Left Atrial Myxoma: Immediate Presentation, Diagnosis, and Treatment
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Cath Lab Digest or HMP Global, their employees, and affiliates.
Richard Casazza MAS1; Neel Patel MD1; Allan Mattia MD2; Paul Saunders MD2; Arsalan Hashmi MD1; Greg Ribakove MD2
1Department of Cardiology; 2Department of Cardiothoracic Surgery
Maimonides Medical Center, Brooklyn, New York
Disclosures: The authors report no conflicts of interest regarding the content herein.
The authors can be contacted via Richard Casazza, MAS, at rcasazza@maimo.org.
Introduction
Cardiac myxomas are histologically benign tumors; however, they may lead to life-threatening complications. Myxomas are the most common primary cardiac tumor. Roughly 75% of myxomas originate in the left atrium at the mitral annulus or the fossa ovalis border of the interatrial septum. Twenty percent (20%) arise from the right atrium, while 5% stem from both atria and the ventricle.1 Myxomas are linked with several difficulties, including outflow obstruction and embolic events. Patients with left-sided myxomas usually develop signs and symptoms associated with mitral valve obstruction or regurgitation, left-sided heart failure, and secondary pulmonary hypertension. The most common symptom is dyspnea with exertion, followed by orthopnea, paroxysmal nocturnal dyspnea, and pulmonary edema.2 The manifestations of cardiac myxomas depend on their location, size, and mobility.3
History of Presentation
We present a 68-year-old male that presented to the emergency department with a complaint of worsening shortness of breath and palpitation. Upon arrival, the patient was hemodynamically stable with tachycardia. An electrocardiogram (EKG) showed atrial flutter at a rapid rate. The patient reported worsening shortness of breath that had been transient for a few years. However, it had gotten worse with dyspnea on exertion for the last month prior to admission, along with palpitations. The patient also reported chest pressure during ambulation in recent months. Subsequently, the point-of-care echocardiogram showed a left atrial mass.
Past Medical History
On further discussions, the patient was diagnosed with this left atrial mass in 2019 in Jamaica. Surgical consultation at that time was performed; however the patient was turned down, as he was deemed a poor surgical candidate. The patient was medically managed with heart rate control. The patient had requested immigration to the United States, but due to the COVID-19 pandemic, his visa was denied until the time of presentation in our emergency department. Multiple attempts were made to obtain the patient’s medical records from Jamaica, but proved unsuccessful. Laboratory work-up showed evidence of congestive hepatopathy with elevated liver enzymes. On physical exam, patient was jaundiced and grossly edematous.
Investigations
Electrocardiogram. ECG showed an atypical atrial flutter with an elevated heart rate of 130 bpm.
Transthoracic echocardiogram (TTE). The left ventricular (LV) ejection fraction, by visual estimation, was approximately 35% with moderately decreased left ventricular systolic function. There was global cardiomyopathy with LV septal wall increasingly hypokinetic. The left atrium (LA) was severely dilated. The LA volume index = 53.99 ml/m² (upper limit of normal = 34 ml/m2). The right ventricle (RV) was also severely enlarged with severely reduced RV systolic function. There were moderately elevated pulmonary artery systolic pressures of approximately 58 mmHg. The right atrium was severely dilated. There was the presence of mild aortic valve sclerosis without stenosis, moderate tricuspid regurgitation, and mild pulmonic valve regurgitation. A small pericardial effusion was also noted.
TTE revealed a large echogenic left atrial mass (measuring about 4.5 cm x 8.2 cm) that appeared attached to interatrial septum. The initial impression was atrial myxoma. Differential diagnosis included thrombus or other cardiac tumor. The mass was protruding into mitral valve orifice during diastole, causing functional mitral stenosis. The anterior mitral leaflet was thickened, in particular, at the leaflet tip, indicating a possible rheumatic mitral valve. The patient also had moderate mitral valve regurgitation.
Computed tomography chest with intravenous contrast. An ovoid homogeneous mass of complex density was within the left atrium, measuring 4.7 x 8.3 x 5.7 cm. The mass occupied most of the left atrium with some left atrial space remaining superiorly. The mass abutted the interatrial septum and mitral valve. The heart was globally enlarged. There was a small circumferential pericardial effusion. There was reflux of contrast into the coronary sinus, and reflux of contrast into the inferior vena cava, with mildly dilated hepatic veins, seen with right heart dysfunction. The mildly dilated main pulmonary artery measured 3.1 cm. The thoracic aorta was of normal caliber.
