Hydrosurgical Debridement System Combined With Negative Pressure Wound Therapy for the Treatment of Large Chronic Wounds Caused by Polyacrylamide Hydrogel Injection for Breast Augmentation
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Wounds or HMP Global, their employees, and affiliates.
Abstract
Background. Polyacrylamide hydrogel (PAHG) is a synthetic, gelatinous chemical substance commonly used as an injectable filler in breast augmentation surgery. However, its decomposition within several years postoperative often leads to various complications, causing significant physical and psychological distress to patients. The key to treatment is thorough debridement and wound drainage. Traditional debridement methods have limitations, such as long duration, significant trauma, and incomplete debridement. A hydrosurgical debridement system offers a new approach to wound healing. Case report. This report presents 2 cases in which a hydrosurgical debridement system combined with negative pressure wound therapy was used to treat large chronic wounds of the trunk, perineum, and lower limbs caused by PAGH injection for breast augmentation. Conclusion. The hydrosurgical debridement system played a key role in the treatment of the 2 cases, resulting in a good prognosis and no recurrence at 12- to 18-month follow-up.
Polyacrylamide hydrogel (PAHG) is a colorless, gelatinous substance that in itself is nontoxic. Commonly known by the brand name Amazingel (manufactured by Intel Farrar, Ukraine) (hereafter “PAHG”), it was introduced as a facial and chest soft tissue filler in China in 1997 and was domestically produced in 2000.1 However, the acrylamide monomers produced by the long-term decomposition of the injected substance in the human body pose significant swelling and toxicity risks. They can combine with water molecules in the human body to travel and penetrate through soft tissue cavities, causing severe displacement of the injected substance, local tissue toxicity, and persistent inflammation. This can lead to various complications, such as pain, hard nodules, infection, diffuse foreign body granuloma, and changes in breast shape.1 This product was banned in China in 2006. Notably, the use of PAGH was also prohibited in other regions, including the United States, around the same period. From 1997 to 2006, PAHG was used for breast augmentation in Eastern Europe, Russia, Iran, and China, with an estimated 300,000 women undergoing the procedure.1 A considerable number of these patients—both symptomatic and asymptomatic—continue to seek medical consultation, with complications reported as late as 18 years after injection.
Common methods for removing PAGH include physical therapy, nodule puncture aspiration, unseen simple aspiration of injected substances, opening surgical incisions, and laparoscopic removal.2 The present study reports 2 cases in which a hydrosurgical debridement system (hereafter “a water knife”) combined with negative pressure wound therapy (NPWT) was used for treating large chronic wounds on the trunk, perineum, and lower limbs caused by PAGH injection for breast augmentation.
Case 1
The patient in case 1 was aged 33 years when she presented to the authors of the present study in 2013. After she gave birth in October 2003, her breasts atrophied, and she underwent PAGH injection for breast augmentation at a local plastic surgery hospital. Eight and a half years later (in 2012), she experienced pain in the left breast and side chest wall, which spread to the lower abdomen and perineum. She had bilateral breast prostheses removed at that same hospital; within 2 months, she underwent local debridement of the lower abdominal incision twice, and the wounds healed after the surgery. However, 3 months later, the affected area experienced skin breakdown, and the wounds became chronic. The patient underwent further debridement surgery, but the surgical incision did not heal, and the surrounding skin turned black and thinned. Hard nodules of varying sizes were palpable under the skin in the lower abdomen and perineum, with some nodules breaking through the skin, causing severe and persistent pain for 1.5 years.
On September 25, 2013, the patient was admitted to our department for further treatment. Her appetite and sleep were affected, and she experienced low mood, depression, and weight loss of 4 kg (8.8 lb). Physical examination revealed that the total area of the skin defect in the lower abdomen was approximately 0.8% of total body surface area. The surrounding skin of the wound had broken down to form granulation tissue, with some skin remaining and hyperpigmentation evident. Subcutaneous exploration revealed communication between the subcutaneous cavity of the lower abdomen and perineum, with yellow-white exudate accompanied by a peculiar smell, a hard and irregular texture, and obvious pain to touch.
