Treatment of Refractory Leg Ulcer in a Patient With Lupus Undergoing Combination Therapies
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Systemic lupus erythematosus can present with refractory chronic leg ulcers, often linked to antiphospholipid syndrome or vasculopathy. Standard immunosuppressive therapy may be unsuccessful in such cases, necessitating alternative approaches such as anticoagulation and advanced wound care. Case report. This report presents the case of a 46-year-old male with systemic lupus erythematosus who developed a persistent leg ulcer on the right lateral ankle that was unresponsive to immunosuppressive treatment of more than 6 months’ duration. A short-term multimodal therapeutic approach incorporating anticoagulation, negative pressure wound therapy, and cultured epidermal homograft successfully induced robust granulation and marked epithelialization with 1 week of hospitalization, a much shorter period than the previously reported 2 months needed for full resolution with anticoagulation alone. Although in the present case complete epithelial closure was not achieved during the brief inpatient period, the wound showed near-total epithelialization with continued outpatient care, indicating rapid and stable wound recovery. This case demonstrates that adjunctive use of negative pressure wound therapy and cultured epidermal homograft appears to shorten the healing process compared with anticoagulation therapy alone. Conclusion. This case underscores the efficacy of multimodal therapy—integrating anticoagulation, negative pressure wound therapy, and cultured epidermal homograft—in rapid induction of granulation and epithelialization of refractory ulcers in a patient with antiphospholipid syndrome-negative systemic lupus erythematosus, highlighting vasculopathy as a potential mechanism amenable to such interventions.
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease characterized by multisystem involvement, including cutaneous manifestations such as malar rash and, less commonly, chronic leg ulcers. Leg ulcers in SLE are rare, with a prevalence of approximately 5% to 6% among affected patients, and these ulcers are often associated with antiphospholipid syndrome (APS) or vasculitis.1 However, in APS-negative patients, the etiology of ulcers may involve vasculopathy or hypercoagulability secondary to chronic inflammation. Standard treatment typically includes immunosuppressive agents such as corticosteroids and methotrexate (MTX), but refractory cases pose a therapeutic challenge.
Chronic full-thickness skin defects, particularly those exceeding 5 mm in diameter, often require advanced interventions such as skin grafts or bioengineered dressings due to impaired natural healing, especially in patients with rheumatologic conditions.2 Negative pressure wound therapy (NPWT) and cultured epidermal homograft (CEH) have emerged as effective adjuncts in wound management by promoting granulation tissue formation and epithelialization.3,4
This report describes a case of a chronic lupus-related leg ulcer for which multiple prior treatments were unsuccessful but that showed rapid granulation and epithelialization following a short-term, multimodal regimen integrating anticoagulation, NPWT, and CEH.
Case Report
Written informed consent was obtained from the patient presented in this report, and the study was conducted in accordance with the Declaration of Helsinki.
A 46-year-old male who was followed by rheumatology specialists for 11 years with a diagnosis of SLE presented to the Department of Plastic and Reconstructive Surgery at Kangwon National University Hospital, Chuncheon, Republic of Korea, with 2 painless, sharply circumscribed chronic ulcers measuring 3 cm × 3 cm and 2 cm × 5 cm on the right lateral ankle. No significant lower leg edema or varicose veins were observed, but mild hyperpigmentation and focal lipodermatosclerotic skin thickening were noted around the ankle. The lesion consisted of yellowish necrotic debris centrally, with surrounding erythematous tissue that had persisted for 6 months. Despite outpatient dermatologic care, the ulcer showed no improvement.
The patient’s SLE had been intermittently managed with MTX 16 mg, prednisolone (PL) 5 mg, hydroxychloroquine (HCQ) 200 mg, and supportive medications (eg, meloxicam, ranitidine). Physical examination showed no active lupus features (eg, photosensitivity, oral ulcers, rash), though mild morning stiffness (<30 minutes) and numbness were noted, with stable vital signs (systolic blood pressure, 130 mm Hg; diastolic blood pressure, 96 mm Hg; pulse, 82/min; height, 180 cm; weight, 68 kg). Laboratory findings were as follows: hemoglobin, 14.4 g/dL; platelet count, 169 × 10³/µL; erythrocyte sedimentation rate, 68 mm/h; C-reactive protein level, 1.013 mg/dL; C3, 139 mg/dL; C4, 21 mg/dL; creatinine, 0.59 mg/dL; antinuclear antibody positive (1:1280); anti-SSA/Ro positive (20.51); and negative anti–double-stranded DNA, anti-cardiolipin, and anti-β2 glycoprotein 1 (anti-β2-GP1). Urinalysis showed a protein-creatinine ratio of 0.09, and nerve conduction studies suggested polyneuropathy with absent peroneal nerve compound muscle action potential amplitude and prolonged tibial F-wave latency. Laboratory tests, including D-dimer and coagulation profiles, were within normal ranges, and the ankle-brachial index was also normal.