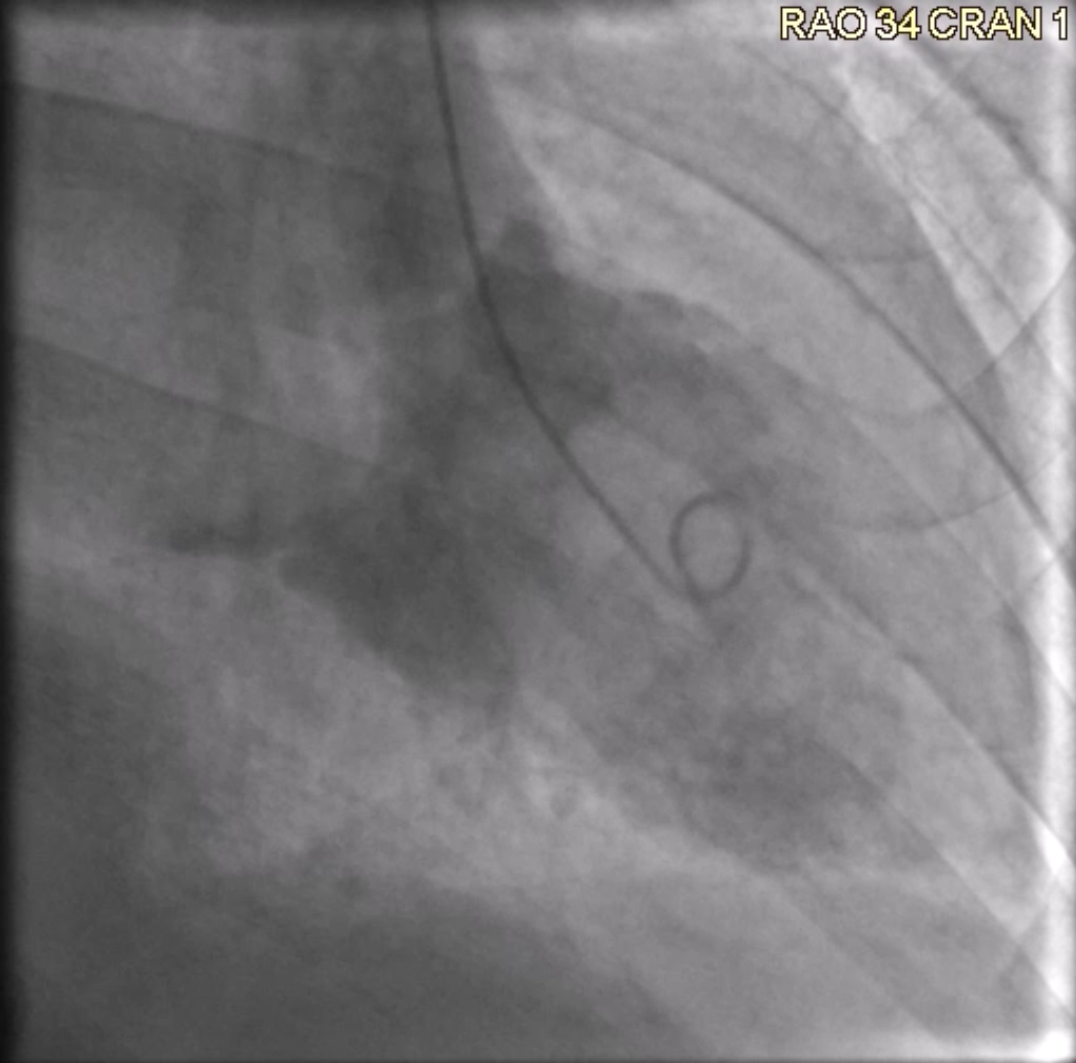
Cardiac Catheterization. Angiography was requested as a pre-procedural workup, which showed normal coronary arteries and branches supplying the left atrial mass. A small LV gram (Figure 1, Video 1) showed global cardiomyopathy with moderately reduced LV ejection fraction and a globular structure prolapsing into the LV during diastole and retracting during systole.
Video 1. Left ventriculogram with mxyoma.
