Preserving Limbs and Lives: The Role of Cellular, Acellular, and Matrix-like Products in Diabetic Foot Ulcer Care
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Diabetic foot ulcers (DFUs) significantly affect patients and health care systems, often resulting in amputation and high morbidity. Objective. To evaluate whether cellular, acellular, and matrix-like products (CAMPs) are more effective than the standard of care in reducing lower limb amputation (LLA) risk and improving survival in DFU patients. Methods. This retrospective cohort study used a proprietary database to identify patients with type 1 or type 2 diabetes and foot ulcers. Two cohorts were compared: those receiving debridement and those receiving CAMPs. Propensity score matching (PSM) balanced baseline characteristics. The primary outcome was 5-year LLA risk and amputation-free survival. Results. Before PSM, the CAMPs cohort (n = 2273) had higher rates of comorbidities compared with the debridement-only cohort (n = 31,050). After matching, cohorts were well-balanced (n = 2272 each). Treatment with CAMPs significantly reduced the risk of LLA compared with debridement alone, with a 4.9% absolute risk reduction (95% CI, -7.4 to −2.4%; P < .0001) and a 24% relative risk reduction (risk ratio, 0.763; 95% CI, 0.664-0.876). Kaplan-Meier analysis demonstrated improved 5-year amputation-free survival in the CAMPs cohort (75.7% vs 71.3. Conclusion. CAMP therapy significantly reduces LLA risk and improves amputation-free survival in DFU patients. These findings underscore the potential of CAMPs as a critical component of advanced DFU care, warranting broader clinical adoption and further investigation.
Diabetic foot ulcers (DFUs) are a prevalent and serious complication of diabetes mellitus (DM), often resulting in infection, gangrene, and lower limb amputation (LLA). In the United States, approximately 38.4 million people are living with DM.1 This population has a 19% to 34% lifetime risk of developing a DFU.2 Among individuals with a DFU, approximately 21% ultimately undergo some level of LLA, based on longitudinal Medicare data.3 Unfortunately, after an amputation, outcomes are extremely poor. Mortality rates after a minor amputation range from 54% to 79% and may exceed 90% after a major amputation.2 In this regard, DFUs contribute significantly to health care costs, morbidity, and mortality.4,5 Given the serious outcomes associated with DFUs, optimizing treatment is critical.
DFU treatment begins with the standard of care, which may consist of debridement, infection management, off-loading, maintaining a moist wound environment, and/or vascular assessment and revascularization.6,7,8 Even with standard-of-care wound management, pooled evidence from randomized controlled trials show that only about 33.15% of DFUs achieve complete healing within 12 to 24 weeks, leaving majority at risk for ongoing complications.9 Less than 50% wound area reduction after 4 weeks of appropriate treatment is a sign that escalation of care to advanced therapies should be considered.10,11 These limitations highlight the need for adjunctive approaches beyond standard care, such as the use of cellular, acellular, and matrix-like products (CAMPs).
CAMPs are traditionally used in wound repair and regeneration of soft tissue in wound management.8,9,12 CAMPs were previously referred to as cellular and/or tissue-based products (CTPs). The adoption of new nomenclature emphasizes the expanding diversity of biomaterials and bioengineered products now available.12 CAMPs can be categorized into 3 main types: cellular, acellular, and matrix-like products. Cellular and acellular products are then divided into autograft, allograft, and xenograft subcategories, and matrix-like products can be divided into natural or synthetic subcategories. Cellular products can also be further divided by whether the cells are viable or nonviable; viability is independent of whether those cells are differentiated or nondifferentiated.12 Currently, over 80 different CAMPs are available. CAMPs are thought to exert their biological effects through mechanisms such as intracellular signaling, intercellular communication, and the formation of structural scaffolds for new tissue growth. While individual studies have reported accelerated wound closure with CAMPs,13 comprehensive data on long-term clinical outcomes such as limb salvage and amputation-free survival remain scarce. Therefore, the present study specifically focuses on CAMPs as the advanced therapy, comparing their effectiveness to standard of care represented by debridement.
This study aims to evaluate the comparative effectiveness of CAMPs vs debridement in preventing long-term complications associated with DFUs, specifically focusing on LLA and amputation-free survival rates.
Methods
Data source
TriNetX (Cambridge, MA), a global federated health research network, was utilized for this study. The platform aggregates de-identified electronic health record data from participating health care organizations (HCOs) across the United States, providing access to diagnoses, procedures, medications, and laboratory values. HCOs choose to join the network and contribute data through institutional subscription agreements that allow them access to the platform. The database encompasses millions of patients, offering a robust resource for clinical research. It also queries patient data up to 20 years before the time of the analysis. The database contains data on over 250 million patients across over 120 HCOs. At the time of the data extraction for this investigation on May 4, 2025, the platform’s US Collaborative Network contained aggregate data for 114 312 208 patients across 55 HCOs.
The database complies with the Health Insurance Portability and Accountability Act (HIPAA). All data displayed on the platform or provided in datasets are de-identified per Section 164.514(a) of the HIPAA Privacy Rule.14 This study was exempt from institutional review board approval because the database only contains de-identified data.
Study population and cohort definitions
Patients with type 1 DM or type 2 DM DFUs were identified in the database using International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes E10.621 or E11.621, in combination with non-pressure chronic ulcer codes for the heel, midfoot, or other parts of the foot (L97.4 or L97.5) (Figure). Patients were included based on the first recorded instance of these combined diagnoses and were subsequently evaluated for treatment received within a defined postdiagnosis window.
