Type 2 Inflammation and the Biology of Itch in Prurigo Nodularis
Clinical Summary
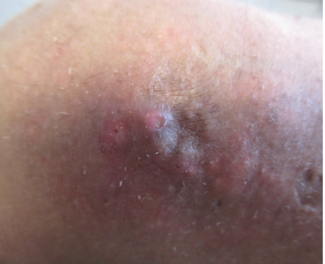
Prurigo Nodularis (PN): Type 2 Inflammation and IL-31–Driven Chronic Itch
-
Prurigo nodularis (PN): Type 2 inflammation is a final common pathway driving chronic itch, neural hypersensitivity, fibrosis, and acanthosis; occurs with or without atopic history (e.g., dry skin–induced barrier disruption).
-
Cytokines (IL-31, IL-13, IL-4): IL-31 most directly linked to pruritus; IL-13 (more prominent than IL-4) promotes IL-31 production. Blocking IL-4/IL-13/IL-31 reduces itch and inflammation; IL-31 levels are particularly high in PN.
-
Neuroimmune loop: Type 2 cytokines directly depolarize sensory neurons, increase mechanical sensitivity, and trigger neuropeptide release → amplifying inflammation, fibrosis, and the itch–scratch cycle. PN reflects variable type 2 intensity across patients.
Reviewed by Riya Gandhi, MA, Associate Editor of Immunology Group
Chronic itch in prurigo nodularis is increasingly understood as a neuroimmune process driven by type 2 cytokines rather than histamine alone. In this interview, Dr Kim explores how IL-4, IL-13, and IL-31 interact with sensory neurons to sustain pruritus—even in patients without classic atopy.
Brian S. Kim, MD, MTR, FAAD, is vice chair of research and site chair of Morningside and Mount Sinai West, in the Kimberly and Eric J. Waldman department of dermatology. He is also director of the Mark Lebwohl Center for Neuroinflammation and Sensation and lead of the Allen Discovery Center for Neuroimmune Interactions at the Icahn School of Medicine at Mount Sinai.
Transcript
What role does type 2 inflammation play in driving chronic itch in PN?
Dr Kim: Type 2 inflammation is actually a default inflammatory reaction when the skin barrier is disrupted, in general, when there's damage to the skin barrier, when there's natural damage. So, whether there's penetration through the skin barrier in some way, not in a traumatic way, but in a very subtle kind of way. In atopic dermatitis and in prurigo nodularis, essentially, what you have is a form of disruption of the deeper actual layers of the skin, and it activates processes like fibrosis, which is also seen with type 2 inflammation. And really, what you get as a consequence of type 2 inflammation in prurigo nodularis is itch number one, but also neural hypersensitivity, which is actually more than itch. It's the proclivity to keep itching and scratching more and more, but also fibrosis and then more inflammation and thickening, and what we refer to as acanthosis, where the epidermis also thickens as well. And this is how you get these really distinct nodular lesions, so type 2 inflammation has many tentacles within the skin by which it promotes pathology in prurigo nodularis or PN.
How can strong type 2 immune signaling occur in PN patients without a clear atopic history?
Dr Kim: Yeah, so atopic history or atopy or allergy as we describe it, is actually a genetic predilection or proclivity to develop type 2 inflammation. So it's a hardwired kind of thing; somehow, I might have a brother or sister with fairly similar genetics, but for some reason they, from birth, are more prone to atopic dermatitis or asthma or food allergy or something like that. There's some genetic predisposition, but that's not necessarily what's absolutely required to develop type 2 inflammation. So type 2 inflammation outside of that genetic predisposition setting. For example, I could just have simply have dry skin, and as a consequence of having dry skin, I'll have a lot of type 2 inflammation in my skin without having any history of atopy. And it's similar in PN, you might have a history of atopy, and as a consequence, half PN, or you might just develop PN in and of itself, in which that local tissue microenvironment has a lot of type 2 inflammation. And the way I describe it is that type 2 inflammation can occur in the setting of atopy, but it can also occur outside of atopy. That doesn't have to be always in the setting of atopy, but it is a final common pathway. So, no matter how you end up having PN, there are lots of ways you can arrive at PN. You do have type 2 inflammation. That's why there's a universal therapeutic response to blockade of type 2 inflammation in PN.
Among IL-4, IL-13, and IL-31, which cytokine appears most critical for sustaining pruritus in PN?