Management
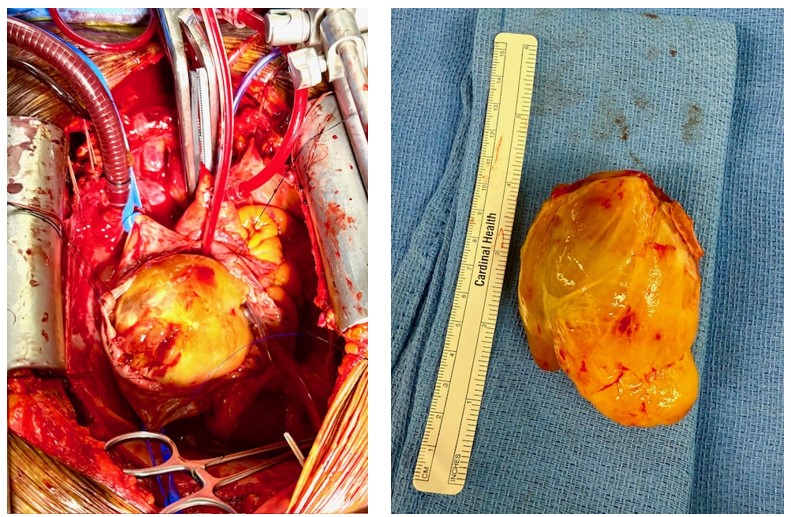
The patient was started on anticoagulation with appropriate rate control medications. The patient was admitted under the cardiothoracic (CT) surgery team with a plan for excision of the suspected left atrial myxoma. Intraoperatively, the patient underwent ascending aorta and bicaval cannulation for cardiopulmonary bypass. The right atrium was opened. The interatrial septum was identified and the fossa ovalis opened. Upon opening, the large cardiac tumor could be found, adherent to the interatrial septum (Figure 2).
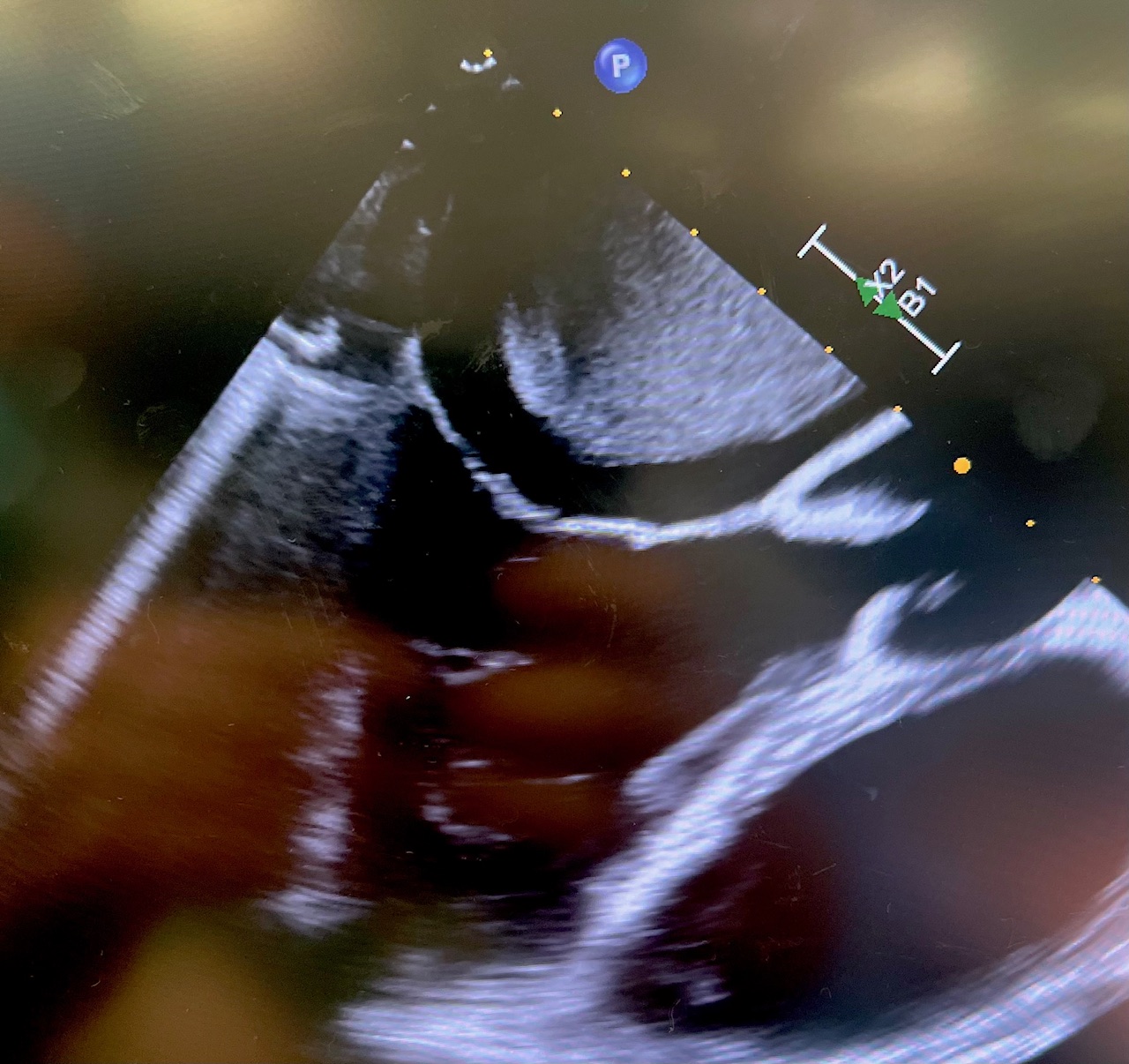
The mass was resected en bloc with the septum. The mitral valve was repaired with a 32 mm CG Future band (Medtronic). The interatrial septum was reconstructed with autologous pericardium. The tricuspid valve was repaired with a Kay annuloplasty. Postoperatively, the patient remained with severe right ventricular dysfunction requiring open chest and central venoarterial extracorporeal membrane oxygenation (VA ECMO) which was successfully weaned on postoperative day 5. Intra-operative echo provided exceptional images of the myxoma, in addition to displaying the nearly complete obstruction of the left atrial outflow tract (Figure 3, Video 2).
Video 2. Intra-operative echo with myxoma.
Outcome and Follow-Up
Pathology from the operating room confirmed the diagnosis of a left atrial myxoma. The patient had modest recovery of right ventricular function. He was started on sildenafil for pulmonary hypertension. The patient was ultimately discharged to a subacute care facility. On follow-up, he reported sternal pain for several weeks post operatively. The patient’s shortness of breath had marked resolution.
Discussion
