Bracing for the Unseen: Navigating the Unpredictable in Heart Failure
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Cath Lab Digest or HMP Global, their employees, and affiliates.
Muhammad Imran Hanif, MBBS, FCPS (Cardiology), FSCAI, FRCP (Edin)1;
Minahil Tariq Sheikh, MBBS, FCPS (Cardiology)2
1Head of Cardiology Department, Gulab Devi Teaching Hospital, Lahore, Pakistan; Visiting Interventional Cardiologist, Pakistan Kidney and Liver Institute and Research Institute, Lahore, Pakistan
2Post Graduate Trainee, Gulab Devi Teaching Hospital, Lahore, Pakistan
The authors can be contacted via Dr. Muhammad Imran Hanif at dr.imranhashmi786@gmail.com.
A sinus of Valsalva aneurysm (SVA) is a rare cardiac anomaly characterized by abnormal dilation of the aortic root between the valve annulus and the sinotubular junction, occurring at the level of the elastic lamina.1 With an estimated prevalence of 0.09%, SVA account for up to 3.5% of congenital cardiac defects. Enlargement of the aneurysm can disrupt nearby structures, with symptoms correlating to the chambers involved and the severity of associated valvular regurgitation. Multimodal cardiac imaging plays a critical role in both diagnosis and guiding therapeutic intervention. Procedural correction is generally recommended, with long-term outcomes showing favorable results.
In cases where rupture occurs, a SVA can lead to severe complications, including the formation of a shunt between the sinus of Valsalva and right-sided cardiac chambers. This condition often manifests as a continuous murmur on physical examination and can have high morbidity and mortality if left untreated.
Case Summary
A 55-year-old female presented with acute onset chest pain and dyspnea as well as orthopnea and paroxysmal nocturnal dyspnea for the past 1 day. Previously asymptomatic from a cardiac standpoint and compliant with her medications, she was admitted to the emergency department where she received emergency treatment for acute heart failure.
Her medical history revealed no recent viral infections, COVID-19, or significant family history of heart disease. On examination, her vital signs were as follows: oxygen saturation of 89% on room air, heart rate of 92 beats per minute, blood pressure of 109/72 mmHg, and a temperature of 36.4°C. Physical examination demonstrated an incidental finding of a continuous machinery cardiac