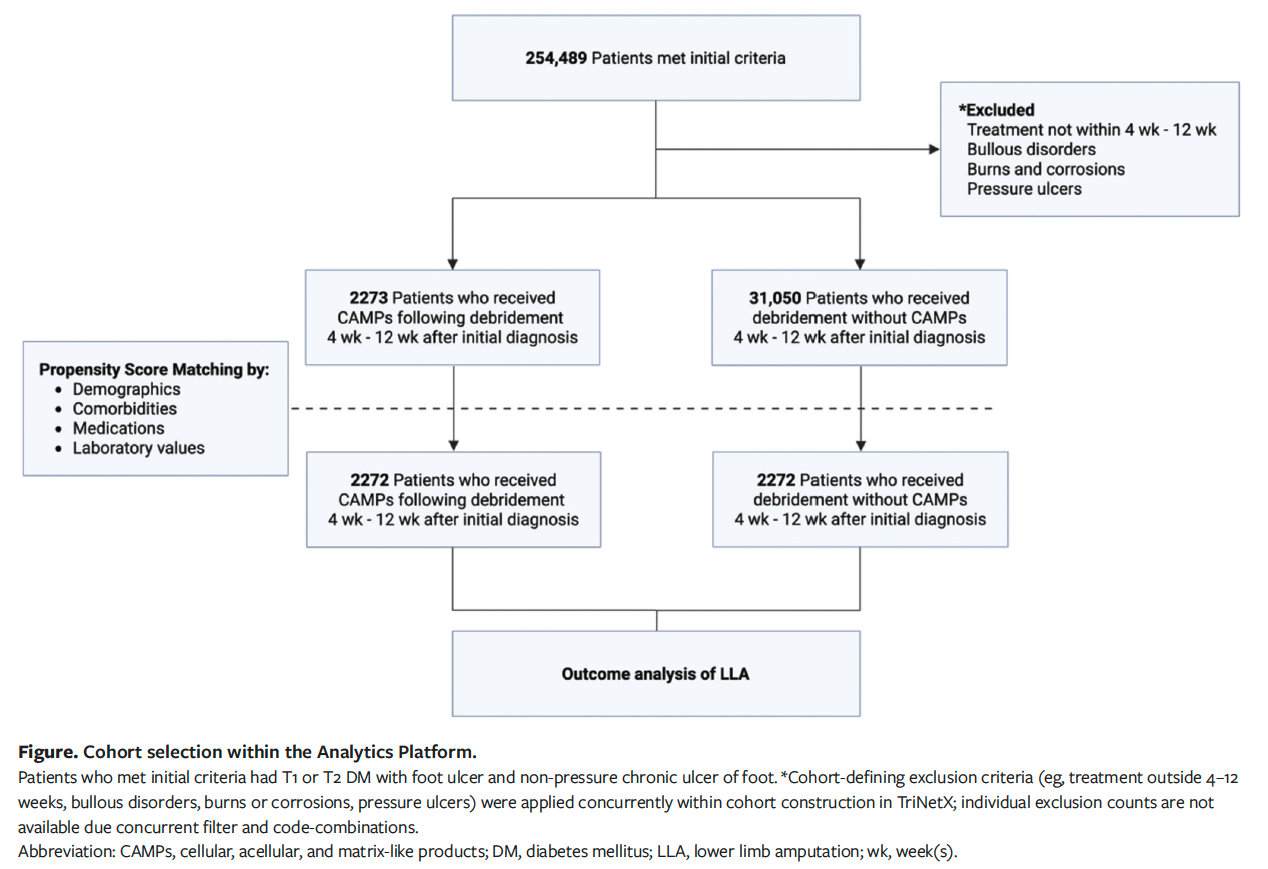
To reflect clinical guidelines recommending escalation to advanced therapies when standard care does not achieve adequate wound healing within 4 weeks, the treatment window was restricted to 4 to 12 weeks after the initial DFU diagnosis.10 This time frame was also selected to exclude early healers and to focus on patients whose ulcers were more likely to be chronic and nonresponsive to conservative management.
The index event was defined as the qualifying treatment (either CAMPs or debridement) received during the 4- to 12-week window. Patients were stratified into 2 cohorts. The debridement cohort included patients who received debridement during the treatment window but did not receive any CAMPs. The CAMPs cohort consisted of patients who received a CAMP following debridement. To isolate treatment effects specific to DFU management, patients with diagnoses of pressure ulcers, bullous disorders, burns, or corrosions were excluded.
Baseline characteristics, including demographics, comorbidities, medications, and laboratory values, were assessed and compared between cohorts before and after propensity score matching (PSM). Matching was performed to reduce confounding and to ensure comparability between groups for downstream outcome analyses.
Outcome measures and statistical analyses
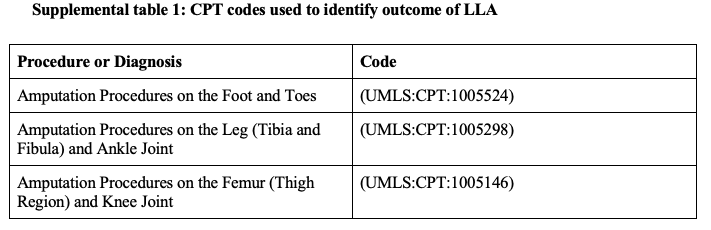
The primary outcome was LLA within 5 years of treatment. Current Procedural Terminology (CPT) codes are noted in Supplemental Table 1. Cohort-level statistics were calculated to assess the risk of LLA, including the number of patients in each cohort, the number of patients experiencing the outcome, the risk of amputation, the risk difference, and the risk ratio. Kaplan-Meier survival analysis was performed, and survival curves were compared using the log-rank test and hazard ratios.