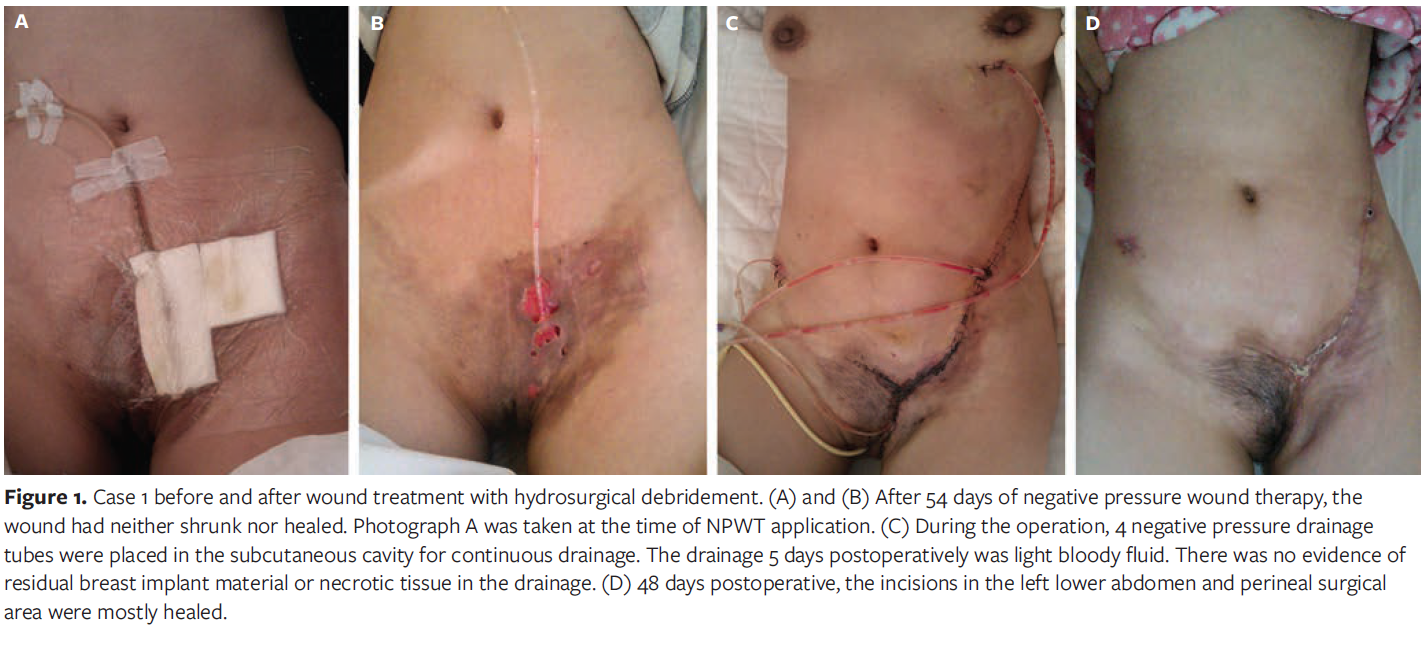
After admission, wound secretion culture revealed the presence of Enterobacter cloacae. Given the contaminated wound, unclean granulation tissue, and the presence of multiple sinuses and cavities, the patient was initially treated with anti-infection measures and NPWT for 54 days; however, the wound did not shrink or heal (Figure 1A, 1B). On November 20, 2013, the patient underwent surgical debridement under general anesthesia. Intraoperatively, a cavity was found encompassing the entire subcutaneous tissue of the anterior two-thirds of the trunk. This extended from the lower abdomen below the umbilicus to the perineum, including the labium majus pudendi and the roots of both thighs. The cavity also involved the left chest wall up to the level of the breast, extending downward to the inguinal region, anteriorly to the sternal border, and posteriorly to the inferior angle of the scapula. After the skin flap was opened, the entire deep fascial surface was found to be covered with a layer of yellow-white necrotic tissue, with an uneven surface. All necrotic tissue on the subcutaneous tissue was removed with a water knife, the original defect margin was trimmed, and the skin was sutured to close the wound. Four negative drainage tubes were inserted into the subcutaneous cavity, and NPWT was continued postoperative. The drainage tubes were gradually removed 1 by 1, and the wounds were closed (Figure 1C, 1D). After 48 days, the patient recovered and was discharged. Her sleep and appetite improved, and her weight increased by 1 kg (2.2 lb). Follow-up for 18 months revealed no recurrence.