murmur best heard at the left parasternal border, loudest in systole (5/6), and 3/6 in diastole with a palpable thrill across the precordium, and a neurological assessment revealed a Glasgow Coma Scale score of 15/15 with normal pupillary responses and brainstem reflexes. Head, neck, respiratory, and abdominal exams were all unremarkable. The baseline electrocardiogram was unremarkable, and her cardiac enzymes were negative.
Based on the diagnosis of acute heart failure, the patient was admitted and her treatment was optimized, with an echocardiogram following, which showed:
• Annular dilatation of the mitral valve with anterior mitral leaflet prolapse;
• Eccentric jet of mitral regurgitation;
• Sclerotic aortic valve with grade II calcification;
• Pressure gradient = 22 mmHg, annulus = 18 mm, sinus of Valsalva = 23 mm;
• Rupture of sinus of Valsalva communicating with the right atrium with continuous flow on continuous wave Doppler.
Device closure utilizing an Amplatzer Duct Occluder (ADO) single disc device (Abbott) was planned. The patient shifted to the cath lab after undergoing informed consent.
Procedure
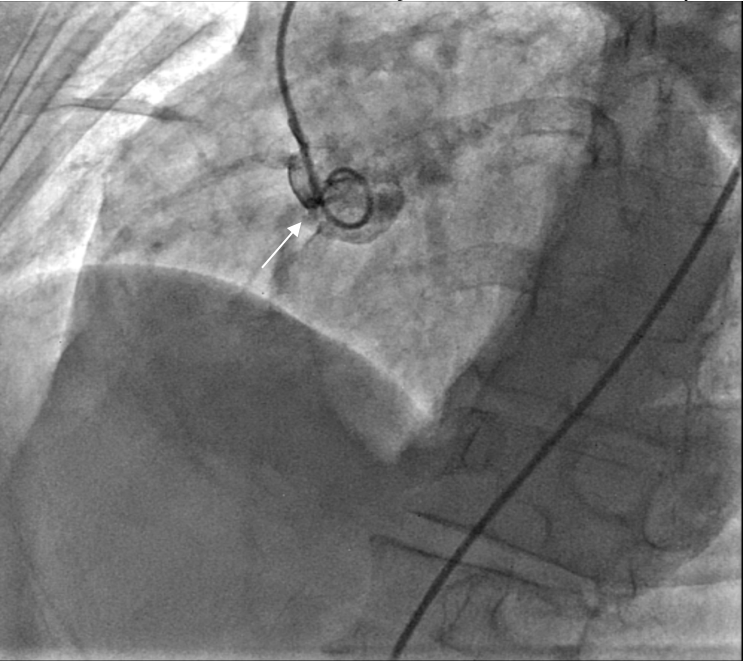
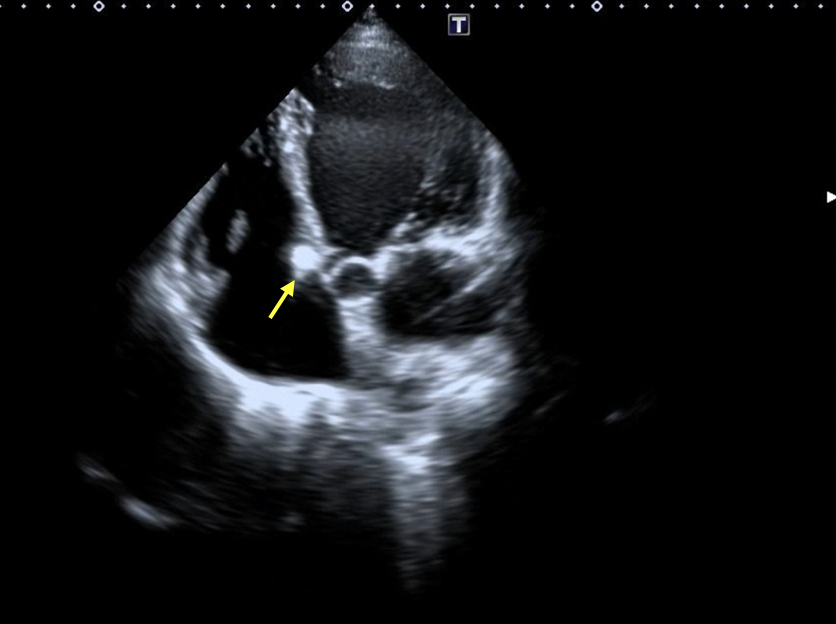
Ultrasound-guided dual access was completed, with a right femoral arterial as well as a venous approach. An aortogram was performed using the arterial approach, following introduction of a pigtail catheter at the level of the non-coronary aortic sinus, which showed a connection between the right atrium and the non-coronary aortic sinus (Figure 1, Video 1).
Figure 1. An aortogram revealing a communication between the right atrium and the non-coronary sinus of Valsalva (white arrow).
Video 1.
Sizing of the lesion was conducted under fluoroscopy and by using periprocedural echo, and determined to be 4 mm. A multipurpose catheter and Glidewire (Terumo Interventional Systems) was used to cross the defect. Another multipurpose catheter was introduced via the venous approach, through which the antegrade wire was snared in the pulmonary artery. The wire was then externalized, leading to the formation of an arteriovenous rail (Figures 2A-B, Videos 2A-B).
Figure 2A. Snaring of the wire.
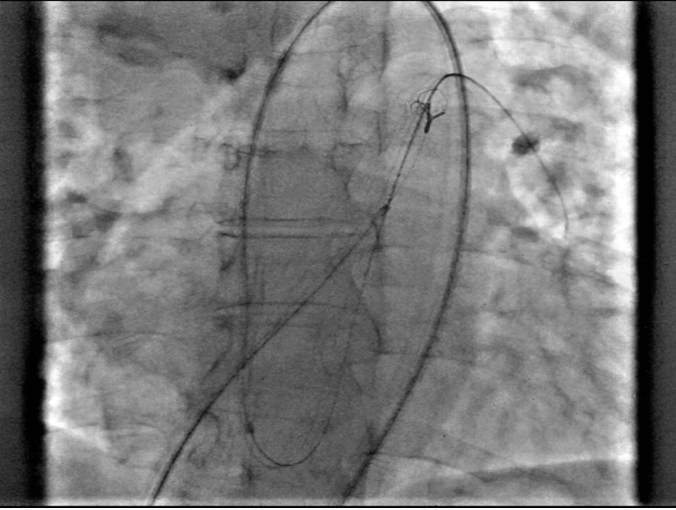
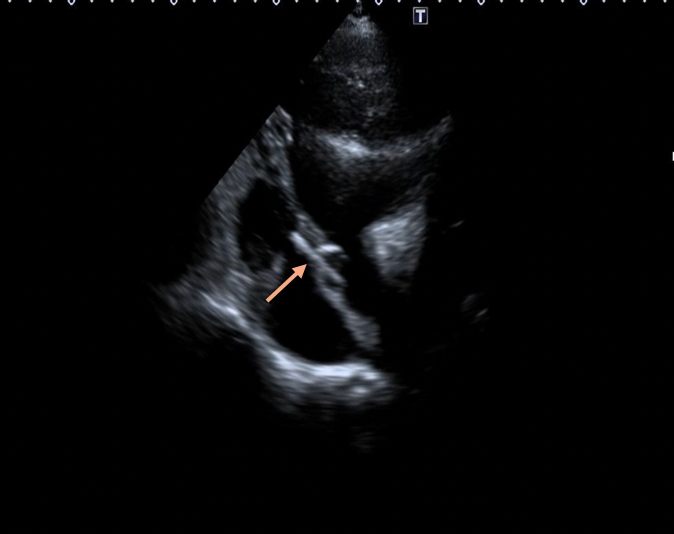
Figure 2B. Catheter advanced across the defect (orange arrow) on transthoracic echocardiography.
Videos 2A-B.
