Successful Application of Bilayer Living Skin Substitute in the Treatment of Pyoderma Gangrenosum: A Case Series
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Pyoderma gangrenosum (PG) is a rare neutrophilic dermatosis marked by painful ulcerations and is commonly associated with autoimmune conditions. Surgical debridement is often avoided due to the risk of pathergy, but when performed under adequate immunosuppression it may be beneficial. Objective. To evaluate outcomes using a bilayer living skin substitute with or without surgical debridement in treating refractory PG. Methods. Eight patients with biopsy-confirmed or clinically diagnosed PG were retrospectively reviewed over a 5-year period using an electronic health record system’s self-reporting tool. All patients underwent serial surgical debridement after controlling systemic inflammation, followed by skin substitute application. Data on comorbidities, laboratory values, prior treatments, and wound healing were analyzed. Results. Patients had significant comorbidities, including hypertension, vascular disease, and autoimmune disorders. Most showed elevated inflammatory markers and varied glycemic control. Initial post-debridement wound enlargement was observed, but all patients demonstrated progressive contraction and full epithelialization. Seven patients developed secondary infections, likely due to immunosuppression. Healing time ranged from several weeks to longer than 6 months. Conclusion. This series supports that surgical debridement combined with bilayer living skin substitute application in a controlled inflammation setting can safely and effectively treat complex PG ulcers. It challenges the traditional avoidance of surgery and promotes a multimodal treatment strategy.
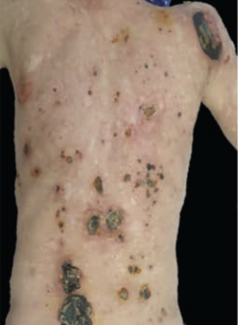
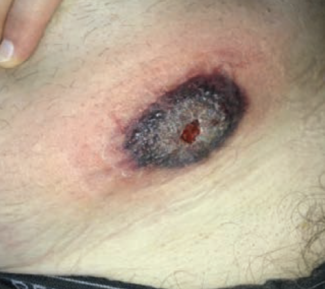
Pyoderma gangrenosum (PG) is an uncommon ulcerative skin disorder categorized as neutrophilic dermatosis.1 PG often presents as a rapidly progressing skin ulceration that is characterized by extensive edema, surface purulent exudate, and erythematous undermined skin edges. Although the pathophysiology is not entirely understood, it is well established that the disease process is autoimmune-mediated.2,3 PG is commonly associated with rheumatoid arthritis and inflammatory bowel disease.
PG’s hallmark clinical presentation is characterized by a painful necrotic ulcer that progresses to a larger, more diffuse wound.3 Diagnosis of PG is difficult because no definitive histologic features or laboratory data identify the disease process specifically.4,5 PG is typically a diagnosis of exclusion after failed attempts at treatment with conservative wound measures.5 Consistent with prior literature, histopathology in the cohort in the present study was supportive but not diagnostic of PG, underscoring the importance of clinicopathologic correlation rather than reliance on biopsy findings alone.
After PG is identified, attempts are made to mitigate the process with intralesional steroid injections, systemic steroids, immunosuppressive agents, and biologic medications.6 Surgical management with debridement alone is contraindicated because it may trigger a worsening immune response, causing the wound to become progressively larger in a process known as pathergy.7 The success of surgical management has been variable when combined with immunosuppression.7 Multiple treatment modalities have been combined to treat PG, including negative pressure wound therapy, split-thickness skin grafts, and xenografts.8
The present study discusses 8 cases of PG successfully treated with immunosuppression followed by excisional debridement and applications of Apligraf (Organogenesis, Inc), a bilayer skin substitute equivalent (BLSE). This US Food and Drug Administration–approved cultured, allogeneic, BLSE comprises living keratinocytes, keratinocyte stem cells, and fibroblasts.9 While further studies are necessary to fully understand the complexities involved, the aim of the present series is to contribute valuable insights into improving wound care strategies for this challenging condition by analyzing clinical outcomes.
Methods
A retrospective review of all patients diagnosed with PG was created over the last 5 years. We reviewed patients from 2020 to 2024 retrospectively using EPIC electronic health recorder’s SlicerDicer to collect the initial dataset of 38 patients. The authors then systematically combed through patient charts to identify who received this BLSE and subsequent wound debridement. Patients’ demographic history, smoking history, comorbidities, wound area prior to debridement, date of surgeries, date of BLSE application, and subsequent follow-up wound area were identified.