Skin biopsy indicated upper dermal necrosis with neovascularization and C3 deposition, suggesting lupus vasculitis or venous ulcer without definitive vasculitis.
Repeated sharp debridement had been attempted twice in the outpatient clinic, but the wound did not show healthy granulation or epithelial advancement. Given the chronicity and poor response to prior management, combination therapy was initiated during hospital admission. Because of the patient’s occupational and geographic constraints, both hospitalization and follow-up were limited. To maximize wound healing within this short admission, a focused multimodal inpatient protocol was planned. Enoxaparin (low-molecular-weight heparin) was started, and a continuous negative pressure setting of −125 mm Hg was applied for 3 days after debridement and meticulous hemostasis.
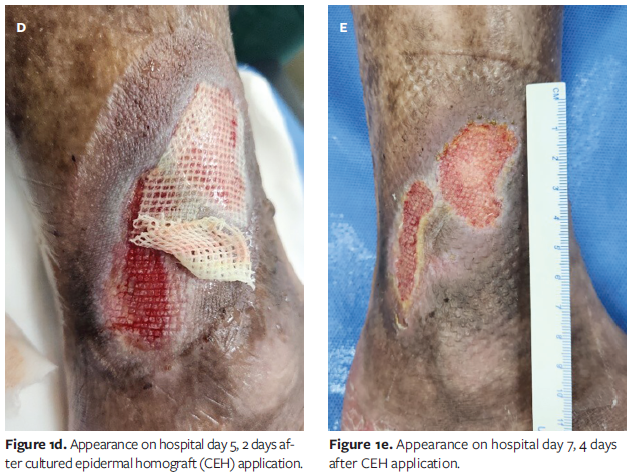
CEH (Kaloderm; Tego Science) was applied with intermittent NPWT at −80 mm Hg for 2 days. When changing the dressing on the fifth hospital day, the CEH and antibiotic ointment gauze were left unchanged, and only the outer gauze was replaced.
Results
After aggressive debridement and enoxaparin initiation, NPWT promoted granulation tissue formation by the third hospital day. CEH was applied under aseptic conditions, and NPWT was resumed. On the fifth hospital day, partial epithelialization was observed. On the seventh hospital day, all dressings were opened and NPWT was discontinued. Near-total epithelial coverage was achieved from both wound margins and the granulation bed (Figure 1).
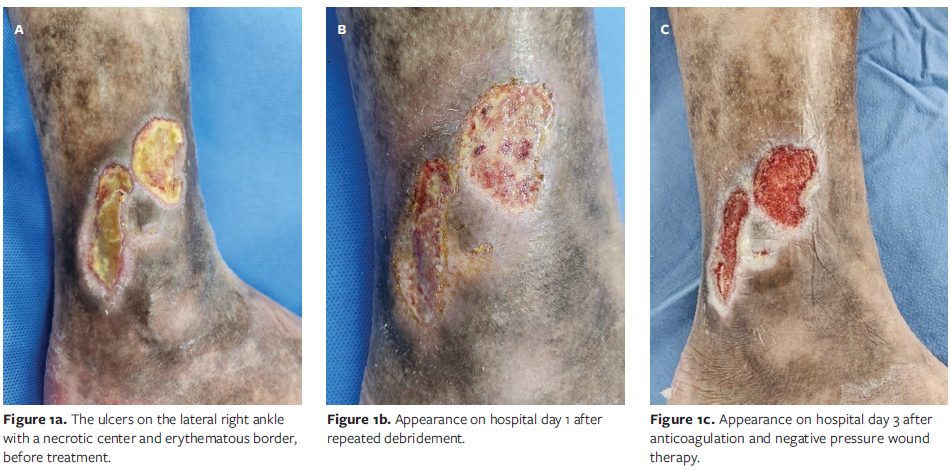
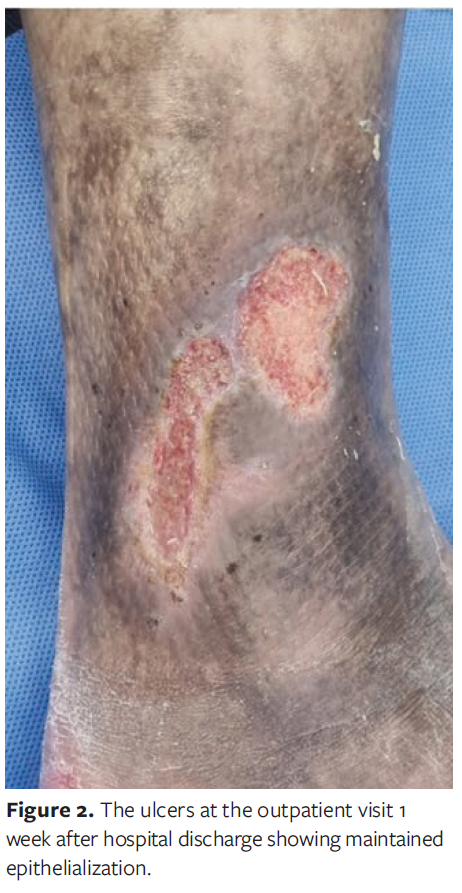
The patient was discharged with ongoing immunosuppression (MTX 16 mg, HCQ 200 mg, PL 5 mg) without additional antibiotics or anticoagulants. At an outpatient visit 1 week after hospital discharge, the ulcer remained stably epithelialized with no recurrence or exudate, indicating successful granulation induction and epithelial recovery within a markedly shortened healing course (Figure 2).