Case 2
The patient in case 2 was aged 39 years when she presented to the authors of the present study in 2015. In 2003, she underwent PAGH injection for breast augmentation at a local plastic surgery hospital. Ten years later (in 2013), she noticed that her breasts were asymmetrical, with the left breast becoming smaller and harder, but she did not experience significant pain. She underwent bilateral breast prostheses removal at that same hospital. Five months after removal of the prostheses, redness and hardening of the skin were observed from the lower edge of the left breast to the left groin, along with some hyperpigmentation. She returned to that hospital and underwent incision drainage at the left lower abdomen and beside the bilateral breasts. However, the incision did not heal postoperatively.
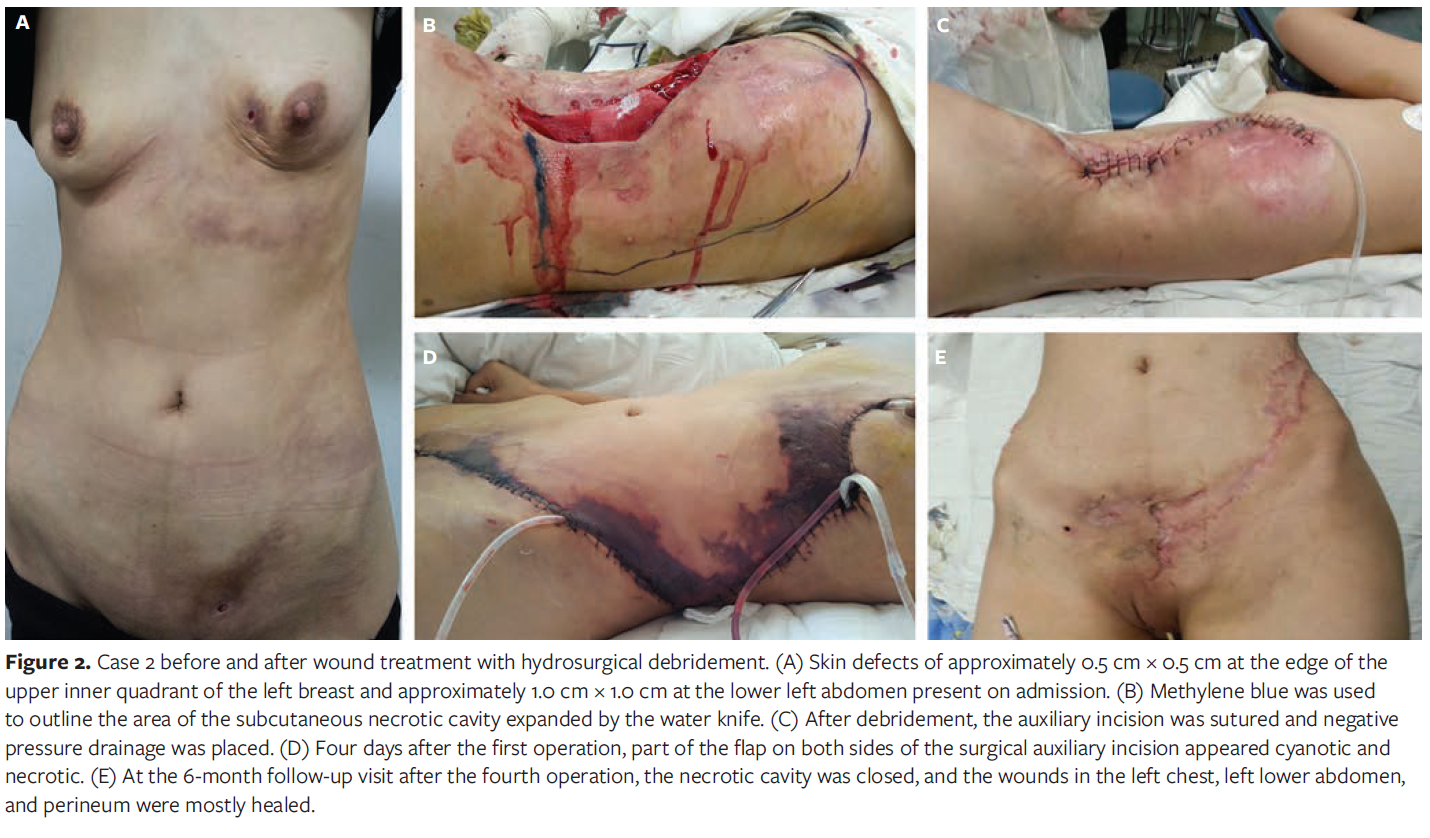
On April 24, 2015, the patient was admitted to the department of the authors of the present report with poor mental and sleep condition and unspecified weight changes. Physical examination revealed a skin defect of approximately 0.5 cm × 0.5 cm at the edge of the upper inner quadrant of the left breast, with a sinus tract forming around it and red purulent fluid flowing out at a depth of approximately 7 cm. A skin defect of approximately 1.0 cm × 1.0 cm was visible in the left lower abdomen, with the surrounding skin floating and separated from the base, forming an irregular subcutaneous cavity, with unclear depth of exploration. Hyperpigmentation was observed in some areas of the skin on the left chest, abdomen, and perineum, with local hardening upon touch and no obvious tenderness (Figure 2A).
The patient was admitted, and wound secretion culture revealed the presence of Staphylococcus epidermidis. The patient received symptomatic antibiotic treatment. On April 29, 2015, she underwent surgical debridement under general anesthesia. Intraoperatively, a cavity was found encompassing the entire area from the upper left side of the trunk to the lower left abdomen and perineum that extended upward to the upper margin of the breast, with complete separation of the left mammary tissue from the base. The cavity extended posteriorly to the left axillary posterior line and anteriorly did not exceed the edge of the left rectus abdominis muscle. It extended downward to the perineum and mons veneris.