(Video 2B)
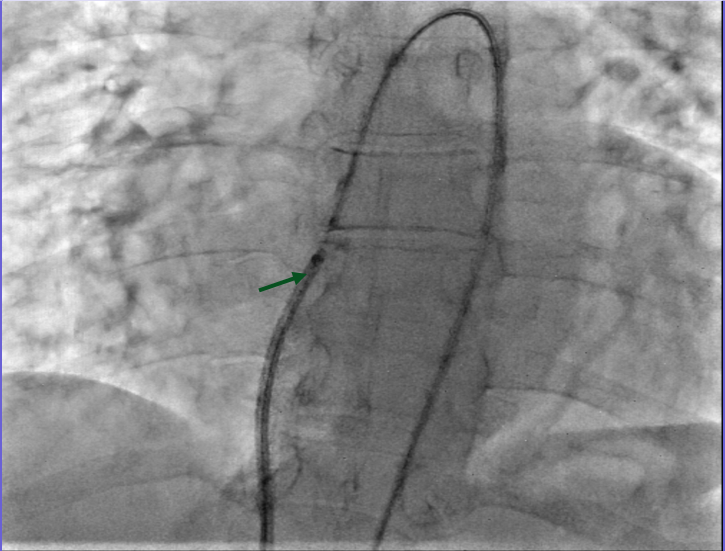
A 7 French delivery sheath was traversed across the ruptured sinus, through the inferior vena cava, to the aorta (Figure 3, Video 3).
Figure 3. Advancement of a 7 French (F) sheath (green arrow) across the arteriovenous (AV) rail.
Video 3.
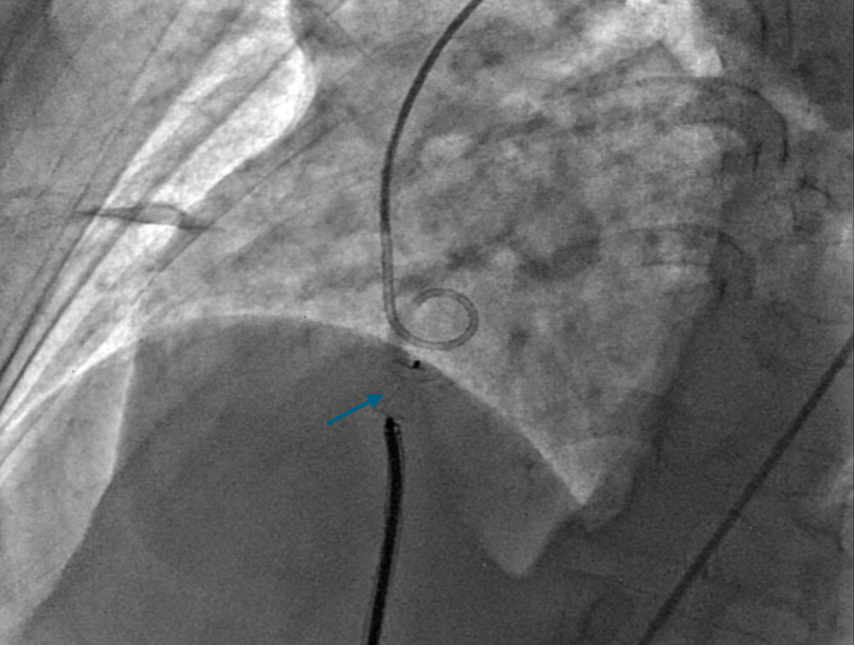
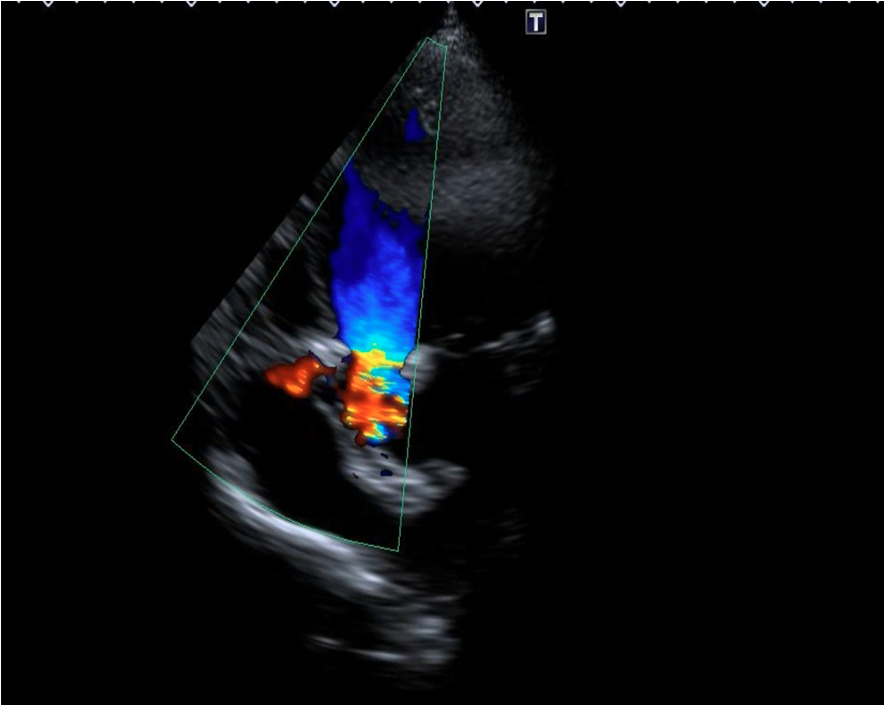
We chose an appropriately sized occluder, which was 20% greater than the entrance diameter of the ruptured SVA. The selected occluder with its attached delivery cable was inserted through the delivery sheath from the venous route, and its aortic disc was deployed in the ascending aorta. Positioning of the device (an ADO single-disc device of 6-8 mm) was then confirmed under transthoracic echocardiography (Figures 4A-C, Videos 4A-C).