The principal method for myxoma diagnosis is echocardiography. Echo provides an excellent evaluation of morphology, size, pedunculation origin, involvement of mitral valve leaflets, and obstruction of the LV outflow tract. LV angiography may add little clinical benefit for diagnosing myxoma; however, on initial catheter insertion into the LV for end diastolic pressures, our catheter was vertical in fashion, and seemingly caught behind the papillary muscle. At this point we shot a 10 cc ventriculogram to ensure the shaft of our pigtail was not in contact with the myxoma before pulling back the catheter into the aorta. The LV gram clearly demonstrated a large globular structure protruding into the ventricle during diastole. After, we inserted an .035-inch regular J-wire through the pigtail and slowly retracted the catheter back into the aorta, monitoring it with the goal of not interrupting left atrial outflow and disturbing the myxoma.
In contemporary cardiac catheterization, left ventriculography may provide little benefit in diagnosing ejection fraction and mitral valve insufficiency. Modern echocardiography has become the gold standard in identifying structural disease and heart function. However, in our case, the use of left ventriculography helped to identify the catheter position in relation to the myxoma and provided a layout on where we wanted our catheter to be on pullback.
Take-Home Message
An LV gram can provide some utility in patients with myxoma in determining catheter position to ensure no disruption occurs between the catheter and tumor.
References
1. Ha JW, Kang WC, Chung N, et al. Echocardiographic and morphologic characteristics of left atrial myxoma and their relation to systemic embolism. Am J Cardiol. 1999 Jun 1; 83(11): 1579-1582, A8. doi:10.1016/s0002-9149(99)00156-3
2. Goswami KC, Shrivastava S, Bahl VK, et al. Cardiac myxomas: clinical and echocardiographic profile. Int J Cardiol. 1998 Feb 28; 63(3): 251-259. doi:10.1016/s0167-5273(97)00316-1
3. Asad D, Abu Moch MA, Amarin H, et al. Left ventricular myxoma: a case report. Cureus. 2023 Sep 28; 15(9): e46112. doi:10.7759/cureus.46112