Discussion
Chronic leg ulcers in SLE are uncommon, with a prevalence of 5% to 6%, and are frequently associated with APS-related hypercoagulability or lupus vasculitis.1,2 The patient in the present report was negative for APS markers (lupus anticoagulant, anti-cardiolipin, anti-b2-GP1), suggesting alternative mechanisms such as microvascular dysfunction or vasculopathy secondary to chronic inflammation. This distinction is clinically significant because APS-positive ulcers are often driven by overt thrombosis, necessitating long-term anticoagulation, whereas APS-negative ulcers may be more amenable to therapies targeting endothelial dysfunction and tissue repair.5 In addition, owing to the patient’s occupational obligations and long travel distance to the hospital in the present case, both hospitalization and follow-up visits had to be limited. These constraints necessitated an intensive short-term multimodal treatment approach to maximize wound healing within a restricted time frame.
The role of antiphospholipid antibody positivity in SLE-related ulcers is critical in guiding treatment strategies. APS-positive patients are predisposed to thrombotic vasculopathy, leading to ischemic ulceration that responds primarily to anticoagulation. In contrast, APS-negative SLE ulcers are more likely due to immune-mediated vasculopathy, microvascular stasis, or chronic inflammatory damage, which may benefit from a multimodal therapeutic approach. This aligns with findings in previous reports in which enoxaparin alone facilitated ulcer healing by improving local circulation, even in the absence of APS. However, the present case demonstrates that integrating NPWT and CEH can substantially shorten healing time.
Unlike prior reports of anticoagulant monotherapy alone requiring approximately 2 months for complete resolution,6 the patient in the present report achieved marked epithelialization within 1 week of hospitalization through the combined use of enoxaparin, NPWT, and CEH. This rapid healing contrasts with historical data indicating prolonged recovery times for rheumatologic ulcers, often ranging from 3 months to 42 months despite active treatment.2 Hussain and Lenz6 demonstrated the efficacy of NPWT with instillation and split-thickness skin grafting in a chronic venous leg ulcer, achieving significant improvement within 2 weeks. Although their patient had a venous etiology rather than SLE, the application of NPWT to enhance granulation and microcirculation parallels the approach used in the present case, suggesting a broader applicability of this modality in complex wounds.
Although mild hyperpigmentation and focal lipodermatosclerotic changes were observed in the present case, there was no significant edema or varicosities, and the ulcers were located on the lateral aspect of the ankle rather than in the gaiter area typical of venous ulcers.7 Laboratory findings, including normal D-dimer level and coagulation profiles, further supported that venous insufficiency was unlikely in the present case. Although venous duplex ultrasonography could not be performed due to the short inpatient course, the absence of edema and the lesion’s atypical location argue against venous stasis as the primary mechanism. Taken together, these findings suggest that the ulcer was more consistent with lupus-related vasculopathy than a primary venous ulcer.
Considering the potential for impaired wound healing at the donor site in patients with SLE undergoing skin grafting, the authors of the present case planned treatment with CEH to mitigate this risk. The addition of CEH likely accelerated epithelialization by providing growth factors and extracellular matrix, as supported by Kim and Lee,3 who reported enhanced wound closure with allogeneic keratinocytes in burn injuries. Enoxaparin’s role in the present case may have addressed subclinical hypercoagulability or microvascular stasis, even in the absence of APS, a hypothesis supported by prior observations of pain reduction and epithelialization following anticoagulant use.1 NPWT, as described by Lee and Kim,4 likely improved local blood flow and reduced edema, creating an optimal wound bed for CEH application. The wound persisted for more than 6 months with conservative treatment, healthy granulation tissue was not induced with wound bed preparation, and epithelialization was not achieved by NPWT and CEH alone. In addition, reduction in wound size or contraction was not observed until the end of wound healing. Importantly, even though complete epithelial closure was not yet finalized during the limited inpatient period, the wound showed sustained epithelial progression thereafter, emphasizing therapeutic success rather than limitation. This outcome demonstrates that prompt granulation and early epithelial induction are key determinants of long-term closure, especially in autoimmune ulcers with compromised regenerative potential. Accordingly, this combination seems necessary and reproducible for selected refractory APS-negative SLE ulcers.