Histopathologic evaluation was performed in 7 of the 8 patients included in this case series. Tissue biopsies were obtained either before or during the course of treatment to support the diagnosis of PG and to exclude alternative etiologies such as infection or malignancy. Biopsy findings were heterogeneous and nonspecific, consistent with the known histopathologic variability of PG, and included ulceration with mixed inflammatory infiltrates, prominent neutrophils, fibrinopurulent exudate, pseudoepitheliomatous hyperplasia, granulation tissue, and features of stasis dermatitis. No biopsies demonstrated evidence of malignancy. One patient (patient 7) underwent wound culture without tissue biopsy. Overall, biopsy findings were interpreted in conjunction with clinical presentation and response to immunosuppressive therapy to establish the diagnosis. All patients were receiving systemic corticosteroid therapy at the time of surgical debridement and/or application of BLSE. Initial prednisone-equivalent doses ranged from moderate- to high-dose therapy during periods of active disease, typically between 0.5 mg/kg/day and 1.5 mg/kg/day, depending on disease severity and patient comorbidities. Prior to surgical intervention and application of BLSE, patients demonstrated clinical stabilization of their inflammatory disease, with reduction in pain, erythema, and wound edge inflammation. Corticosteroid doses were subsequently tapered as clinically tolerated following improvement in wound healing and in conjunction with adjunctive immunosuppressive or biologic therapy.
Results
All patients
A total of 8 patients with biopsy-proven or clinically diagnosed PG were included in this case series. Patient demographics are summarized in Table 1 and include age, sex, and body mass index (BMI). The median age was 63 years (range, 29 years-87 years), and the cohort included 5 females and 3 males. BMI varied widely, with 4 patients classified as having obesity (BMI ≥30), including 2 with morbid obesity.
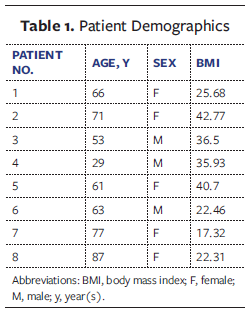
The cohort consisted of patients with a variety of comorbidities, as detailed in Table 2. Hypertension was present in 7 of 8 patients, and 6 patients had vascular disease, including peripheral artery disease, chronic venous insufficiency, and atherosclerosis of lower extremities. Three patients had type 2 diabetes, and 4 had underlying autoimmune or inflammatory conditions such as rheumatoid arthritis, ulcerative colitis, or Antineutrophilic cytoplasmic autoantibodies (ANCA)-associated vasculitis. None of the patients were active smokers at the time of treatment; all were either former smokers or had never smoked.
Seven of 8 patients underwent tissue biopsy as part of their diagnostic evaluation. Histopathologic findings varied across cases and included ulceration with neutrophilic and mixed inflammatory infiltrates, fibrinopurulent exudate, pseudoepitheliomatous hyperplasia, granulation tissue, and features of stasis dermatitis. These findings were consistent with reported patterns in PG and helped exclude infectious or malignant processes. One patient (patient 7) was diagnosed clinically and underwent wound culture without biopsy.
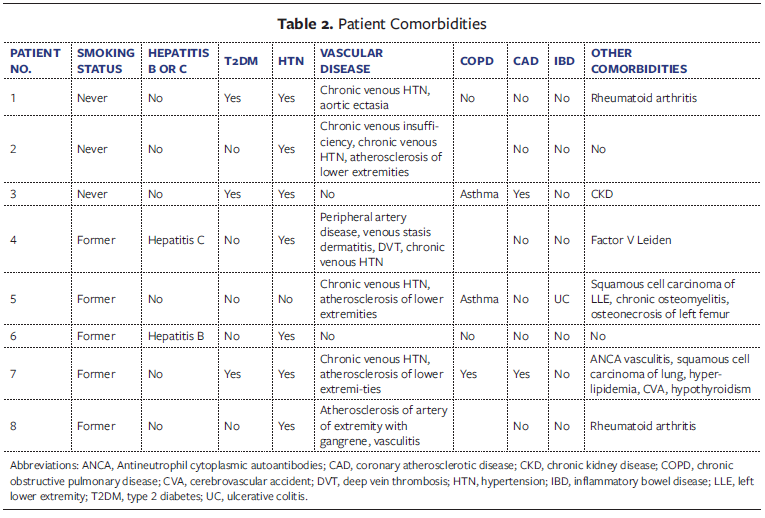
Treatment regimens prior to surgical debridement are outlined in Table 3. All patients received systemic corticosteroids during the active disease phase, with prednisone being the most common. Topical corticosteroids along with additional immunosuppressive or biologic agents were used in 5 of 8 patients. This included cyclosporine, tacrolimus, adalimumab, and dupilumab, typically tailored to the patient’s underlying disease.