Figure 4. (A) Positioning of the Amplatzer Duct Occluder (ADO) device (Abbott) (blue arrow). (B) Transthoracic echocardiography. (C) No residual shunt observed across the defect following device placement on transthoracic echocardiography.
(Figure 4A)
(Figure 4B)
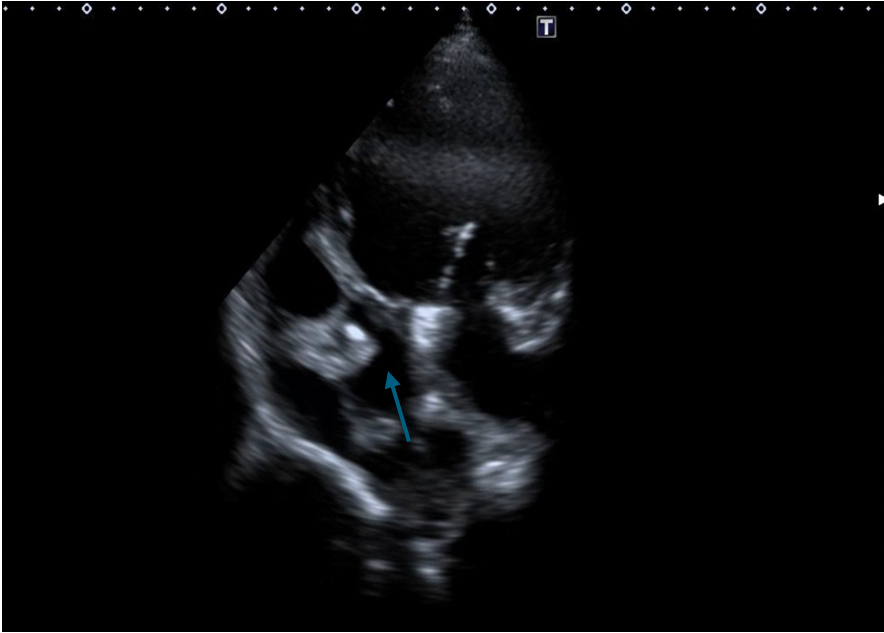
(Figure 4C)
Videos 4A-C.
(Video 4B)
(Video 4C)
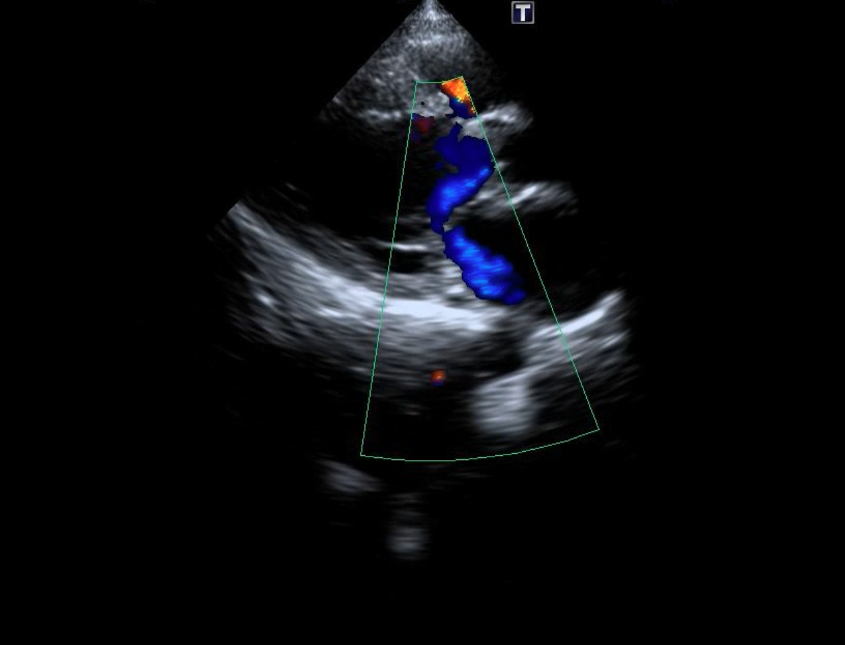
Gentle traction was exerted on the delivery cable to confirm seating of the left disc on the aortic side without slippage into the ruptured SVA. The rest of the occluder was deployed on the right side across the lesion (Figure 5A-B, Videos 5A-B).