To minimize confounding effects, PSM was performed to balance baseline characteristics between cohorts. Matching variables included race, ethnicity, age, sex, comorbidities, body mass index (BMI), concurrent medications, and relevant laboratory values. Race and ethnicity were assessed as baseline demographic characteristics and included in PSM to account for potential confounding related to demographic differences between treatment cohorts. Cohort matching was performed using the platform’s built-in analytics functionality, which uses a 1:1 nearest neighbor greedy matching methodology with a caliper of 0.1 pooled standard deviations (SDs). This method ensured that the baseline characteristics of the cohorts were comparable.
The risk of LLA after PSM was compared between cohorts using cohort-level statistics, and Kaplan-Meier survival analysis provided additional insight into amputation-free survival probabilities. Statistical significance was determined using a 2-sided P value threshold of less than .05, and 95% CIs were provided for all risk estimates.
Results
Cohort description
Before PSM, the CAMPs cohort (n = 2 273) and the debridement cohort (n = 31 050) demonstrated several statistically significant differences in baseline characteristics. Patients in the CAMPs group were older, with a mean (SD) age of 61.5 (11.7) years, compared with 60.4 (12.5) years in the debridement group (P < .001) (Table 1). Ages ranged from 18 to 90 years in both cohorts. The CAMPs group also had a higher proportion of male patients (71.2%) than the debridement group (67.8%; P = .001). In contrast, the debridement group had a higher proportion of female patients (28.3%) than the CAMPs group (25.5%, P = .004).
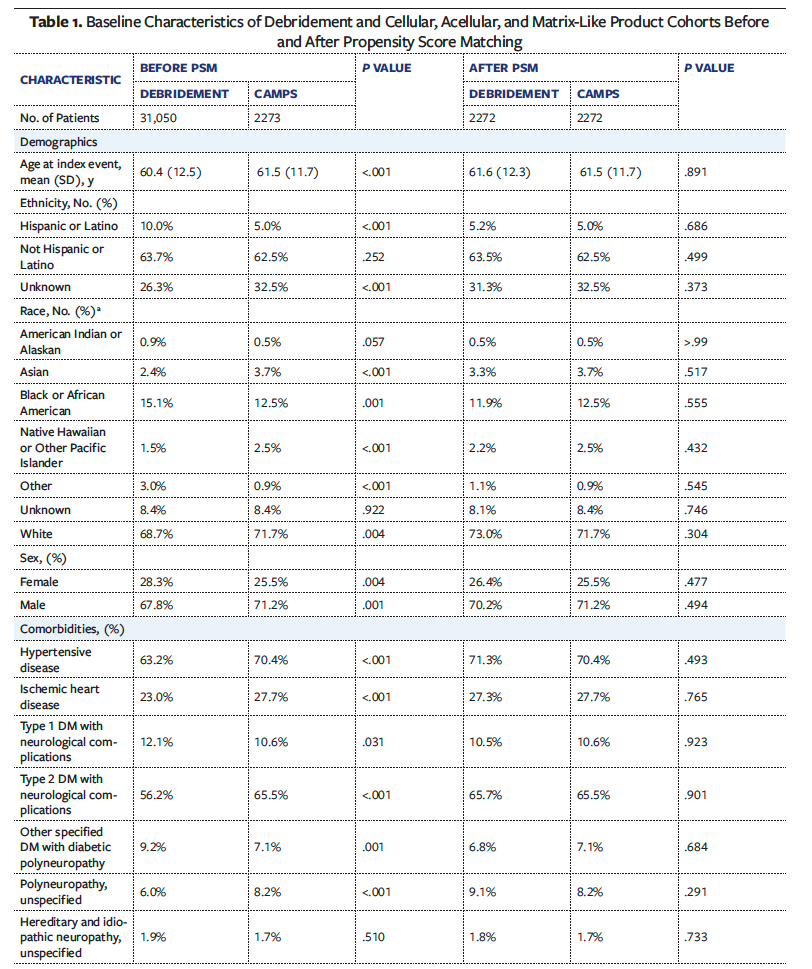
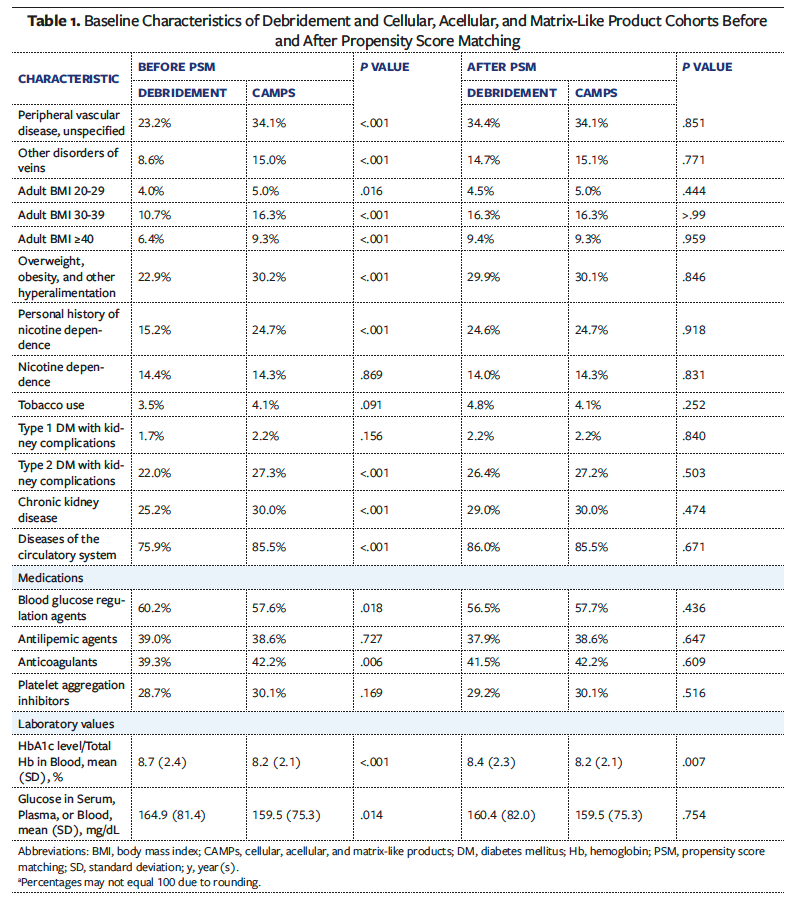
Racial and ethnic composition varied between groups. White patients made up 71.7% of the CAMPs cohort vs 68.7% of the debridement cohort (P = .004). Hispanic or Latino patients represented 5.0% of the CAMPs cohort and 10.0% of the debridement cohort (P < .001). Black or African American patients represented 12.5% of the CAMPs cohort and 15.1% of the debridement cohort (P < .001).