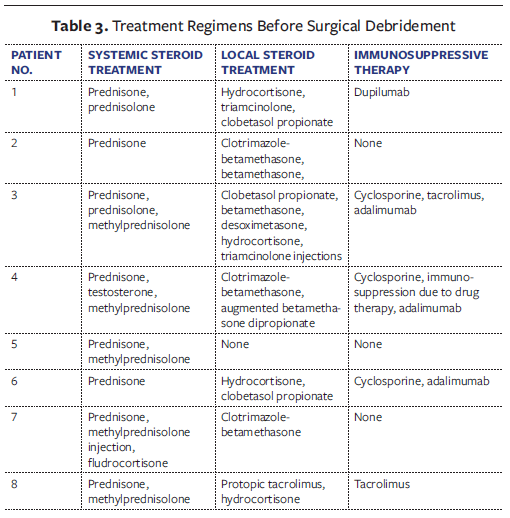
Inflammatory markers and hemoglobin A1c (HbA1c) levels were studied (Table 4). Erythrocyte sedimentation rate (ESR) (range, 39 mm/h-97 mm/h) and C-reactive protein (CRP) level, which suggests the systemic inflammation consistent with active PG, were also evaluated. White blood cell counts remained within normal limits across cases, and HbA1c levels ranged from 5.0% to 9.4%, with 1 patient showing poorly controlled diabetes. Only 1 patient tested positive for autoantibodies (anti-Smith); 1 patient tested positive for ANCA, myeloperoxidase, and hydralazine-induced lupus; and 1 patient tested positive for rheumatoid factor, potentially pointing to an autoimmune component. These findings support the complexity of PG pathophysiology and suggest that BLSE was used across a heterogeneous patient population, including some with metabolic or autoimmune dysregulation.
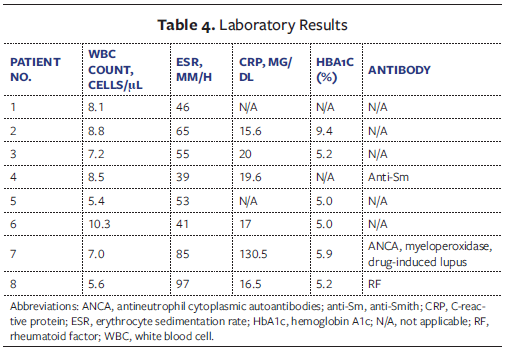
All patients underwent surgical debridement, with a median of 6 procedures (range, 1-11) (Table 5). BLSE was applied in 8 patients following debridement, with between 1 and 6 applications (median, 2). In 2 cases, adjunctive therapies such as split-thickness skin grafts or biologic wound matrices (eg, PuraPly; Organogenesis, Inc) were also used. Transient wound enlargement post-debridement was observed in several cases, but this was followed by gradual wound contraction and healing.
Seven patients developed superimposed wound infections during treatment, confirmed either clinically or microbiologically. Wound cultures were obtained in all 8 patients during the course of treatment, primarily in the setting of clinical concern for secondary infection. Positive cultures were common and demonstrated a range of organisms typical of chronic lower extremity wounds. Identified pathogens included Staphylococcus aureus (patients 2, 4, and 7), Pseudomonas aeruginosa (patients 3, 4, and 5), Enterococcus faecalis (patients 1 and 8), Escherichia coli (patient 1), group B Streptococcus (Streptococcus agalactiae, patient 5), coagulase-negative Staphylococcus species (patient 6), and Alcaligenes xylosoxidans (patient 8). These infections were managed with targeted antimicrobial therapy and did not preclude eventual wound healing in any patient. Elevated inflammatory markers were common: median ESR was approximately 52 mm/h, and CRP levels exceeded 10 mg/dL in most cases, reflecting ongoing systemic inflammation. Table 5 highlights the number of BLSE applications in each patient along with the number of debridements they received. All patients achieved complete wound healing by 50 weeks.