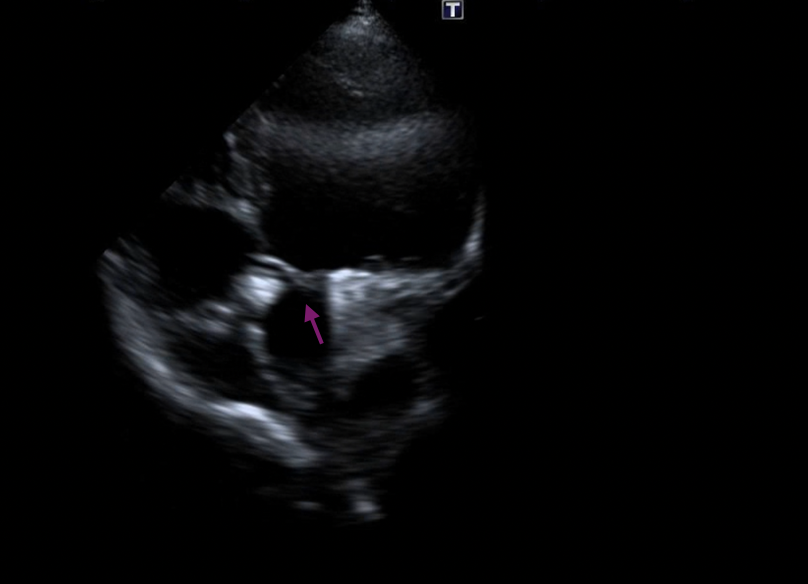
Figure 5. (A) Device deployed (purple arrow). (B) Transthoracic echocardiography.
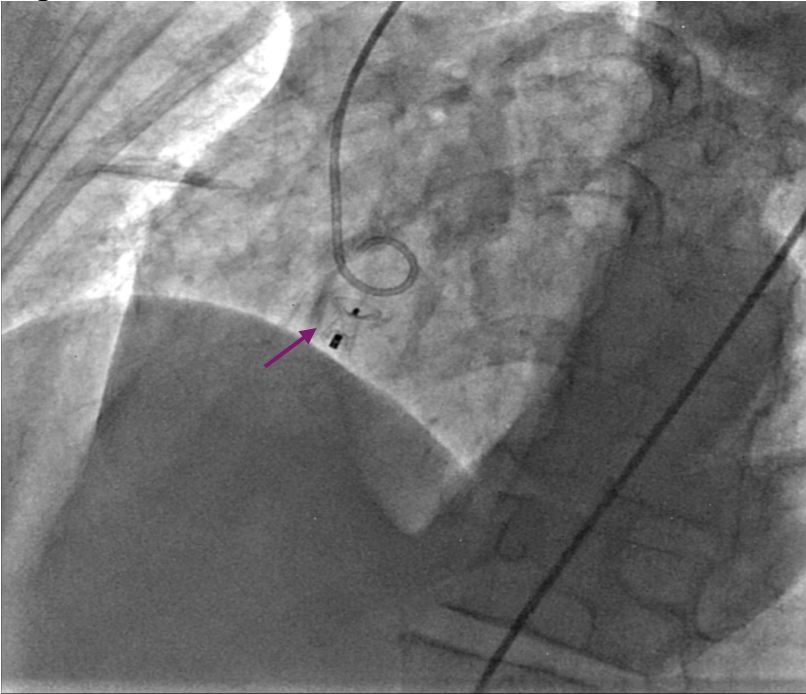
(Figure 5B)
Videos 5A-B.
(Video 5B)
The device was released after making certain that there was no significant aortic regurgitation, tricuspid regurgitation, or encroachment on coronary arteries (Figure 6A-B, Videos 6A-B and Figure 7A-B, Videos 7A-B).
Figure 6. (A-B) Check injections demonstrating a well-seated device with no encroachment on the coronary arteries and absence of tricuspid regurgitation.