Comorbidities were more prevalent among those who received treatment with CAMPs across nearly all categories. Hypertensive disease was observed in 70.4% of the CAMPs cohort vs 63.2% of the debridement cohort (P < .001). Ischemic heart disease was present in 27.7% of CAMPs patients, compared with 23.0% of debridement patients (P < .001). Type 2 DM with neurological complications occurred in 65.5% of CAMPs patients vs 56.2% of debridement patients (P < .001). Peripheral vascular disease was also more common in the CAMPs group (34.1%) than in the debridement group (23.2%; P < .001), as was chronic kidney disease (30.0% and 25.2%, respectively; P < .001).
Obesity and elevated BMI were more frequent in the CAMPs cohort. Adult BMI of 30 to 39 was documented in 16.3% of CAMPs patients, compared with 10.7% of the debridement group (P < .001), and BMI greater than or equal to 40 was reported in 9.3% of CAMPs patients vs 6.4% of debridement patients (P < .001). Overall diagnoses related to overweight or obesity were also more common in the CAMPs cohort (30.2%) than in the debridement cohort (22.9%; P < .001).
Despite having fewer metabolic comorbidities, the debridement cohort had higher medication usage and worse glycemic control. Blood glucose-lowering agents were prescribed to 60.2% of debridement patients, compared with 57.6% of those in the CAMPs cohort (P = .018). Mean (SD) hemoglobin A1c level was higher in the debridement cohort at 8.7% (2.4%), compared with 8.2% (2.1%) in the CAMPs cohort (P < .001). Likewise, mean serum glucose levels were elevated in the debridement cohort (164.9 [81.4] mg/dL) relative to the CAMPs group (159.5 [75.3] mg/dL; P = .014).
After PSM, each cohort included 2 272 patients. All previously significant differences between groups were eliminated, resulting in well-balanced cohorts with comparable distributions across demographics, comorbidities, medication use, and laboratory values.
Risk of LLA and amputation-free survival analysis over 5 years
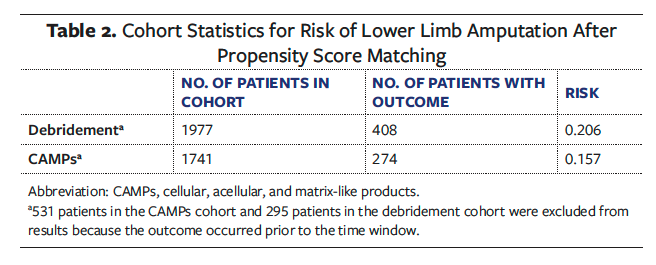
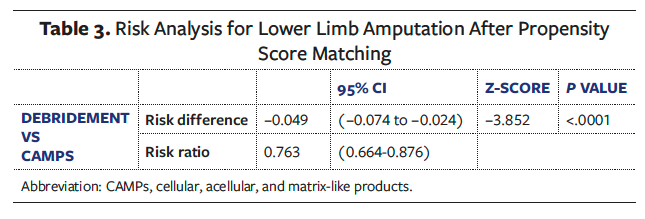
After excluding patients who experienced LLA prior to the treatment window, 1 741 patients remained in the CAMPs cohort and 1 977 remained in the debridement cohort. The 5-year risk of LLA was significantly lower in the CAMPs group (15.7%) compared with the debridement cohort (20.6%), yielding a risk difference of −4.90% (95% CI, −7.4 to −2.4%; P < .0001) (Tables 2 and 3). The corresponding risk ratio was 0.763 (95% CI, 0.664-0.876), indicating a 23.7% lower risk of amputation in the CAMPs group (Table 3).