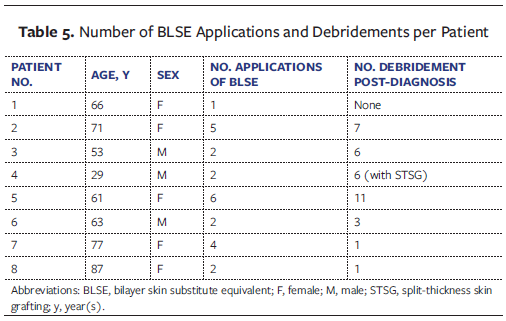
Despite the complexity of the cases and the presence of multiple comorbidities, all 8 patients ultimately achieved complete wound closure. Healing timelines varied, ranging from several weeks in mild cases to longer than 6 months in more severe, refractory wounds. An association between initial wound area and time to complete healing was observed. Patients with a larger baseline wound area generally required a longer duration to achieve full epithelialization, whereas smaller ulcers healed more rapidly. Figure 1 summarizes the wound area in centimeters squared per patient at 10-week intervals from week 0 through week 50 after debridement. Patient 8 experienced an increase in the wound area after debridement, but for the other 7 patients the wound area substantially decreased during that time period.
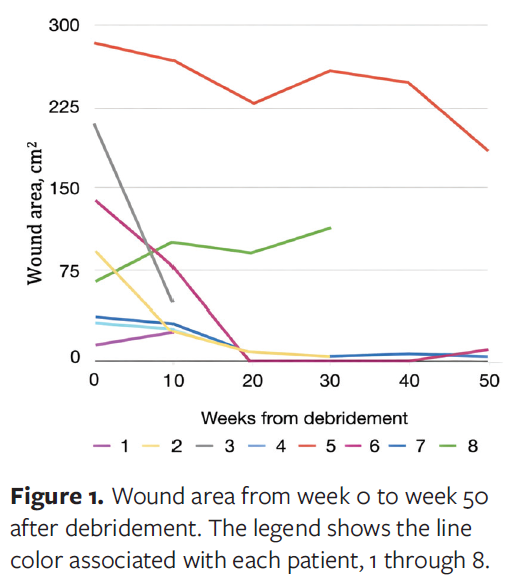
Patient 3
Patient 3, a 53-year-old male, presented to the outpatient wound center on March 1, 2023, with an 8-month history of nonhealing ulcer of the left leg and a presumptive diagnosis of PG (Figure 2). At presentation, the patient was on a prednisone taper. The initial dosage was 1.5 mg/kg, but because of the patient’s insulin-dependent diabetes, it was not tolerated due to hyperglycemia. The patient had prior hyperbaric oxygen therapy, since hyperbaric oxygen therapy has been shown to alleviate symptoms in Chron’s disease phenotypes, without significant improvement of the wound.10 He was treated with antibiotic therapy as well as topical wound therapies, with slight improvement of the wound. A recent magnetic resonance image of the left lower extremity revealed periostitis. The bone scan was negative for osteomyelitis. Tissue biopsy was negative for malignancy. The rheumatologic workup was nondiagnostic. Noninvasive vascular studies were negative for peripheral arterial disease or venous insufficiency. The patient’s past medical history is significant for uncontrolled diabetes, asthma, and hypertension, and his surgical history is significant for bilateral tympanostomy tubes and laminectomy.
Physical examination showed a wound on the midanterior leg with intermittent granulation tissue, slough, and rolled wound edges. The wound measured 6.4 cm × 4.7 cm × 0.2 cm. As of April 26, 2023, no significant change in the wound was noted with conservative management. Biopsy was inconclusive.
The patient was referred to dermatology for evaluation before any surgical debridement. Treatment with gentian violet, betamethasone, and intralesional triamcinolone injections was initiated at that time. Pain symptoms improved; however, there was no significant progress in wound healing. On May 18, 2023, the wound dimensions were 6.7 cm × 5.1 cm × 0.2 cm.