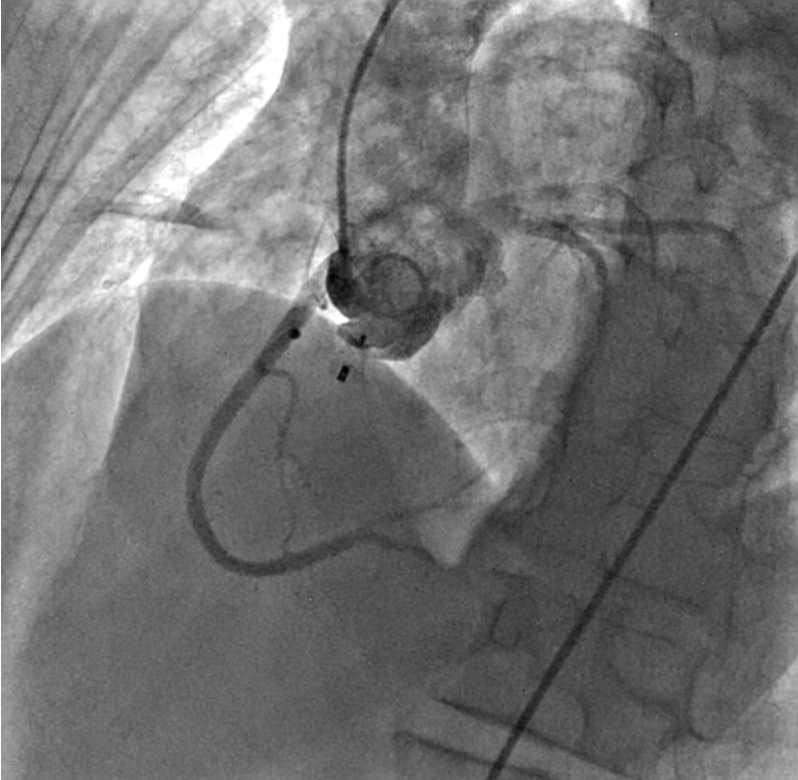
(Figure 6B)
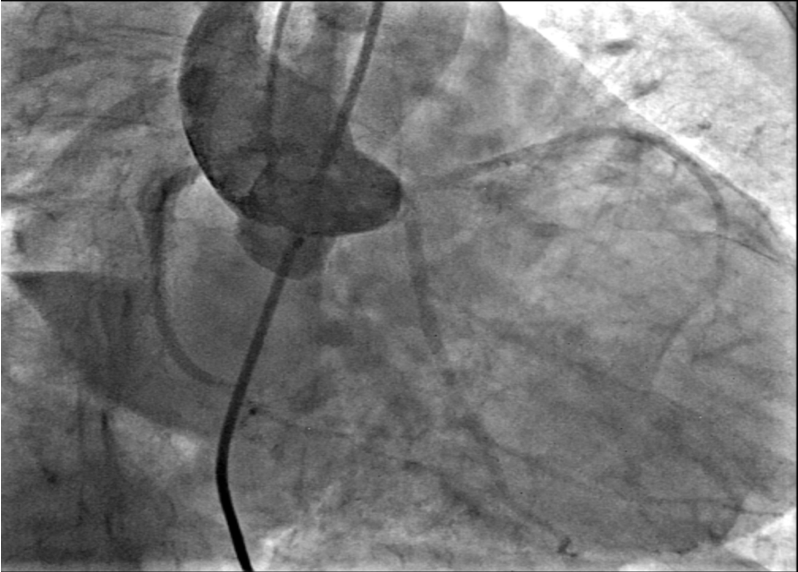
Videos 6A-B.
(Video 6B)
Figure 7. (A-B) After device placement, no progression of tricuspid regurgitation was noted on transthoracic echocardiography.
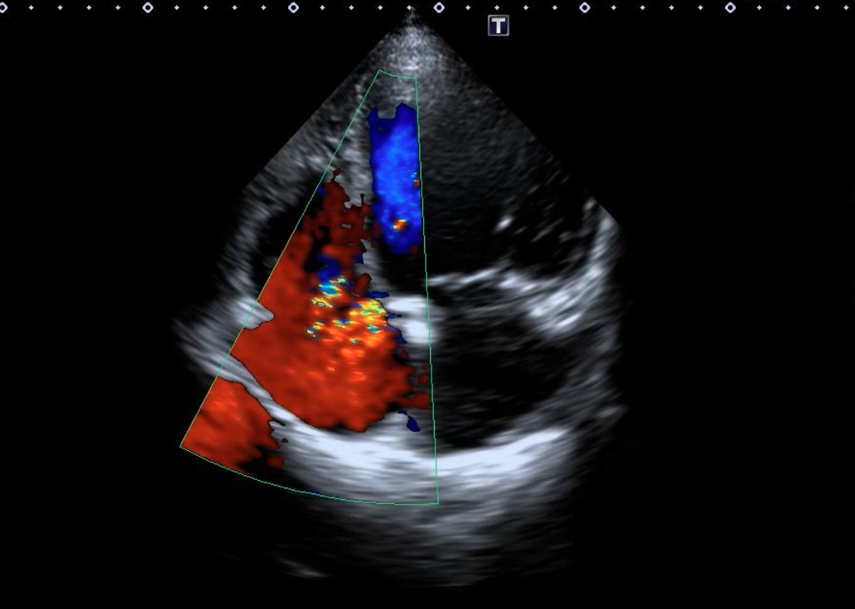
(Figure 7B)
Videos 7A-B.