After opening the skin flap, the bottom of the wound was found to be hard, uneven, and rough to the touch. Fat infiltration and degeneration were observed, with small abscesses forming in some areas and brownish purulent fluid overflowing. Local congestion and necrosis of the dermis were visible in some areas. The hard fibrous tissue and degenerated fat tissue on the bottom of the wound and under the skin flap were removed with a water knife (Figure 2B) until a fresh wound formed. Afterward, each flap and base were sutured intermittently to close the dead space, the surgical auxiliary incision was sutured, and 5 drainage tubes were placed under the flaps (Figure 2C). NPWT was continued postoperative, with regular replacement of the tubes. The drainage tubes were gradually removed, and all the tubes were removed after 22 days. Four days after the first operation, the flaps on both sides of the surgical auxiliary incision were partially cyanotic and necrotic, with purulent secretion (Figure 2D). After removing necrotic tissue via hydrosurgical debridement, NPWT was performed to remove necrotic tissue and cultivate granulation tissue, and skin grafts were transplanted to repair multiple skin defects. Later, the patient developed multiple recurrent subcutaneous abscesses in the trunk, and chest and abdominal exploration and debridement were performed multiple times after using NPWT. Severe adhesion of the trunk skin flaps was observed, forming multiple fibrous septa. Multiple surgeries were performed for local repair, resulting in gradual closure of the subcutaneous soft tissue cavities. The patient underwent 4 surgical procedures and was discharged 11 days after the fourth operation, after the wound had healed. Follow-up for 1 year revealed no recurrence (Figure 2E).