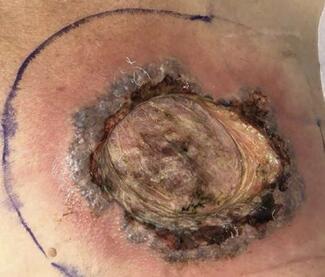
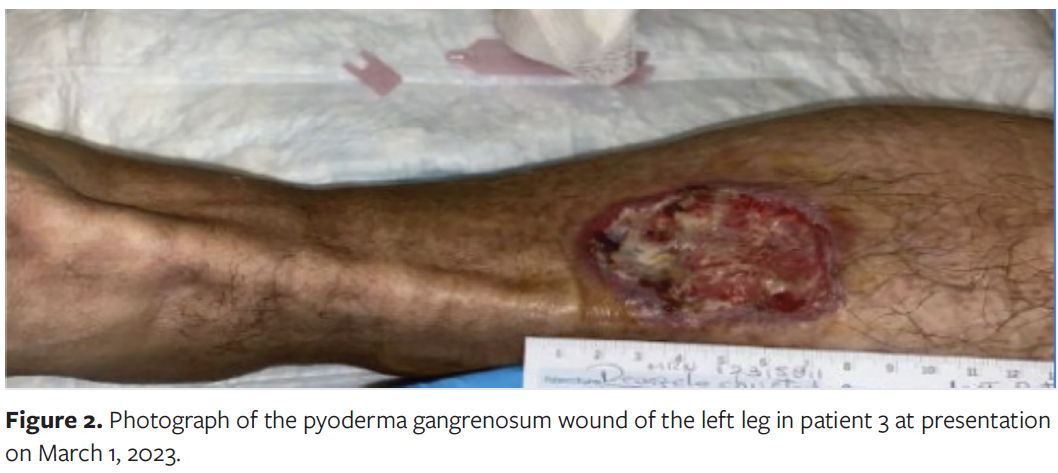
On May 26, 2023, the patient presented with swelling, worsening pain, and necrosis of the wound. Debridement of the epidermis, dermis, and subcutaneous tissues was performed. The post-debridement wound measured 7.2 cm × 5.2 cm × 0.5 cm. The patient was started on adalimumab for 1 month, but the wound continued to deteriorate. This is evident from his July visit before surgical debridement (Figure 3).
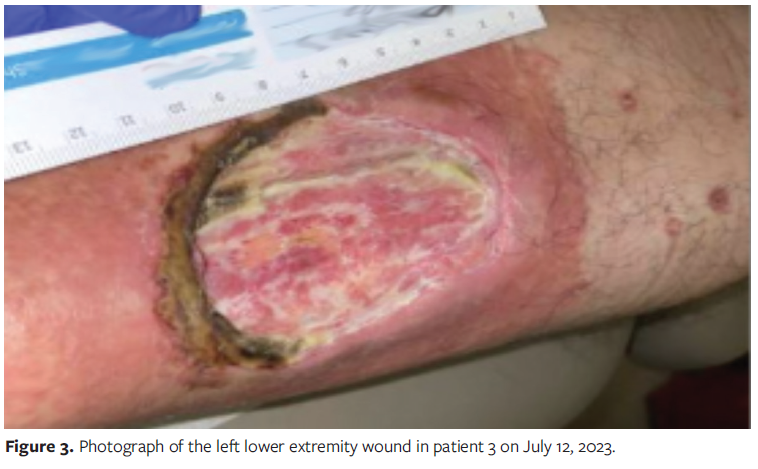
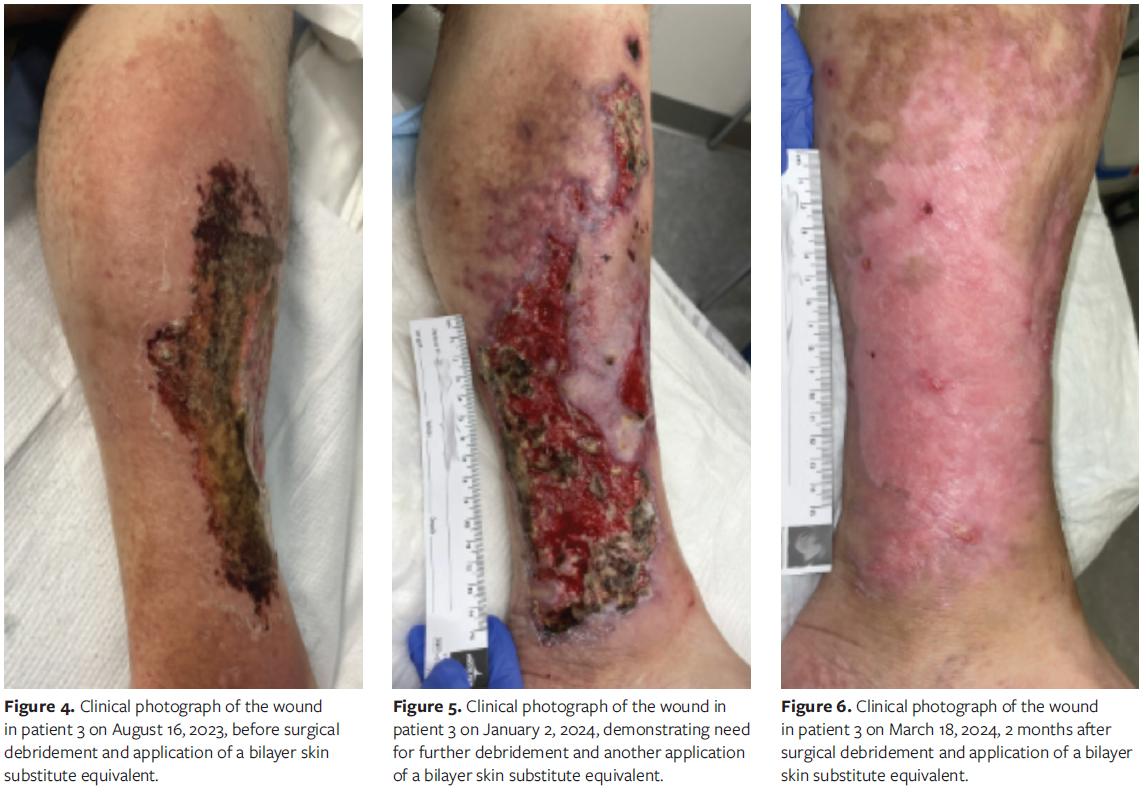
He was then switched to cyclosporine and dapsone. Because there was no significant improvement with topical treatment, dermatology specialists initiated tumor necrosis factor (TNF) inhibitor treatment, betamethasone, and gentian violet. The wound continued to progressively worsen. The patient developed cellulitis and was admitted to the hospital for intravenous antibiotic therapy and pain management. It was decided to perform excisional surgical debridement and apply BLSE. Figure 4 demonstrates how the wound looked prior to his debridement and application of BLSE in August 2023. Postoperatively, the wound was without significant local inflammation and began to contract and epithelialize. A second application of BLSE was performed in the clinic of the authors of the present series. Figure 5 demonstrates how the wound appeared prior to the surgical debridement and application of BLSE. The wound eventually fully epithelialized and healed. Figures 3-6 highlight the progression of the patient’s wound with eventual wound healing after application of BLSE.
Discussion
This case series highlights the potential of a multimodal approach—systemic immunosuppression, conservative surgical debridement, and application of BLSE—in managing complex cases of PG. While debridement historically has been avoided due to the risk of pathergy, the findings of the present case series support its selective use when the underlying inflammatory state is adequately controlled. Previous studies have demonstrated that the combination of systemic corticosteroids, immunomodulators, and adjuvant wound therapies can mitigate recurrence and enable safe surgical intervention.11 Importantly, in those