(Video 7B)
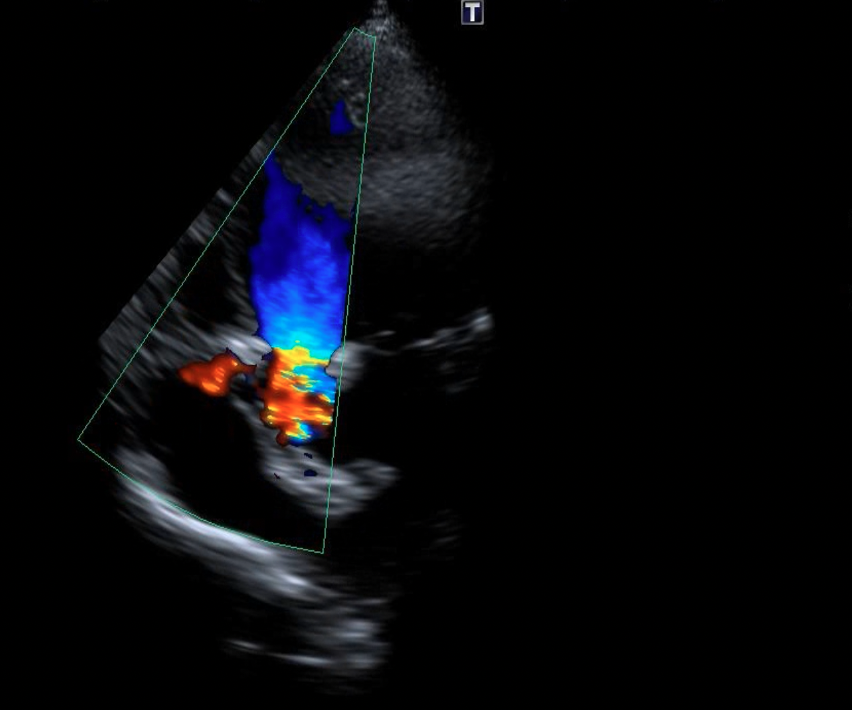
The patient was then shifted to the intensive care unit, kept on aspirin, and discharged after 2 days. She returned for follow-up at 1 month post procedure and remains in follow-up with no residual shunt visible on repeat echocardiography (Figure 8A-B, Videos 8A-B).
Figure 8. (A-B) Follow-up transthoracic echocardiography demonstrated satisfactory device seating (yellow arrow), and stable mitral and tricuspid valve function without increased regurgitation.
(Figure 8B)
Videos 8A-B.
(Video 8B)
Discussion
A sinus of Valsalva aneurysm (SVA) is a rare congenital heart defect, representing less than 1% of all congenital heart diseases. It typically presents as a blind pouch extending from one of the aortic sinuses into the right atrium or ventricle, resulting from a congenital weakness at the junction between the aortic media and annulus fibrosus.1 Approximately 20% of SVA remain unruptured.
Although it is predominantly a congenital condition, a SVA can also be associated with conditions such as infective endocarditis, atherosclerosis, trauma, syphilis, aortic dissection, ulcerative colitis, Behçet’s disease, and Marfan syndrome. The condition exhibits a male predominance, with a male-to-female ratio of 4:1, and symptoms typically develop in adulthood,2-4 with patients most commonly between 30 and 45 years of age.5 Approximately 30-40% of patients may present with coexisting cardiac anomalies, most commonly ventricular septal defects or aortic valve regurgitation. Clinical symptoms manifest in 80% of patients, which include shortness of breath, chest pain and easy fatigability.6 The severity of symptoms depends on factors such as the size of the shunt, the presence of associated cardiac defects, and age at presentation.5 Sakakibara and Konno proposed a classification system to delineate the anatomic origin and protrusion sites of SVA, commonly referred to as the Sakakibara classification.7 This system can be succinctly summarized into five types:
- Type I: Rupture into the right ventricle immediately beneath the pulmonary valve.
- Type II: Rupture into or just below the crista supraventricularis of the right ventricle.
- Type III: Rupture into the right atrium or right ventricle at or near the tricuspid valve annulus.
- Type IV: Rupture directly into the right atrium.
- Type V: Rare or atypical presentations, including rupture into the left ventricle, left atrium, pericardial cavity, or pulmonary artery.
Aneurysms most frequently involve the right coronary sinus (70%), followed by the noncoronary sinus (29%), and rarely, the left coronary sinus (1%).8 Rupture of right coronary sinus aneurysms often occurs into the right ventricle or atrium, while noncoronary sinus aneurysms tend to rupture into the right atrium. In rare instances, rupture can occur into the pulmonary artery, left ventricle, left atrium, or pericardial cavity.
Rupture typically presents as a sudden onset of chest pain and acute heart failure. If left untreated, death often occurs within one year. Transesophageal echocardiography is regarded as the gold standard for diagnosing SVA due to its superior sensitivity and accuracy.
Surgical intervention remains the primary treatment for ruptured SVA, but percutaneous closure is emerging as new treatment modality, especially in patients at high risk for surgical closure.9 Different devices used for closing this defect include Gianturco coils (Cook Medical), Amplatzer duct occluders, and Amplatzer atrial septal occluders (Abbott).9,10
Following a comprehensive review and appraisal of 1,017 relevant articles identified across three databases, Ayati et al11 found 94 studies involving 407 patients who underwent transcatheter closure of a ruptured sinus of Valsalva. Males represented 65% of patients, with an average age of 34.5 years. The overall success rate of transcatheter closure was 95.6%. Complications occurred in 49 patients (12%), with the most notable being sustained residual shunts in 7 patients (1.7%), significant new or worsening aortic insufficiency in 6 patients (1.5%), and ruptured sinus of Valsalva recurrence in 6 patients (1.5%). Post-procedural mortality was reported in only 2 patients (0.5%).
The case reported herein is notable due to the patient's underlying cardiomyopathy and heart failure, which did not respond to emergency interventions despite receiving fully optimized heart failure treatment. The patient's condition was complicated by an acute rupture of a Sakakibara Type IV sinus of Valsalva aneurysm. Additionally, the patient, a woman in her mid-50s, presented outside the typical age range for this condition and had no prior history of dyspnea or other cardiac issues. Uniquely, she underwent device closure during the acute phase of the presentation, rather than waiting for clinical stabilization over several days before planning the intervention.
