Conclusion: DVA could potentially offer a salvage alternative in patients with ALI with no conventional treatment options, but based on this series with 3 patients, the data remain limited. Further research is required to define optimal patient selection and long-term outcomes.
J CRIT LIMB ISCHEM 2026:6(1):E10-E15. doi: 10.25270/jcli/CLIG-2500008
Key words: lower extremity limb salvage, vein arterialization, chronic limb-threatening ischemia, acute limb ischemia
Introduction
Chronic limb-threatening ischemia (CLTI) represents the most advanced form of peripheral arterial disease, often resulting in nonhealing wounds, rest pain, and a high risk of major amputation. In recent years, deep vein arterialization (DVA) has emerged as an alternative technique to salvage limbs in “no-option” patients with CLTI when all conventional surgical and endovascular revascularization options have been exhausted.
The PROMISE I and PROMISE II trials have demonstrated the safety and efficacy of percutaneous DVA using the LimFlow system, reporting 12-month amputation-free survival rates of 70% and 69%, respectively.1,2 Similar results have been reported using conventionally available (“off-the-shelf”) endovascular tools rather than the proprietary LimFlow system.3 These pivotal studies have established DVA as a viable alternative to amputation in patients with advanced CLTI not amenable to further endovascular or surgical revascularization.
Although venous arterialization has been evaluated in CLTI, the underlying principle of DVA may also be used in the setting of acute limb ischemia (ALI), particularly in patients with no option because of an inability to restore pedal perfusion or identify distal targets for surgical bypass after attempting thrombectomy, thrombolysis, or other acute procedures. However, there are very limited reports of the utilization of DVA in the setting of ALI.4,5 We present procedural and clinical outcomes in 3 patients who underwent DVA for non-reconstructible arterial disease and absent pedal runoff following initial unsuccessful endovascular therapy for ALI.
Methods
This study describes a retrospective case series from a single hospital reviewing 3 patients with ALI who underwent percutaneous DVA between March 18, 2025 and April 9, 2025. All patients were deemed no-option for conventional surgical or endovascular revascularization due to the persistent absence of flow to the foot following prior interventions, including thrombectomy and thrombolytic therapy.
Three patients (2 female, 1 male; mean age 68.3) underwent percutaneous DVA for no-option ALI. Data collected include demographics, comorbidities, procedural details, postoperative management, and clinical outcomes (Table 1).
Procedural description
All patients were treated percutaneously using antegrade or retrograde access to the affected vessels. DVA access was achieved using a combination of wire and catheter techniques.
Donor artery for the DVA was the posterior tibial artery (PTA) in 2 patients and peroneal artery in 1 patient; 1 patient had a prior anterior tibial artery (ATA) DVA that was then revised to the posterior tibial distribution. Patients were followed until either resolution of their clinical ischemia or the occurrence of a major (above-ankle) amputation. Repeat endovascular procedures to maintain DVA patency were performed as needed based upon clinical and duplex ultrasound findings. All patients were treated after DVA with oral antiplatelet and/or anticoagulant medications. Details regarding the location and timing of the DVA are shown in Table 2.
Two patients ended up undergoing major amputation; 1 had above-knee amputation and 1 had below-knee amputation. One patient has maintained limb salvage and an ambulatory foot surface after undergoing a transmetatarsal amputation (TMA).