studies BLSE was not applied during periods of uncontrolled inflammation; all patients remained on concomitant systemic corticosteroids at stabilized doses at the time of application, supporting safe surgical intervention and minimizing the risk of pathergy. Nevertheless, PG remains difficult to treat, with reported recurrence rates as high as 46% despite optimal medical therapy.12
All patients in the present cohort were treated with systemic corticosteroids, and the majority required additional immunosuppressive or biologic agents such as anti-TNF therapies, calcineurin inhibitors, or interleukin 4 or interleukin 13 blockers. These regimens were tailored to each patient’s underlying autoimmune or inflammatory conditions.13 Importantly, patients were maintained on systemic steroids prior to the application of BLSE, and only after achieving resolution of their inflammatory status was BLSE attempted. The findings of the present series echo prior reports that suggest biologics lead to superior healing outcomes compared with corticosteroids alone, with some studies showing healing rates exceeding 90% when biologics are used.14,15 As inflammation subsided and wound healing progressed, steroid tapering was possible in several cases, which highlights the importance of timely escalation to steroid-sparing agents. BLSE was applied in 7 of 8 patients, typically after initial debridement. Its use appeared to promote faster granulation, reduce open wound time, and improve wound bed quality, especially in large or refractory ulcers. All BLSE applications were secured with a standardized multilayer dressing protocol consisting of Adaptic, abdominal pads, Kerlix, ACE wrap, and Coban, which provided stability and protection. Due to the use of a mesher, most grafts were applied at a 1:1.5 ratio, and the maximum number of BLSEs used for the largest wound was 3. Patients requiring multiple graft applications generally had larger or bilateral ulcers, but all achieved eventual closure. Of note, patients 3 and 8 initially experienced worsening of their inflammation and associated wound deterioration; however, after their inflammatory states were stabilized and BLSE was subsequently applied, both achieved complete wound resolution by week 50.