Kaplan-Meier survival analysis supported this finding. The estimated 5-year amputation-free survival probability was 75.7% in the CAMPs cohort compared with 71.3% in the debridement cohort (Table 4). This difference was statistically significant based on the log-rank test (P = .001). Additionally, the Cox proportional hazards model yielded a hazard ratio of 0.763 (95% CI, 0.655-0.889; P = .006), consistent with a sustained survival benefit associated with CAMPs usage. Overall, patients treated with CAMPs had a significantly lower 5-year risk of amputation and a higher probability of amputation-free survival compared with those who did not receive CAMPs.

Discussion
In this large, multicenter, real-world study of patients with DFUs, treatment with CAMPs following debridement between 4 weeks and 12 weeks after initial diagnosis was associated with a significantly lower 5-year risk of LLA and higher amputation-free survival compared with debridement alone. After balancing baseline characteristics through PSM, patients treated with CAMPs had a 23.7% lower risk of amputation over 5 years (risk ratio, 0.763; 95% CI, 0.664-0.876), with a corresponding 4.9% absolute risk reduction. These findings suggest that CAMPs may have durable clinical benefits beyond initial wound healing. Given the chronic, recurrent nature of DFUs, analyzing long-term outcomes is essential to fully understand the effect of biologic therapies on patient trajectories.
These findings contrast with the existing literature, which has largely emphasized short-term end points. Landmark randomized controlled trials (RCTs) for Apligraf (Organogenesis, Inc; hereafter “Product A”)15, Grafix (Smith & Nephew; hereafter “Product B”)16, and Epifix (Mimedx Group, Inc; hereafter “Product C”)17 demonstrated faster wound healing but did not evaluate long-term downstream outcomes such as amputation or limb salvage. A meta-analysis echoed this limitation, showing robust effects on wound closure but noting a lack of evidence linking these improvements to long-term outcomes.18 Armstrong et al19 reported reduced 1-year amputation rates and mortality in Medicare recipients treated with skin substitutes. However, the present study expands upon this previous research by demonstrating that CAMP use may be associated with a reduced risk of amputation over 5 years. The 4.9% absolute risk reduction observed in this study corresponds to a number needed to treat of approximately 20, indicating that for every 20 patients treated with a CAMP, 1 amputation may be prevented. This effect size is clinically meaningful, suggesting that the benefits of CAMPs may be cumulative rather than transient. In addition, the present study found that 5-year
amputation-free survival was significantly higher in the CAMP group (75.7%) than in the debridement-only group (71.3%), further supporting the durability of benefit. This study builds on prior short-term efficacy data to suggest that CAMPs may offer durable benefits in limb preservation, while highlighting the need for future prospective studies comparing specific products and assessing long-term functional and quality-of-life outcomes.
To better understand the long-term outcomes of CAMPs usage, it is critical to account for baseline differences between the 2 treatment modalities and offer important context for interpreting downstream outcomes. Prior to PSM, patients in the CAMP group were generally more medically complex than those in the debridement group, with higher mean BMI (30.6 and 29.3, respectively; P < .001), greater prevalence of obesity (34.9% and 30.2%, respectively; P < .001), and higher rates of chronic kidney disease (21.5% and 17.4%, respectively; P < .001), coronary artery disease (27.6% and 24.3%, respectively; P < .001), and peripheral vascular disease (35.9% and 32.0%, respectively; P < .001). Nicotine use was also more common in the CAMPs cohort (13.9% and 10.5%, respectively; P < .001). Each of these comorbidities has been independently associated with delayed wound healing, increased infection risk, and increased likelihood of major amputation in patients with chronic lower extremity wounds.6,20-22 These differences highlight a potential selection bias, in which patients with more severe or nonhealing wounds are more likely to receive advanced therapies such as CAMPs.
Despite having more complex medical conditions, patients in the CAMPs group had lower mean hemoglobin A1c values (8.2% and 8.7%, respectively; P < .001) and were less likely to be prescribed glucose-lowering agents (57.6% and 60.2%, respectively; P = 0.018) compared with those who received debridement alone before PSM. These findings may seem paradoxical, particularly given that CAMP recipients had higher rates of chronic kidney disease, coronary artery disease, peripheral vascular disease, and obesity. However, this pattern likely reflects the influence of these comorbidities on both DM management and the interpretation of hemoglobin A1c values. Patients with advanced renal or vascular disease are at increased risk for hypoglycemia and medication-related complications, which may lead clinicians to use more conservative glycemic targets or reduced pharmacologic treatment intensity.23, 24 In addition, chronic illness can interfere with the accuracy of hemoglobin A1c measurements through mechanisms such as anemia, malnutrition, and altered red blood cell turnover, potentially underestimating true glycemic burden.24, 25 Because patients with more severe, nonhealing ulcers and multiple comorbidities may be more likely to receive advanced therapies such as CAMPs, the observed differences in hemoglobin A1c level and medication use likely reflect clinical limitations in treatment and measurement, rather than better metabolic control. These observations underscore the importance of accounting for baseline differences to ensure that outcome comparisons reflect the effects of treatment rather than difference in patient health status or disease severity.