Case 1
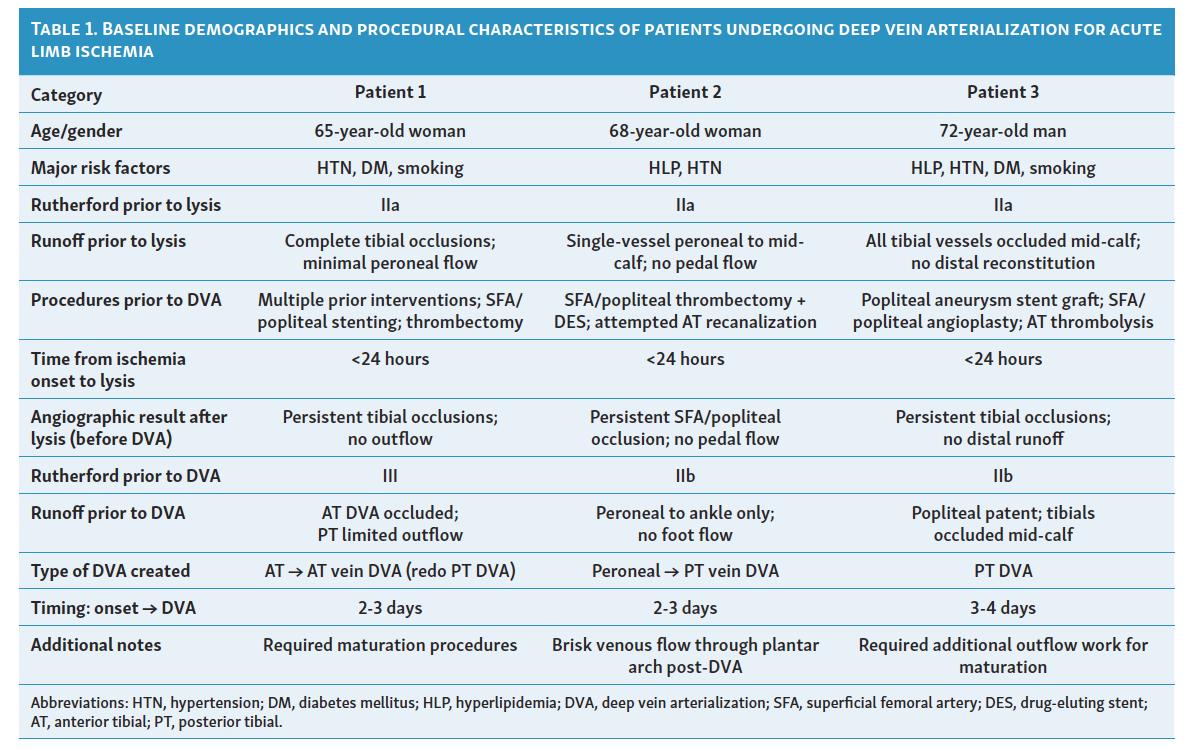
The first patient, a 65-year-old woman with Rutherford class IIb acute-on-chronic limb ischemia (Figure 1), developed abrupt superficial femoral artery (SFA)–popliteal–ATA thrombosis within 1 day of revascularization; DVA was undertaken within 36 hours after overnight catheter-directed thrombolysis failed to restore durable runoff. Baseline anatomy (after thrombolytic therapy) showed chronic total occlusion of the stented SFA, a chronic total occlusion of the PTA with no reconstitution, and single-vessel ATA runoff with severe dorsalis pedis disease and occlusion of the pedal arch.
Before proceeding with venous arterialization, inflow was optimized using femoropopliteal atherectomy, angioplasty, and stent placement across the SFA–popliteal segment. Because the ATA was the only patent vessel supplying the foot, it was selected for the initial DVA creation. Access was obtained via the dorsalis pedis vein. An Outback Elite re-entry catheter (Cordis) was used to cross back into the ATA from the venous system. Balloon angioplasty was performed to establish the arterial–venous connection, and a Viabahn 5 mm x 100 mm stent graft (Gore) was deployed across the anastomosis, followed by additional balloon angioplasty of the ATA–venous loop. While technically successful,
his approach effectively sacrificed the patient’s only remaining arterial runoff, making the limb more vulnerable despite good angiographic results.
Twelve days later, the procedure was revised to a PTA-based DVA in an effort to improve venous arch perfusion and redirect flow to the foot. Angiography at 3 months showed maintained brisk flow, and clinic exams documented strong DVA signals. The patient subsequently underwent TMA due to continued clinical deterioration and absent pedal signals; a later below-knee amputation occurred at an outside hospital, but no further clinical details regarding this secondary event are available.