Dr Kim: Yeah. So if you think about pruritus itself, it's really hard to break down. But if you really think of just pruritus itself, IL-31 is the one that kind of sticks its head up above everything else. But as we've been talking about, there's also inflammation in PN as well. And what incites IL-31 production oftentimes is IL-4 and IL-13, and amongst those 2 in general, this is not just in PN, but in atopic dermatitis, the cytokine between IL-4 and IL-13 that tends to pop up a lot more tends to be more IL-13. That's not to say IL-4 doesn't contribute, but it's really a combination of these cytokines that creates the soup that's in PN. And we think that these cytokines push slightly different buttons, and we're still learning what all the outputs of those different buttons are. But really, there are different ways you can tackle PN. That's why you have therapies that work, whether they block IL-4, IL-13, or IL-31. And presumably, if you wanted to block all of the above, you might even have a better effect overall as well, potentially.
What distinguishes IL-31–mediated itch in PN from itch pathways in other inflammatory skin diseases?
Dr Kim: Yeah, it's tricky. It's interesting. For instance, IL-31, there are lots of ways to think about IL-31. So IL-31 levels can be very high, and they are very, very high in PN, but they're not necessarily high in other conditions per se, objectively. But by blocking IL-31 in PN, that's an obvious one. The levels are so high that when you shut it down, you get rid of the itch. But what we also believe is that IL-31 levels may not even have to be terribly high. And by blocking that, you could actually shut down itch as well. And we know that this is true for IL-4 and IL-13 as well. You don't have to have sky-high levels. But blocking even just kind of what we consider low levels or even maybe what we refer to as physiologic levels also limits itch because it actually decreases the sensitivity of the nerve to not just IL-13 or IL-4 or IL-31, but also to many other what we refer to as pruritogens, like histamine or even other things like leukotrienes. We don't know the full extent of all that causes it, but we know that the electricity of the nerve can be dialed down when we block molecules like IL-31.
How do type 2 cytokines interact with sensory neurons to perpetuate the itch–scratch cycle?
Dr Kim: Yeah, they interact directly. So what we think is that there are a number of things that these cytokines do. For one, they directly activate the nerve, and they cause the nerve to actually depolarize and make it more sensitive to many other factors, maybe even mechanical factors. And this is why, for instance, if you have atopic dermatitis, you cannot wear wall sweater because the mechanical wool fibers will set off your itch. Why is that? Those aren't pruritogens, those aren't itch molecules, those are mechanical fibers that they set off itch because your itch nerves have now become really sensitive. But also what we do know is that these cytokines also not only trigger itch, so when we talk about itch, they trigger a signal towards the spinal cord into the brain. That's the sensation of itch. But what they also do is they trigger the release of neuropeptides, neuropeptides get released into the tissue and cause more inflammation in the setting of PN, more fibrosis, and turn those inflammatory molecules, and maybe even the fibroblasts, can turn back on the nerve and cause more itch. So the cytokines cause direct itch and indirect itch by acting on the nerves and maybe other things as well.
Do emerging immune endotypes suggest that PN is not a uniform type 2 disease?
Dr Kim: I'm not so sure there are such a thing as necessarily a type 2 disease. There are atopic diseases, which we talked about earlier, in which there's a genetic predisposition to a certain kind of form of type 2 inflammation, but anyone can develop type 2 inflammation. If I scratch my skin right now, I'll cause myself type 2 inflammation. That does not mean I have type 2 disease. But there are many diseases in which modules of type 2 inflammation exist. And what do they do? They might cause itch, they might cause fibrosis, and they might even cause elements of pain through indirect mechanisms as well. We don't know all that type 2 inflammation does. So the idea is that if you block a type 2 process within a disease, then you will block certain kinds of outputs. Now, does that mean that if you make a pie chart of PN, there are different kinds of PN in which type 2 inflammation is bigger in terms of proportion of that pie chart versus others? Absolutely. But I think that's true even in atopic dermatitis. I think that's true in general. In fact, I would even argue that even conditions that are not type 2 diseases, if you want to use that term, even in a condition like psoriasis, my personal belief is that people who are very itchy, who have psoriasis, have some type 2 inflammation that's driving the itch, even in a TH17 disease. So it's really about how much type 2 inflammation is there, how operative it is, and then if you block it, would you get resolution of different outputs, whether it be itch, inflammation, or fibrosis.