Disscusion
In past breast augmentation surgeries, PAHG was often injected into the posterior gap of the breast or between the chest muscles. During the years following surgery, owing to muscle traction, drastic changes in body position, and local external forces, PAHG and acrylamide monomers can infiltrate the subcutaneous tissue gaps, causing toxic effects on adjacent soft tissues of the chest, back, and abdomen. This infiltration induces persistent inflammation in tissues, leading to pathological changes such as hard nodules, subcutaneous abscesses, and foreign body granulomas.3 Additionally, the diseased tissue containing PAHG in the soft tissue gaps adheres strongly to the normal tissue, making debridement challenging. Therefore, the key to successful surgery lies in effectively removing subcutaneous foreign bodies and necrotic tissue. Traditional debridement methods are highly subjective, and for such patients, surgeons might struggle to distinguish diseased tissue from normal tissue to the greatest extent possible. Increased time and effort are required to manage tissue adhesions to ensure maximum preservation of healthy tissue. Traditional debridement is neither thorough nor easy, increasing the risk of infection and secondary surgery and imposing additional physiological, psychological, and economic burdens on patients.
Both cases in the present report involved breast augmentation using PAHG injection. In both cases, many years after injection the filler material ruptured and leaked, causing ulceration, redness, necrosis, and secretions in multiple areas of the skin. Hard nodules were palpable under the skin in several areas, and sinuses of varying depths could be probed beneath the skin defects. Ruptured and leaked PAHG caused extensive damage, affecting many tissues with deep infiltration and severe damage, making removal difficult. The treatment focused on thoroughly removing the infiltrated fillers and diseased tissue from various areas. After complete drainage, the dead cavity and sinus were closed, and the skin defect was repaired. The symptoms did not significantly improve but instead worsened after traditional debridement and drainage, and the wounds did not heal.
Two possible reasons for rupture and leakage of the injection material were considered in these cases: (1) breast augmentation had been in place for a long time, and under prolonged gravitational and external pressure the injected material ruptured and moved into the surrounding tissue gaps, and (2) the hydrophilic nature of acrylamide monomers produced by the decomposition of PAHG allows it to bind with water molecules in the body and then travel and infiltrate the lymph nodes and cavities, causing local tissue toxicity and other adverse reactions.
The use of a water knife offers significant advantages in addressing such issues. This tissue separation technology involves rapidly spraying sterile normal saline from a hydrosurgery system, selectively cutting tissue and using the Venturi effect to adsorb necrotic tissue, bacteria, and pollutants from the wound surface. It is a relatively mature new debridement method.4 In clinical use, a water knife can be chosen on the basis of the wound depth and the texture of the tissue, allowing for the removal of dead tissue by adjusting the handle direction and pressure to tailor treatment to the situation.
Matsumura et al5 used imaging analysis and reported that after debridement with a water knife, the median percentage of local necrotic tissue decreased from 50% to 0%. The median wound area and depth increased by only 0.3 cm2 and 0.5 mm, respectively, and 91% of the wounds in their study did not expand to deeper structural areas after debridement, indicating that the use of a water knife can effectively preserve active tissue. Through its unique tissue selectivity and local vacuum negative suction, a water knife significantly reduces the amount of degenerative tissue and bacteria on the wound, increases blood flow to the wound, promotes capillary regeneration and local microcirculation improvement, and accelerates the growth and repair of granulation tissue, thus shortening the wound healing time.6
Yeh et al7 reported that in cases of wounds to complex anatomical structures with abundant and dense blood vessels and nerves, such as the head, face, fingers, toes, and perineum, and other special parts, the use of a water knife is more efficient than traditional debridement in separating complex adhesion tissues, removing biofilms on the wound to the maximum extent, and reducing the risk of excessive resection of normal tissue through pressure regulation and feedback systems. The use of a water knife can maximize the preservation of viable tissue, effectively protect normal dermis, control infection, reduce scarring,8 greatly reduce the psychological burden on patients, and help patients better cope with daily life after treatment.