Case 2
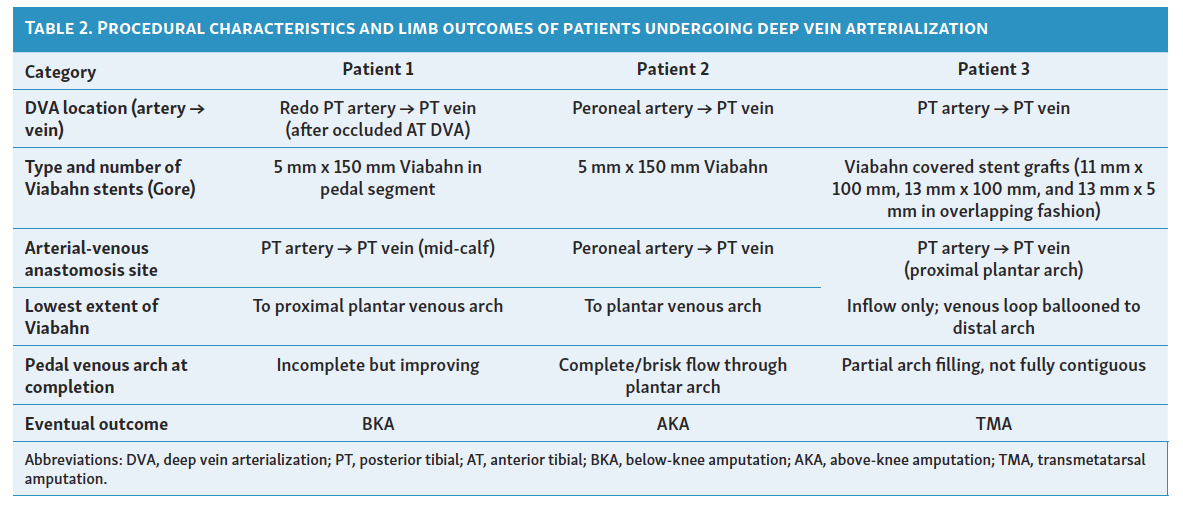
The second patient, a 68-year-old woman (Figure 2), presented with Rutherford class IIa ALI from extensive femoropopliteal and tibial thrombosis, with single-vessel peroneal runoff to the ankle but no perfusion to the foot and occluded ATA and PTA. After an overnight course of catheter-directed thrombolysis, extended with a repositioned infusion, inadequate distal flow remained. Inflow was then optimized with femoropopliteal thrombectomy, angioplasty, and stent relining as well as tibioperoneal trunk angioplasty and peroneal ballooning. Despite these measures, distal runoff remained inadequate, with sluggish peroneal flow and minimal collaterals into the foot. A peroneal-to-posterior tibial vein DVA was then created using a Viabahn 5 mm x 150 mm stent graft across the arteriovenous anastomosis. Completion angiography demonstrated a widely patent, high-flow DVA with brisk opacification of the common/lateral plantar veins and plantar venous arch and strong Doppler signals. Despite these measures, tissue perfusion did not recover and the patient ultimately required an above-knee amputation, which was performed at an outside hospital 3 months after the index procedure; no additional clinical information is available regarding the circumstances leading to this secondary event.
Case 3
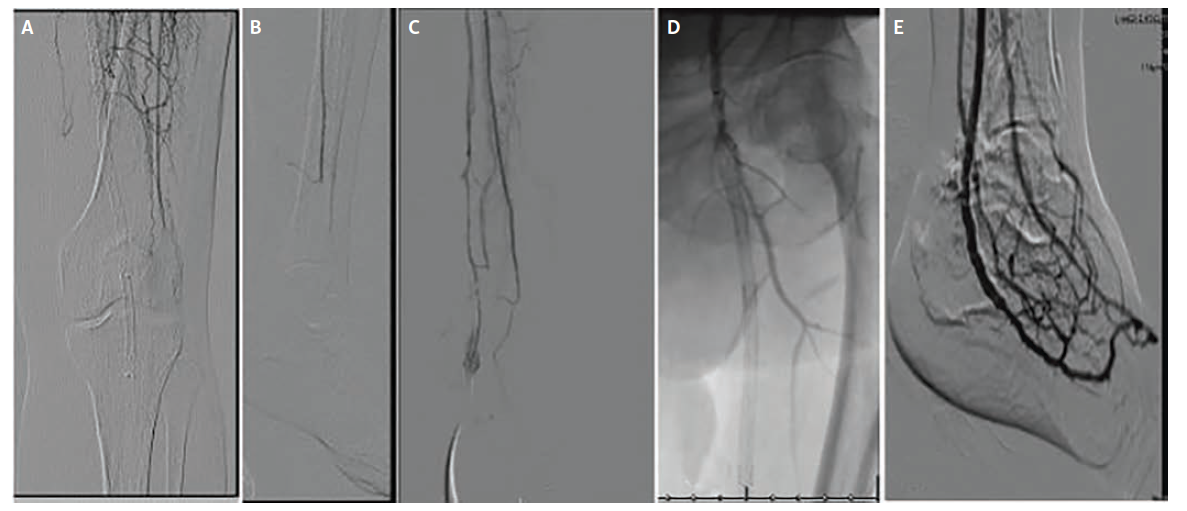
The third patient, a 72-year-old man (Figure 3), presented with Rutherford class IIa ALI manifested by a cold, painful foot and absent pedal signals. Imaging demonstrated thrombosis of a prior popliteal artery aneurysm stent graft with 3-vessel tibial runoff to the ankle but no pedal perfusion.