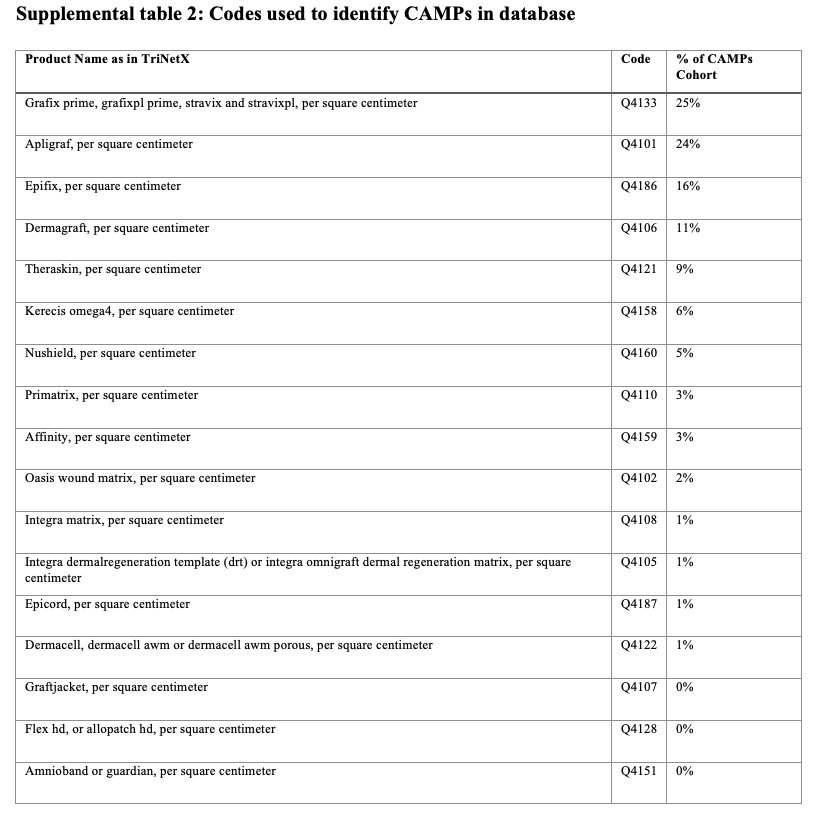
After adjusting for baseline differences, the authors of the present study also characterized the distribution of CAMPs used within the cohort to provide context of how these therapies are applied in real-world practice. Product B (25%), Product A (24%), and Product C (16%) were the most frequently used agents, together representing the most common choices among CAMP applications (Supplemental Table 2). Their prominence likely reflects a combination of product composition, method of preservation, timing of market entry, availability of supporting evidence, and reimbursement status. Product A, which was approved by the US Food and Drug Administration for DFUs in 2000, is a bilayered living skin equivalent and was among the first CAMPs supported by RCTs and included in clinical guidelines.26 Product B, a cryopreserved placental membrane introduced in 2012, is designed to retain viable endogenous cells and growth factors, and it has also been evaluated in RCTs.15 Product C, a dehydrated amnion/chorion membrane allograft that entered the United States market in 2011 is widely used due to its room-temperature storage, long shelf life, and ease of application in outpatient settings. 27 In addition to these product-specific features, factors such as institutional familiarity and access likely contribute to their more frequent use. Although the present study did not compare outcomes across individual CAMPs, the concentration of use among these agents suggests that clinician preferences align not only with perceived clinical effectiveness but also with logistical convenience, regulatory support, and economic feasibility.
While these usage patterns reflect how clinicians navigate product characteristics and practical considerations, they also raise important questions about who has access to these advanced therapies. This study found notable disparities in access to CAMPs by race and ethnicity. Prior to PSM, Black or African American patients were more likely to receive debridement alone and were less likely to receive CAMPs (15.1% and 12.5%, respectively; P = .001), despite evidence that this group experiences the highest national rates of LLA due to DFU.2, 28 Hispanic patients were similarly underrepresented in the CAMP cohort in the present study, despite having higher odds of major amputation compared with White patients in the context of DFUs.29 Although this study was not designed to assess disparities directly, these patterns may reflect structural inequities in access to specialized wound care, provider-level referral decisions, or systemic barriers such as insurance coverage, transportation, and limited continuity of care. 28-31 These findings highlight a potential gap in the equitable delivery of advanced therapies and underscore the need for further investigation into racial and ethnic disparities in limb-salvaging interventions.
Limitations
This study has several limitations that should be considered when interpreting the findings. The retrospective design and reliance on administrative coding data from the proprietary database introduce the potential for misclassification and coding errors. The accuracy of diagnoses, procedures, and comorbidities is dependent on correct and consistent documentation across contributing institutions.
While PSM was used to reduce confounding, unmeasured variables may still have influenced treatment selection and outcomes. Important clinical details such as wound size, depth, duration, infection severity, and off-loading practices—factors known to affect healing and amputation risk—were not available in the dataset. Similarly, standardized DFU grading systems such as Wagner grade could not be applied, because ICD-10 system does not capture formal grading. The study authors were unable to account for differences in debridement techniques or secondary wound dressings, either, which may vary across institutions and influence healing and amputation risk. Additionally, the decision to use CAMPs may have been influenced by provider experience, institutional protocols, or patient preferences, none of which could be controlled for in this analysis.
Another limitation involves the included billing codes and outcomes captured in this analysis. The dataset was limited to the diagnosis and procedure codes selected for cohort identification, and the study authors did not incorporate CPT codes for other aspects of standard DFU management, such as off-loading. In addition, LLAs were analyzed as a single composite outcome rather than stratified into major and minor categories, because doing the latter would have yielded smaller subgroups after PSM and reduced reliability of those comparisons.
Lastly, to ensure a sufficiently large patient population for meaningful analysis, the study authors combined various CAMPs into a single treatment group. This approach may have introduced heterogeneity, because the CAMPs group included a mix of biologic and scaffold-based products, some of which were more commonly used than others (Supplemental Table 2). Additionally, due to limitations in the available data, the study authors could not account for differences in dosing, frequency, or timing of CAMP application—factors that may also influence treatment outcomes. Furthermore, certain products (eg, Product B) are available in both cryopreserved and lyopreserved formulations, but these are grouped under the same billing code. As a result, it was not possible to differentiate outcomes by preservation method, which may have obscured formulation-specific effects.