Unlike autografts, BLSEs avoid the need for a donor site, thereby minimizing the risk of inducing additional lesions through pathergy. The use of cellular- and/or tissue-based products in the management of PG has been increasingly reported as a strategy to promote wound healing while reducing donor site morbidity. Chan et al13 described a case series in which patients with PG underwent surgical debridement and treatment with cellular- and/or tissue-based therapies, including fetal bovine dermis, resulting in pain reduction, improved granulation tissue formation, and durable wound coverage when used in conjunction with systemic immunosuppression. These findings support the role of biologic wound matrices as effective adjuncts to medical therapy, particularly in complex or refractory PG wounds. The results of the present series similarly demonstrate that advanced cellular tissue products, when applied after adequate inflammatory control, can facilitate wound contraction and epithelialization without exacerbating disease activity. These outcomes are consistent with prior reports on living cellular skin substitutes in PG. For example, Duchini et al14 reported complete healing of a refractory PG ulcer following multiple applications of a bilayer skin substitute.
Superimposed wound infections were common in the present series, affecting 7 of 8 patients. Although wound cultures were frequently positive, these findings were interpreted as secondary colonization or superimposed infection rather than an alternative diagnosis, because all patients demonstrated clinical features consistent with PG and ultimately responded to immunosuppressive therapy and advanced wound care. Although PG is not inherently infectious, prolonged immunosuppression and chronic ulceration likely increased susceptibility. Nonetheless, with appropriate antimicrobial therapy, infections were managed without impeding overall healing.
Taken together, the results of the present series support a treatment algorithm that begins with early immunologic control and is followed by staged conservative debridement and adjunctive use of cellular tissue products. This integrated approach challenges traditional surgical avoidance in PG and aligns with emerging evidence that controlled debridement—when combined with biologic therapies and advanced wound care—can significantly improve healing outcomes.13,14 Future studies may further clarify optimal timing, graft application frequency, and long-term recurrence risk in surgically managed PG.
Limitations
This case series is limited by a small sample size of 8 patients, a retrospective design, and lack of a control group. This restricts the generalizability of the findings and the ability to draw firm conclusions about the success of BLSE in PG. Additionally, heterogeneity in immunosuppressive regimens introduces a confounding variable, because the interaction between BLSE and specific medications remains insufficiently characterized. Although the dressing protocol was standardized, the small cohort prevents definitive conclusions about its reproducibility or comparative effectiveness. Similarly, the use of a mesher at a 1:1.5 ratio and the maximum of 3 BLSE per wound represent procedural choices that may not extrapolate to larger populations. Finally, although patients 3 and 8 demonstrated resolution after initial worsening of inflammation, the limited number of such cases precludes broader insights into the timing and safety of BLSE initiation following inflammatory flares. Future prospective studies with larger cohorts are warranted to validate these observations.
Conclusion
This study shows that a multidisciplinary, multimodal approach was key in the management of PG. Combining systemic therapy (eg, steroids, biologics) to control the disease, judicious surgery to manage the wounds, and innovative wound healing adjuncts such as BLSE to promote tissue regeneration were the hallmark in management of PG wounds. Surgical intervention in PG is a careful balancing act. Too much or ill-timed debridement can exacerbate the disease, but targeted debridement and grafting, done under cover of immunosuppression, can markedly speed up healing in otherwise intractable cases. In this series, the successful closure of patients’ ulcers despite severe PG underscores that even in challenging presentations, remission is achievable with persistent and comprehensive care.