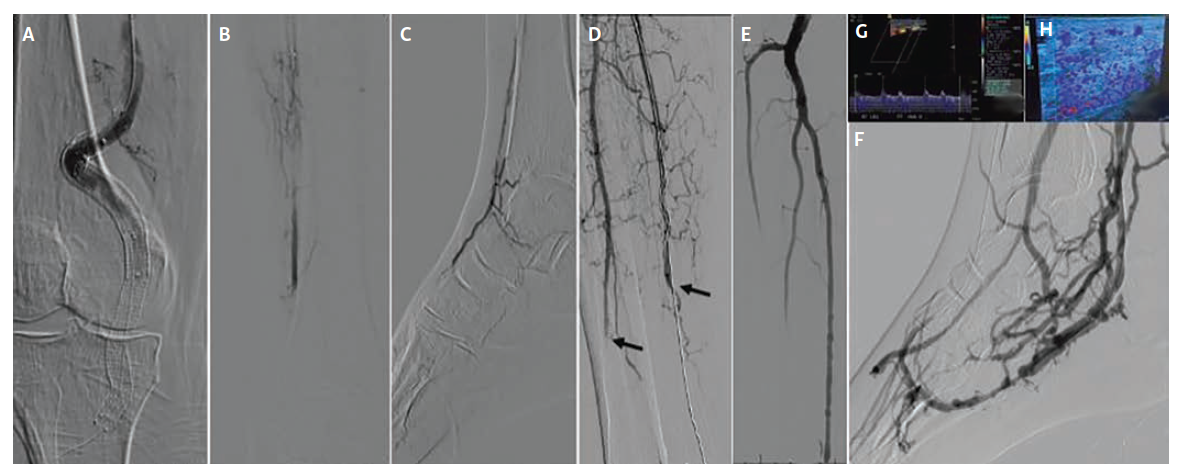
Initial management consisted of catheter-directed overnight thrombolysis aimed at restoring tibial flow. Given sluggish inflow, endovascular optimization was also performed with stent-graft relining of the superficial femoral and popliteal arteries and angioplasty of the tibioperoneal trunk and peroneal artery. Despite these maneuvers, there was no restoration of flow into the foot.
As no suitable distal arterial target was identified, the decision was made to proceed with PTA-based DVA. Using an arterial-venous crossing technique, the PTA was connected to the posterior tibial vein, and Viabahn covered stent grafts (11 mm x 100 mm, 13 mm x 100 mm, and 13 mm x 5 mm in overlapping fashion) were deployed to establish an arterialized venous outflow tract. Completion angiography demonstrated brisk hindfoot venous flow but only partial opacification of the plantar pedal venous loop.
At 1-month follow-up, additional pedal venous arch angioplasty was performed, which improved venous loop filling and forefoot perfusion. Ultimately, the patient required an anticipated TMA 26 days post-DVA, but major amputation was avoided and durable limb perfusion was maintained through the venous circuit.
At 6-month follow-up, Doppler ultrasound demonstrated excellent flow to the foot with sustained patency of the DVA, confirming durable revascularization and long-term effectiveness of the venous arterialization strategy.
Discussion
DVA is increasingly established for the treatment of patients with no-option CLTI, but there are few case reports on the use of DVA in ALI. A successful DVA in the acute setting following an embolic complication after an endovascular procedure was reported by Rodriguez et al.4 Another case described DVA for subacute ischemia (symptoms lasting >14 days and up to 3 months) where conventional techniques, including surgical, endovascular, and medical therapy, were not an option.5 These cases suggest that the underlying principle of DVA may be effective, even in the dynamic setting of ALI.
This case series includes 3 patients who underwent DVA for no-option ALI after failed revascularization, including thrombolytic therapy. All patients would have had inevitable major amputation prior to performing the DVA due to absent pedal flow. Prevention of major amputation was achieved for 1 patient, with the other 2 patients eventually needing above- and below-knee amputations, respectively. Amputation was needed in all cases due to extensive occlusion of the pedal arteries, either from de novo thrombosis or thrombus propagation during thrombolytic therapy, together with clinical deterioration and worsening Rutherford class. Importantly, for the second patient, the subsequent above-knee amputation occurred months later as the result of a separate ischemic event. The initial DVA preserved limb viability during the acute episode and successfully prevented immediate major amputation, indicating that the conduit functioned as intended in the short term.
While there is a strong rationale to avoid performing a DVA using the only arterial outflow to the foot, anatomic constraints often limit donor selection. The decision to use the only visualized tibial runoff vessel is based on the clinical determination that the alternative is inevitable major amputation. In our series, both unsuccessful patients had single-vessel runoff used for DVA, eliminating any residual native arterial flow despite robust collaterals. This likely contributed to poor outcomes despite technically successful procedures. In contrast, the 1 successful patient retained patent tibials to the ankle, permitting preserved native perfusion parallel with the DVA. Prior case reports4,5 similarly document that maintaining at least 2 patent tibial vessels correlates with better outcomes, reinforcing the importance of preserved native arterial flow alongside the DVA conduit.