NPWT is recognized as a treatment method for various refractory wounds and has several advantages.9 First, NPWT effectively removes secretions and exudates, provides a closed and moist environment, creates conditions for moist healing, and prevents infection and maceration caused by excessive exudates. Second, the continuous negative pressure and local vacuum of NPWT compress the wound, reduce tissue edema, and improve local blood perfusion. NPWT stimulates the formation of new blood vessels and granulation tissue, providing a good base for epithelial migration, and induces the growth and proliferation of epithelial cells, fibroblasts, and endothelial cells, thus shortening wound healing time. Third, NPWT serves as a barrier to isolate the wound from the external environment, thereby reducing the possibility of secondary infection, decreasing the bacterial load on the wound, and disrupting the formation of a biofilm on the wound.
In both cases in the present report, a water knife was used. Intraoperatively, an auxiliary incision was made to fully expose the necrotic cavity. The flap was fully opened to thoroughly remove the hardened fibrous tissue and degenerated necrotic fat at the bottom of the wound. To the extent possible, normal tissue was preserved to form a fresh wound. Local NPWT greatly reduced the bacterial load on the wound, increased blood flow, promoted capillary regeneration and improved local microcirculation, and accelerated the growth and repair of granulation tissue on the wound. After thorough debridement and suturing of the wound, NPWT tubes were placed segmentally under the skin flap to adequately drain necrotic tissue and exudate, close dead spaces and sinuses, improve wound conditions, and promote granulation tissue growth. This was followed by skin grafting to close the wound.
In a cohort analysis study on the treatment of burn wounds with a hydrosurgery system, Legemate et al10 reported that using a hydrosurgery system for debridement before skin grafting can significantly increase the survival rate of the grafted skin. During water knife debridement, the secretion and necrotic tissue are removed, making it difficult to evaluate bleeding in the surgical area and increasing the likelihood of postoperative anemia. Therefore, preoperative blood preparation should be adequate. Intraoperatively, attention should be given to bleeding, patient blood pressure, timely hemostasis, and active correction of anemia during the early postoperative period. After debridement, the wound was washed with hypochlorous acid, which has weak acidity and strong oxidizing properties, is nonirritating to the wound, and can quickly control infection, indirectly promoting blood circulation around the wound. In case 2, after bilateral breast prosthesis removal at another hospital, severe scar adhesion was observed under the trunk skin flap, resulting in the formation of multiple fibrous septa. The treatment at that hospital was partial, with only local drainage performed, and the displacement filling was not completely removed. Multiple operations resulted in the inelasticity of subcutaneous granulation tissue and hyperplasia of surrounding fibrous tissue. Subcutaneous abscesses repeatedly formed in multiple parts of the trunk, likely due to continuous stimulation from interstitial leakage. With respect to abscess formation, the drainage time can be extended appropriately according to the drainage site. For example, if the perineal area is at the lowest position of the trunk and if leakage easily accumulates there and aggravates the invasion of the skin and subcutaneous tissue, resulting in repeated ulceration, the drainage time can be extended appropriately at that site.
Limitations
This case report has several limitations. It is a descriptive study with a small sample size, which cannot yield epidemiological measures, establish causality, or be generalized. It is also subject to bias, is susceptible to overinterpretation, may result in overemphasis on rare occurrences, raises confidentiality concerns, and has methodological limitations such as retrospective design and subjectivity.
Conclusion
Women who undergo breast augmentation should adhere to follow-up care, and appropriate treatment should be administered in the event of complications. The hydrosurgical debridement system played a key role in the treatment of the 2 cases reported herein. For the large, complex, chronic wounds in these patients, a water knife combined with NPWT was used to close the necrotic cavity and sinuses, improve wound conditions, and promote granulation tissue growth. Direct suturing or skin grafting was subsequently performed to close the wound, achieving maximum debridement and drainage, accelerating wound healing, and shortening treatment time. This method is suitable for treating complex chronic wounds caused by extensive foreign body residue, leakage, displacement, and tissue infiltration, and it is worth considering in clinical treatment.