Author and Public Information
Authors: Sibat Noor, MD; Soniya Singh, BS; Katherine Cordero Bermudez, MD; and Scott A. Gorenstein, MD
Affiliation: Department of Surgery, New York University Langone Hospital-Long Island, Mineola, NY
Acknowledgment: ChatGPT was used, in particular deep research and basic gpt, in various aspects of manuscript preparation for polishing the manuscript with regard to sentence structure, grammar, and verbiage.
Disclosure: The authors declare no financial or other conflicts of interest.
Correspondence: Sibat Noor, MD; Department of Surgery, New York University Langone Hospital-Long Island, Mineola, NY, 550 First Avenue, New York, NY, 10016; sibat.noor@gmail.com
Manuscript Accepted: January 14, 2026
References
- Su R, Tan Y, Peng S. Clinical characteristics of pyoderma gangrenosum: case series and literature review. Medicine (Baltimore). 2024;103(37):e39634. doi:10.1097/MD.0000000000039634
- Croitoru D, Naderi-Azad S, Sachdeva M, Piguet V, Alavi A. A wound care specialist’s approach to pyoderma gangrenosum. Adv Wound Care (New Rochelle). 2020;9(12):686-694. doi:10.1089/wound.2020.1168
- Moura RR, Brandão L, Moltrasio C, et al. Different molecular pathways are disrupted in pyoderma gangrenosum patients and are associated with the severity of the disease. Sci Rep. 2023;13(1):4919. doi:10.1038/s41598-023-31914-z
- Maronese CA, Pimentel MA, Li MM, Genovese G, Ortega-Loayza AG, Marzano AV. Pyoderma gangrenosum: an updated literature review on established and emerging pharmacological treatments. Am J Clin Dermatol. 2022;23(5):615-634. doi:10.1007/s40257-022-00699-8
- George C, Deroide F, Rustin M. Pyoderma gangrenosum: a guide to diagnosis and management. Clin Med (Lond). 2019;19(3):224-228. doi:10.7861/clinmedicine.19-3-224
- Dissemond J, Marzano AV, Hampton PJ, Ortega-Loayza AG. Pyoderma gangrenosum: treatment options. Drugs. 2023;83(14):1255-1267. doi:10.1007/s40265-023-01931-3
- Alam M, Grossman ME, Schneiderman PI, Blume RS, Benvenisty AI. Surgical management of pyoderma gangrenosum: case report and review. Dermatol Surg. 2000;26(11):1063-1066. doi:10.1046/j.1524-4725.2000.0260111063.x
- Bar D, Beberashvili I. Assessing the role of wound debridement in pyoderma gangrenosum: a retrospective cohort study. Wound Repair Regen. 2024;32(6):941-948. doi:10.1111/wrr.13219
- Dolynchuk K, Hull P, Guenther L, et al. The role of Apligraf in the treatment of venous leg ulcers. Ostomy Wound Manage. 1999;45(1):34-43.
- McCurdy J, Siw KCK, Kandel R, Larrigan S, Rosenfeld G, Boet S. The effectiveness and safety of hyperbaric oxygen therapy in various phenotypes of inflammatory bowel disease: systematic review with meta-analysis. Inflamm Bowel Dis. 2022;28(4):611-621. doi:10.1093/ibd/izab098
- Guliyeva G, Janis JE. Postsurgical pyoderma gangrenosum requiring plastic surgical intervention: a practical review. Plast Reconstr Surg Glob Open. 2024;12(1):e5505. doi:10.1097/GOX.0000000000005505
- Mlika RB, Riahi I, Fenniche S, et al. Pyoderma gangrenosum: a report of 21 cases. Int J Dermatol. 2002;41(2):65-68. doi:10.1046/j.1365-4362.2002.01329.x
- Chan MCY, James C, Patel M, Ellis S, Lantis JC. The use of cellular- and/or tissue-based therapy for the management of pyoderma gangrenosum: a case series and review of the literature. Wounds. 2021;33(6):161-168.
- Duchini G, Itin P, Arnold A. A case of refractory pyoderma gangrenosum treated with a combination of Apligraf and systemic immunosuppressive agents. Adv Skin Wound Care. 2011;24(5):217-220. doi:10.1097/01.ASW.0000397898.56121.3f
- Argüelles-Arias F, Castro-Laria L, Lobatón T, et al. Characteristics and treatment of pyoderma gangrenosum in inflammatory bowel disease. Dig Dis Sci. 2013;58(10):2949-2954. doi:10.1007/s10620-013-2762-2