The rationale to perform DVA in an acute setting remains unknown. Traditionally, open surgical or endovascular strategies are used first in ALI. When initial angiography shows reconstitution of distal limb or pedal flow, primary DVA is not intuitive, since it is usually reserved for patients with no pedal outflow. However, early DVA may be beneficial when there is complete absence of pedal perfusion on initial angiography, or rapid progression to pedal outflow occlusion during catheter-directed therapy. In such cases, DVA may be an option to prevent irreversible tissue loss, even when a patent conduit is ultimately created.
In our series, worsening ischemia and tissue loss despite DVA patency likely reflected profound microcirculatory injury. Even when thrombolysis restored conduit patency or a high-flow DVA was created, irreversible capillary damage rendered restored inflow non-nutritive. Prolonged ischemia produces capillary “no-reflow” through endothelial swelling, leukocyte–platelet plugging, interstitial edema, and compartmental hypertension.7 In such cases, DVA cannot resuscitate necrotic microvascular beds. This mechanism may also explain patients with CLTI who fail DVA, as extensive microvascular occlusion may prevent neovascularization and maturation of the venous arterialization circuit.
Our case series is limited by the small sample size, retrospective design, and short follow-up. Outcomes were also influenced by major confounding factors, including single-vessel runoff, variable collateral circulation, and differing degrees of tissue ischemia at presentation. These limitations highlight that even technically successful DVAs may yield poor clinical outcomes and make it difficult to determine the true therapeutic benefit of DVA. Larger prospective studies with standardized selection criteria are needed to better define the role of DVA in the management of acute and subacute limb ischemia.
Conclusion
This case series adds to the limited evidence supporting the use of percutaneous DVA as a limb-salvage strategy in patients with ALI who have exhausted conventional revascularization options. Although a DVA can be successfully created, its use in the acute ischemic setting remains limited, and outcomes are heavily influenced by the severity of microvascular injury and available native runoff. Nevertheless, in select patients, DVA may delay or prevent an otherwise inevitable major amputation. Continued experience is needed to refine patient selection, optimize procedural technique, and better define the long-term role of DVA in acute ischemia.
Affiliation and Disclosures
Anxhela Hasanlli, MD; John H. Rundback, MD, FAHA, FSVM, FSIR; Kevin Herman, MD; and Edel Mendoza, MD, are from Advanced Interventional & Vascular Services in Teaneck, New Jersey.
The authors report no financial relationships or conflicts of interest regarding the content herein.
Manuscript accepted March 2, 2026.
Address for Correspondence: John Rundback, MD, Advanced Interventional & Vascular Services, 718 Teaneck Rd, Teaneck, NJ 07666. Email: jrundback@aivsllp.com
References
1. Clair DG, Mustapha JA, Shishehbor MH, et al. PROMISE I: Early feasibility study of the LimFlow System for percutaneous deep vein arterialization in no-option chronic limb-threatening ischemia: 12-month results. J Vasc Surg. 2021;74(5):1626-1635. doi: 10.1016/j.jvs.2021.04.057
2. Clair DG. PROMISE II: 12M results from the pivotal trial of the LimFlow system. Presented at: VIVA 23; October 30-November 3, 2023; Las Vegas, NV.
3. Zaman NS, Shackles C, Herman K, Rundback JH. Outcomes of reintervention in percutaneous deep venous arterialization. J Crit Limb Ischem. 2023; 3(4):E119-E126. doi:10.25270/jcli/CLIG23-00016
4. Rodriguez AP, Solano A, Babool SK, Zhang SM, Lavery LA, Siah MC. Deep venous arterialization in the acute setting for embolic complication after endovascular procedure. Ann Vasc Surg - Brief Reports and Innovations. 2025;5(11):100355. doi:10.1016/j.avsurg.2024.100355
5. Chim MSM, How GY, Hong QT, Lo ZJ, Pua U. Case report: Deep vein arterialization for limb salvage in a patient with subacute limb ischemia. Vascular. 2025 Jun;33(3):695-697. doi: 10.1177/17085381241257316
6. Kutsenko O, Sommerset J, Chandra V, Bryce Y. Techniques providing endpoints for revascularization in chronic limb-threatening ischemia. Semin Intervent Radiol. 2023;40(2):177-182. doi:10.1055/s-0043-1768608
7. Blaisdell FW. The pathophysiology of skeletal muscle ischemia and the reperfusion syndrome: a review. Cardiovasc Surg. 2002;10(6):620-630. doi:10.1016/s0967- 2109(02)00070-4