Author and Publicn Information
Authors: Xun Xu, MS; Ruoyu Wang, MS; Honglin Wu, MS; Jinxiong Xu, MD; and Yulan Miao, PhD
Affiliation: Second Affiliated Hospital of Kunming Medical University, Wuhua District, Kunming, Yunnan Province, China
Acknowledgments: The senior authors would especially like to thank Honglin Wu, Jinxiong Xu, and Yulan Miao for their contributions to the drafting of the manuscript, and especially thank Dr Miao for his guidance.
Author Contributions: Ms. Xu and Mr. Wang contributed equally to this work.
Data Availability Statement: The data presented in the study are included in the article and the Supplementary Material. Further inquiries can be directed to the corresponding author.
Disclosure: The authors disclose no financial or other conflicts of interest.
Ethical Approval: The studies involving humans were approved by The Second Affiliated Hospital of Kunming Medical University. The studies were conducted in accordance with local legislation and institutional requirements. The participants provided written informed consent to participate in this study. Written informed consent was obtained from the individuals for the publication of any potentially identifiable images or data included in this article.
Correspondence: Yulan Miao, MD; Department of Burn Surgery, Second Affiliated Hospital of Kunming Medical University, No. 374 Dianmian Avenue, Wuhua District, Kunming, Yunnan Province, 630000, China; miaoyulanll@163.com
Manuscript Accepted: November 21, 2025
References
- Ono S, Ogawa R, Hyakusoku H. Complications after polyacrylamide hydrogel injection for soft-tissue augmentation. Plast Reconstr Surg. 2010;126(4):1349-1357. doi:10.1097/PRS.0b013e3181ead122
- Zarini E, Supino R, Pratesi G, et al. Biocompatibility and tissue interactions of a new filler material for medical use. Plast Reconstr Surg. 2004;114(4):934-942. doi:10.1097/01.PRS.0000133425.22598.D0
- Sun YH, Song JM, Wen W, Guan H, Xu MQ. Breast lesions following breast augmentation with polyacrylamide hydrogel injection. Chin J Tissue Eng Res. 2011;15(25):4623-4626. doi:10.3969/j.issn.1673-8225.2011.25.016
- Lillieborg S, Aanderud L. EMLA anaesthetic cream for debridement of burns: a study of plasma concentrations of lidocaine and prilocaine and a review of the literature. Int J Burns Trauma. 2017;7(6):88-97.
- Matsumura H, Nozaki M, Watanabe K, et al. The estimation of tissue loss during tangential hydrosurgical debridement. Ann Plast Surg. 2012;69(5):521-525. doi:10.1097/SAP.0b013e31826d2961
- Su H, Li Z, Li Y, Zhou Z, Liu Z, Guo G. Treatment of deep facial and cervical burn wounds with hydrosurgery system: six cases. Zhonghua Shao Shang Za Zhi. 2017;33(5):315-316. doi:10.3760/cma.j.issn.1009-2587.2017.05.012
- Yeh CC, Lin YS, Huang KF. Resurfacing of total penile full-thickness burn managed with the Versajet hydrosurgery system. J Burn Care Res. 2010;31(2):361-364. doi:10.1097/BCR.0b013e3181d0f4f1
- Acton C, Morris C, Gray D, et al: Consensus guidance for the use of debridement techniques in the UK. Wounds UK. 2011;7(1):77-84. doi:10.12968/jowc.2011.20.12.585.
- Nie B, Yue B. Biological effects and clinical application of negative pressure wound therapy: a review. J Wound Care. 2016;25(11):617-626. doi:10.12968/jowc.2016.25.11.617
- Legemate CM, Goei H, Middelkoop E, et al. Long-term scar quality after hydrosurgical versus conventional debridement of deep dermal burns (HyCon trial): study protocol for a randomized controlled trial. Trials. 2018;19(1):239. doi:10.1186/s13063-018-2599-2
- Nissen T, Wynn R. The clinical case report: a review of its merits and limitations. BMC Res Notes. 2014;7:264. doi:10.1186/1756-0500-7-264

