The device was implanted through the following procedure:
1. An aortogram was performed to confirm the presence and characteristics of the ruptured SVA and to assess the severity of associated aortic regurgitation.
2. Additionally, a left ventricular angiogram was conducted to exclude the presence of a ventricular septal defect.
3. The ruptured SVA was accessed from the aortic side using a multipurpose catheter and a Glidewire.
4. The wire was carefully advanced into the aorta through the inferior vena cava and subsequently snared via the right femoral vein using a multi-loop snare, thereby establishing an arteriovenous loop.
5. A 7 French delivery sheath was introduced through the venous access site and advanced over the wire across the ruptured SVA.
6. The occlusion device was then loaded into the delivery sheath.
7. The aortic retention disc was first deployed in the ascending aorta, and the system was gently retracted to ensure secure anchorage at the aortic end of the ruptured SVA.
8. Subsequently, the distal portion of the device was deployed by stabilizing the delivery cable and withdrawing the sheath.
9. An angiogram was performed to verify the optimal positioning of the device. The entire procedure was guided by fluoroscopy and contrast-enhanced transthoracic echocardiography.
10. Upon satisfactory deployment and positioning, the device was released.
11. Ten minutes post-procedure, a follow-up aortogram was performed to assess for any residual shunt and to re-evaluate the degree of aortic regurgitation.
After the procedure, the patient was transferred to the intensive care unit for close monitoring. Her symptoms improved, and she achieved clinical stability within two days, after which she was discharged. The patient remains under regular follow-up, with repeat echocardiograms during her visits confirming that the device is securely in place with no evidence of residual shunt.
References
1. Prian GW, Diethrich EB. Sinus of Valsalva abnormalities. A specific differentiation between aneurysms of an aneurysms involving the sinuses of Valsalva. Vasc Surg. 1973 May-Jun;7(3): 155-164. doi:10.1177/153857447300700304
2. Babacan KM, Tasdemir O, Zengin M, et al. Fistulous communication of aortic sinuses into the cardiac chambers. Fifteen years surgical experience and a report of 23 patients. Jpn Heart J. 1986 Nov; 27(6): 865-870. doi:10.1536/ihj.27.865
3. Taguchi K, Sasaki N, Matsuura Y, Uemura R. Surgical correction of aneurysm of the sinus of Valsalva. A report of forty-five consecutive patients including eight with total replacement of the aortic valve. Am J Cardiol. 1969 Feb; 23(2): 180-191. doi: 10.1016/0002-9149(69)90065-4
4. Chu SH, Hung CR, How SS, et al. Ruptured aneurysms of the sinus of Valsalva in Oriental patients. J Thorac Cardiovasc Surg. 1990 Feb; 99(2): 288-298.
5. Moustafa S, Mookadam F, Cooper L, et al. Sinus of Valsalva aneurysms--47 years of a single center experience and systematic overview of published reports. Am J Cardiol. 2007 Apr 15; 99(8): 1159-1164. doi:10.1016/j.amjcard.2006.11.047
6. Reichert CL. Ruptured sinus Valsalva aneurysm, a rare cause of heart failure. Neth Heart J. 2008 Feb; 16(2): 60-61. doi:10.1007/BF03086120
7. Sakakibara S, Konno S. Congenital aneurysm of the sinus of Valsalva. Anatomy and classification. Am Heart J. 1962 Mar; 63: 405-424. doi:10.1016/0002-8703(62)90287-9
8. Takach TJ, Reul GJ, Duncan JM, et al. Sinus of Valsalva aneurysm or fistula: management and outcome. Ann Thorac Surg. 1999 Nov; 68(5): 1573-1577. doi:10.1016/s0003-4975(99)01045-0
9. Trehan VK, Mukhopadhyay S, UmaMahesh CR, et al. Successful transcatheter closure of ruptured sinus of Valsalva aneurysm. Indian Heart J. 2002 Nov-Dec; 54(6): 720-722.
10. Chang CW, Chiu SN, Wu ET, et al. Transcatheter closure of a ruptured sinus of valsalva aneurysm. Circ J. 2006 Aug; 70(8): 1043-1047. doi:10.1253/circj.70.1043
11. Ayati A, Toofaninejad N, Hosseinsabet A, et al. Transcatheter closure of a ruptured sinus of valsalva: a systematic review of the literature. Front Cardiovasc Med. 2023 Aug 25; 10: 1227761. doi:10.3389/fcvm.2023.1227761
Read more from Dr. Hanif and colleagues:
CASE REPORT
Muhammad Imran Hanif, MBBS, FCPS (Cardiology), FSCAI, FRCP; Muhammad Sikandar Saleem, MBBS, FCPS (Cardiology); Muhammad Ansar Maqsood, MBBS, FCPS (Cardiology)
Find More:
Renal Denervation Topic Center
Cardiovascular Ambulatory Surgery Centers (ASCs) Topic Center
Grand Rounds With Morton Kern, MD
Peripheral Artery Disease Topic Center