Despite these limitations, this study offers valuable real-world evidence on the association between CAMP therapy and improved clinical outcomes in patients with DFUs, supporting the need for further prospective and controlled investigations.
Conclusion
This study adds to the growing body of evidence supporting the use of CAMPs as an effective adjunctive therapy in DFU management. In this large, multicenter, real-world analysis, treatment with CAMPs after debridement for DFUs was associated with a significant long-term reduction in LLA and improved amputation-free survival over 5 years. These findings extend prior evidence focused primarily on short-term wound closure, suggesting that CAMPs may confer durable benefits in limb preservation. While barriers such as cost and limited familiarity with these therapies remain, the demonstrated improvements in patient outcomes justify their broader adoption into standard of care practices. Future research should focus on elucidating the effect of early intervention and exploring personalized approaches to enhance the effectiveness of these promising therapies.
Author and Public Information
Authors: Abigail Royfman, BS1; Sarah Lux, BA1; Kennedy Couch, BS1; Molynna Nguyen, BS1; Mandy Wong, BS1; Emily Gong, BS1; and Richard Simman, MD2,3
Affiliations: 1University of Toledo College of Medicine and Life Sciences, Toledo, OH, USA; 2Jobst Vascular Institute/ProMedica, Toledo, OH, USA; 3Department of Surgery, University of Toledo College of Medicine and Life Sciences, Toledo, OH, USA.
Author Contributions: The submitted manuscript has been reviewed and approved by all authors, who accept full responsibility for the design and conduct of the study, had access to the data, and controlled the decision to publish.
Disclosure: The authors disclose no relevant financial or nonfinancial interests.
Correspondence: Richard Simman, MD; Jobst Vascular Institute, 2109 Hughes Drive, CJT Suite 400, Toledo, OH 43606; Richard.simmanmd@promedica.org
Manuscript Accepted: November 21, 2025.
References
1. Centers for Disease Control and Prevention. National diabetes statistics report. Published May 15, 2024. Accessed September 12, 2025. https://www.cdc.gov/diabetes/php/data-research/index.html
2. McDermott K, Fang M, Boulton AJM, Selvin E, Hicks CW. Etiology, epidemiology, and disparities in the burden of diabetic foot ulcers. Diabetes Care. 2023;46(1):209-221. doi:10.2337/dci22-0043
3. Petersen BJ, Linde-Zwirble WT, Tan TW, et al. Higher rates of all-cause mortality and resource utilization during episodes-of-care for diabetic foot ulceration. Diabetes Res Clin Pract. 2022;184:109182. doi:10.1016/j.diabres.2021.109182
4. Ndosi M, Wright-Hughes A, Brown S, et al. Prognosis of the infected diabetic foot ulcer: a 12-month prospective observational study. Diabet Med. 2018;35(1):78-88. doi:10.1111/dme.13537
5. Rice JB, Desai U, Cummings AKG, Birnbaum HG, Skornicki M, Parsons NB. Burden of diabetic foot ulcers for Medicare and private insurers. Diabetes Care. 2014;37(3):651-658. doi:10.2337/dc13-2176
6. Chen L, Sun S, Gao Y, Ran X. Global mortality of diabetic foot ulcer: a systematic review and meta-analysis of observational studies. Diabetes Obes Metab. 2023;25(1):36-45. doi:10.1111/dom.14840
7. Jeffcoate WJ, Harding KG. Diabetic foot ulcers. Lancet. 2003;361(9368):1545-1551. doi:10.1016/S0140-6736(03)13169-8
8. Coye, T. L., Bargas Ochoa, M., Zulbaran-Rojas, A., Martinez Leal, B., Quattas, A., Tarricone, A., Chung, J., Najafi, B., & Lavery, L. A. (2025). Healing of diabetic neuropathic foot ulcers receiving standard treatment in randomised controlled trials: A random effects meta-analysis. Wound repair and regeneration : official publication of the Wound Healing Society [and] the European Tissue Repair Society, 33(1), e13237. https://doi.org/10.1111/wrr.13237
9. Apelqvist J, Bakker K, van Houtum WH, Schaper NC. Practical guidelines on the management and prevention of the diabetic foot: based upon the International Consensus on the Diabetic Foot (2007) prepared by the International Working Group on the Diabetic Foot. Diabetes Metab Res Rev. 2008;24(Suppl 1):S181-S187. doi:10.1002/dmrr.848
10. Nabuurs-Franssen MH, Huijberts MSP, Nieuwenhuijzen Kruseman AC, Willems J, Schaper NC. Health-related quality of life of diabetic foot ulcer patients and their caregivers. Diabetologia. 2005;48(9):1906-1910. doi:10.1007/s00125-005-1856-6
11. American Diabetes Association Professional Practice Committee. 12. Retinopathy, neuropathy, and foot care: Standards of Care in Diabetes—2025. Diabetes Care. 2025;48(Suppl 1):S252-S265. doi:10.2337/dc25-S012
12. Wu S, Carter M, Cole W, et al. Best practice for wound repair and regeneration: use of cellular, acellular and matrix-like products (CAMPs). J Wound Care. 2023;32(Suppl 4b):S1-S31. doi:10.12968/jowc.2023.32.Sup4b.S1
13. Hughes OB, Rakosi A, Macquhae F, Herskovitz I, Fox JD, Kirsner RS. A review of cellular and acellular matrix products: indications, techniques, and outcomes. Plast Reconstr Surg. 2016;138(3 Suppl):138S-147S. doi:10.1097/PRS.0000000000002643
14. Malin B. Summary of the assessment of TriNetX data privacy principles through an empirical analysis; December 8, 2020. Accessed January 29, 2026. https://trinetx.com/wp-content/uploads/2021/12/TriNetX-Empirical-Summary-by-Brad-Malin-2020.pdf?_gl=1*g232rf*_up*MQ..*_ga*MTc5NjY2NDE0MS4xNzY3NjY2ODM4*_ga_ND9FN4NT6M*czE3Njc2NjY4MzckbzEkZzAkdDE3Njc2NjY4MzckajYwJGwwJGgw.
