FDA Approves Cosentyx for the Treatment of Pediatric Patients Aged 12 Years and Older With Moderate-To-Severe Hidradenitis Suppurativa
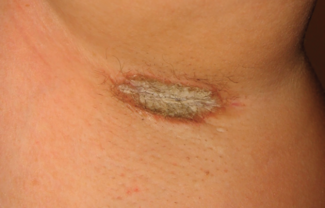
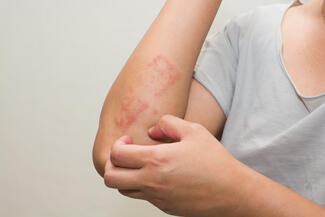
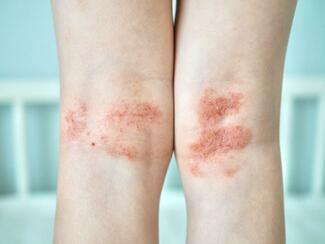
On March 13, 2026, the US Food and Drug Administration (FDA) approved Cosentyx (secukinumab) for the treatment of pediatric patients aged 12 years and older with moderate-to-severe hidradenitis suppurativa (HS). Secukinumab, a fully human interleukin-17A (IL-17A) inhibitor, is the only IL-17A inhibitor approved for this population and is administered with weight-based dosing.
The approval is supported by well-controlled adult HS trials, including SUNSHINE and SUNRISE, along with pharmacokinetic modeling and pediatric data from other approved indications. These analyses demonstrated that weight-based dosing in pediatric patients achieves drug exposure comparable to that observed in adults with HS. The indication addresses a gap in care for adolescents, as HS often begins around puberty and can lead to progressive disease and scarring if not treated early.
“Hidradenitis suppurativa often begins in adolescence and can cause irreversible scarring and disabilities,” said Alexa B. Kimball, MD, MPH, lead investigator of the SUNSHINE and SUNRISE trials. “The approval of Cosentyx represents an important advancement for younger HS patients who have had limited treatment options.”
Secukinumab targets IL-17A, a key cytokine involved in inflammatory pathways across multiple immune-mediated diseases. With this approval, secukinumab now holds 4 pediatric indications, including plaque psoriasis, psoriatic arthritis, enthesitis-related arthritis, and HS.
The safety profile in pediatric HS is consistent with prior indications. Secukinumab may increase the risk of infections, including tuberculosis, and requires screening prior to initiation. Other important risks include hypersensitivity reactions, inflammatory bowel disease exacerbations, and rare severe skin reactions. The most common adverse events include upper respiratory infections, diarrhea, and cold-like symptoms.
“HS affects far more than skin; it impacts confidence, emotional well-being, and relationships during a formative period,” said Brindley Brooks, founder and CEO of HS Connect.
Reference
Novartis Cosentyx receives FDA approval for pediatric patients aged 12+ with moderate to severe hidradenitis suppurativa. News release. March 13, 2026. Accessed March 17, 2026. https://www.novartis.com/us-en/news/media-releases/novartis-cosentyx-receives-fda-approval-pediatric-patients-aged-12-moderate-severe-hidradenitis-suppurativa