The synergy of these modalities differentiates the present case from typical lupus ulcers, which often align with venous or vasculopathic etiologies rather than classic vasculitis, as evidenced by the biopsy showing necrosis and C3 deposition without definitive vasculitic changes.8 Differential diagnoses such as pyoderma gangrenosum were considered but excluded due to the absence of violaceous borders or cribriform scarring and poor response to prior corticosteroids, consistent with the diagnostic challenges in lupus-related ulcers described by Reddy et al.8
Moreover, the rapid success of this multimodal regimen suggests that combining anticoagulation with advanced wound therapies may overcome the therapeutic resistance seen in immunosuppressive-
only approaches. Future studies should compare this protocol to standard care in larger APS-negative SLE cohorts to validate its efficacy and elucidate the precise mechanisms of action.
Limitations
This report has several limitations. First, it describes the outcome of a single patient and lacks a control group for comparison. Second, additional diagnostic evaluations such as venous duplex ultrasonography or repeat biopsy were not performed due to the short inpatient course and the patient’s limited availability. Third, nontuberculous mycobacterial infection was not completely ruled out, although the clinical features were inconsistent with infection. Moreover, complete epithelial closure could not be fully documented within the brief hospitalization period; however, continuous epithelial progression and stable wound coverage were confirmed during outpatient follow-up, demonstrating the effectiveness of the multimodal regimen.
Conclusion
An APS-negative patient with SLE and with refractory leg ulcers unresponsive to immunosuppression, combination therapy with anticoagulation, NPWT, and CEH achieved rapid induction of granulation and epithelialization within a short clinical window. Such strategies may provide a reproducible and time-efficient option for otherwise treatment-resistant lupus-related ulcers.
Author and Public Information
Authors: Chi Young Bang, MD, PhD; Seung Ho Lee, MD; and Kun Yong Sung, MD, PhD
Affiliation: Department of Plastic and Reconstructive Surgery, Kangwon National University Hospital, Chuncheon, Republic of Korea
Disclosure: The authors disclose no financial or other conflicts of interest.
Ethical Approval: Written informed consent was obtained from the patient for publication of this case report and accompanying images. This study was conducted in accordance with the Declaration of Helsinki.
Correspondence: Kun Yong Sung, MD, PhD; Department of Plastic and Reconstructive Surgery, Kangwon National University Hospital, Baekryeong-ro 156, Chuncheon 200-722, Republic of Korea; ps@kangwon.ac.kr
Manuscript Accepted: November 24, 2025
References
- Gönüllü Ö, Kahraman FC, Yıldırım UM. Leg ulcers in a patient with systemic lupus erythematosus and successful treatment with anticoagulant therapy. Int J Low Extrem Wounds. Published online April 18, 2024. doi:10.1177/15347346241248260
- Chia HY, Tang MBY. Chronic leg ulcers in adult patients with rheumatological diseases: a 7-year retrospective review. Int Wound J. 2014;11(6):601-604. doi:10.1111/iwj.12012
- Kim EH, Lee SH. Efficacy of cultured allogenic keratinocytes in treatment of deep second-degree burn. J Burn Care Res. 2021;42(3):533-537. doi:10.1093/jbcr/iraa191
- Lee DH, Kim YJ. Negative pressure wound therapy applied to a meshed split-thickness skin graft. Arch Reconstr Microsurg. 2016;25(2):29-36. doi:10.15596/ARMS.2016.25.2.29
- Ronicke M, Berking C, Erfurt-Berge C. Occlusive cutaneous vasculopathies as cause of chronic ulcers. J Dtsch Dermatol Ges. 2024;22(4):553-567. doi:10.1111/ddg.15276
- Hussain F, Lenz R. Coupling negative pressure wound therapy with instillation and split-thickness skin graft for a large chronic venous leg ulceration: a case report. Wounds. 2023;35(2):E59-E62. doi:10.25270/wnds/22037
- Singer AJ, Tassiopoulos A, Kirsner RS. Evaluation and management of lower-extremity ulcers. N Engl J Med. 2017;377(16):1559-1567. doi:10.1056/NEJMra1615243
- Reddy V, Dziadzio M, Hamdulay S, Boyce S, Prasad N, Keat A. Lupus and leg ulcers: a diagnostic quandary. Clin Rheumatol. 2007;26(7):1173-1175. doi:10.1007/s10067-006-0306-2

