15. Veves A, Falanga V, Armstrong DG, Sabolinski ML. Graftskin, a human skin equivalent, is effective in the management of noninfected neuropathic diabetic foot ulcers: a prospective randomized multicenter clinical trial. Diabetes Care. 2001;24(2):290-295. doi:10.2337/diacare.24.2.290
16. Lavery LA, Fulmer J, Shebetka KA, et al. The efficacy and safety of Grafix for the treatment of chronic diabetic foot ulcers: results of a multi-centre, controlled, randomised, blinded clinical trial. Int Wound J. 2014;11(5):554-560. doi:10.1111/iwj.12329
17. Zelen CM, Serena TE, Denoziere G, Fetterolf DE. A prospective randomised comparative parallel study of amniotic membrane wound graft in the management of diabetic foot ulcers. Int Wound J. 2013;10(5):502-507. doi:10.1111/iwj.12097
18. Monami M, Ragghianti B, Scatena A, et al. Effectiveness of different advanced wound dressings versus standard of care for the management of diabetic foot ulcers: a meta-analysis of randomized controlled trials for the development of the Italian guidelines for the treatment of diabetic foot syndrome. Acta Diabetol. 2024;61(12):1517-1526. doi:10.1007/s00592-024-02320-2
19. Armstrong DG, Tettelbach WH, Chang TJ, et al. Observed impact of skin substitutes in lower extremity diabetic ulcers: lessons from the Medicare database (2015-2018). J Wound Care. 2021;30(Suppl 7):S5-S16. doi:10.12968/jowc.2021.30.Sup7.S5
20. Luo Y, Liu C, Li C, Jin M, Pi L, Jin Z. The incidence of lower extremity amputation and its associated risk factors in patients with diabetic foot ulcers: a meta-analysis. Int Wound J. 2024;21(7):e14931. doi:10.1111/iwj.14931
21. Bechara N, Hng TM, Gunton JE. The association between tobacco smoking and systolic toe pressures in active foot ulceration. Sci Rep. 2024;14(1):8550. doi:10.1038/s41598-024-59158-5
22. Chin BZ, Lee P, Sia CH, Hong CC. Diabetic foot ulcer is associated with cardiovascular-related mortality and morbidity: a systematic review and meta-analysis of 8062 patients. Endocrine. 2024;84(3):852-863. doi:10.1007/s12020-024-03696-5
23. Galindo RJ, Beck RW, Scioscia MF, Umpierrez GE, Tuttle KR. Glycemic monitoring and management in advanced chronic kidney disease. Endocr Rev. 2020;41(5):756-774. doi:10.1210/endrev/bnaa017
24. American Diabetes Association Professional Practice Committee. 11. Chronic kidney disease and risk management: Standards of Care in Diabetes—2025. Diabetes Care. 2025;48(Suppl 1):S239-S251. doi:10.2337/dc25-S011
25. Sacks DB, Arnold M, Bakris GL, et al. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Diabetes Care. 2023;46(10):e151-e199. doi:10.2337/dci23-0036
26. Zaulyanov L, Kirsner RS. A review of a bi-layered living cell treatment (Apligraf) in the treatment of venous leg ulcers and diabetic foot ulcers. Clin Interv Aging. 2007;2(1):93-98.
27. Zelen CM, Serena TE, Gould L, et al. Treatment of chronic diabetic lower extremity ulcers with advanced therapies: a prospective, randomised, controlled, multi-centre comparative study examining clinical efficacy and cost. Int Wound J. 2016;13(2):272-282. doi:10.1111/iwj.12566
28. Akinlotan MA, Primm K, Bolin JN, et al. Racial, rural, and regional disparities in diabetes-related lower-extremity amputation rates, 2009–2017. Diabetes Care. 2021;44(9):2053-2060. doi:10.2337/dc20-3135
29. Tan TW, Shih CD, Concha-Moore KC, et al. Disparities in outcomes of patients admitted with diabetic foot infections. PLoS One. 2019;14(2):e0211481. doi:10.1371/journal.pone.0211481
30. Creager MA, Matsushita K, Arya S, et al. Reducing nontraumatic lower-extremity amputations by 20% by 2030: time to get to our feet. a policy statement from the American Heart Association. Circulation. 2021;143(17):e875-e891. doi:10.1161/CIR.0000000000000967
31. Gallagher KA, Mills JL, Armstrong DG, et al. Current status and principles for the treatment and prevention of diabetic foot ulcers in the cardiovascular patient population: a scientific statement from the American Heart Association. Circulation. 2024;149(4):e232-e253. doi:10.1161/CIR.0000000000001192