Temporary Occlusion Embolectomy: A Cost-Effective Approach to Distal Embolic Protection in Peripheral Artery Disease
Key Summary
- In a retrospective case series (n = 104 peripheral vascular interventions [PVIs]) that evaluated embolic debris retrieval and procedural feasibility of temporary occlusion embolectomy (TOE) during complex PVI, TOE was used in 103/104 complex peripheral arterial disease cases.
- Embolectomy device delivery succeeded in 100%, with debris retrieved in 101/103 cases (98.1%). TOE added a mean 6 minutes; 97.1% of procedures were rated “Excellent” for usability.
- In this descriptive office-based lab experience, embolic debris was nearly universal during complex PVI. There were no comparator arm or quantitative particle or perfusion/limb outcomes; prospective studies are needed to assess clinical impact.
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Vascular Disease Management or HMP Global, their employees, and affiliates.
VASCULAR DISEASE MANAGEMENT. 2026;23(3):E29-E39
Abstract
Background: Embolization accompanies nearly all plaque-modifying peripheral vascular interventions (PVI) when systematically evaluated. Angiography visualizes only the major named vessels and does not assess obstructions within the distal microcirculation. These microvascular beds represent the functional endpoint of limb perfusion—where ischemia, ulceration, and tissue loss ultimately occur—and even small embolic particles may therefore carry physiological relevance beyond what is angiographically apparent. Despite this, embolic containment strategies remain selectively applied in lower extremity interventions. Temporary occlusion embolectomy (TOE) was developed as a containment-and-extraction approach designed to enable embolic removal prior to restoration of distal flow. Methods: This retrospective observational case series evaluated embolic debris retrieval and procedural feasibility of TOE during complex PVI. A total of 104 PVIs were performed across 2 office-based laboratories (OBLs) between April 2023 and August 2024, with TOE performed in 103 cases. A self-regulating tourniquet was applied distal to the lesion prior to plaque modification to achieve temporary flow arrest, maintained during angioplasty, atherectomy, and stenting. Mechanical embolectomy was then performed prior to tourniquet deflation and restoration of distal perfusion. Procedural and outcome data were recorded using standardized case forms and analyzed descriptively. Results: Of the 104 patients treated, the mean age was 71.0 years and 74.0% were male. TOE was performed in 103/104 procedures, with atherectomy utilized in 103/104 cases. Adjunctive therapy was common, including pre-atherectomy angioplasty (16.5%), post-atherectomy percutaneous transluminal angioplasty/drug-coated balloon (99.0%), and stent placement (44.0%), based on available procedural data. Embolic debris was retrieved and photographically documented in 101/103 TOE-assisted procedures (98.1%), demonstrating near-universal embolic liberation during plaque-modifying intervention when systematically assessed. TOE achieved 100.0% successful embolectomy device delivery. Lesions were highly complex and associated with elevated embolic risk, with a mean length of 304 mm, chronic total occlusions in 76.5%, in-stent restenosis in 30.4%, and moderate-to-severe calcification in 54.8%. Use of TOE added a mean of 6 minutes to procedure time, with physician-reported usability rated as “Excellent” in 97.1% of cases. Conclusions: In this real-world OBL cohort, embolic debris retrieval approached universality during complex PVI when systematically assessed. TOE enabled consistent embolic containment and removal with 100% device deliverability and minimal incremental workflow burden. These findings support further evaluation of tourniquet-based temporary occlusion as a safe and effective embolic protection strategy during lower extremity revascularization.
Introduction
Endovascular intervention, by definition, involves mechanical interaction between foreign devices and diseased arterial tissue. The passage of wires, catheters, balloons, and atherectomy systems through atherosclerotic vessels disrupts plaque and thrombotic material, liberating particulate debris throughout the procedural spectrum, from diagnostic manipulation and lesion crossing to angioplasty and atherectomy. As plaque modification intensifies, embolic generation becomes increasingly likely.
Embolization accompanies nearly all plaque-modifying peripheral vascular interventions (PVI) when systematically evaluated.1-4 When distal embolization is systematically measured, it appears nearly universal. Filter-based analyses during femoropopliteal atherectomy have demonstrated embolic debris in 100.0% of procedures, with grossly visible particulate captured in 91.5% of cases.2 Continuous translesional Doppler monitoring during superficial femoral artery intervention has likewise detected embolic signals in 100.0% of treated vessels.1 In contrast, registry-reported rates of clinically recognized distal embolization requiring bailout intervention range from approximately 1% to 5%.5,6
This discrepancy reflects differences in detection rather than differences in embolic occurrence. Most clinical registries define embolization by angiographically visible events or those requiring procedural rescue, inherently overlooking microembolization that may not produce immediate angiographic consequence. As a result, distal embolization has often been characterized as an infrequent complication of PVI, whereas direct particulate assessment suggests it is a routine procedural phenomenon.
Distal embolic protection is standard practice in select vascular territories. Current guidelines recommend embolic protection during carotid artery stenting to reduce the risk of cerebral embolization.7 In contrast, embolic containment strategies during lower extremity PVI remain inconsistently applied despite the functional importance of limb microcirculation. One-year mortality following major lower extremity amputation in peripheral arterial disease (PAD) approaches 40% to 50%,8 underscoring the clinical consequences of compromised distal perfusion.
The adverse consequences of chronic distal embolization have long been recognized in other peripheral arterial conditions. Patients with peripheral arterial aneurysms, particularly popliteal artery aneurysms, frequently present with chronic microembolization to the distal circulation, which can manifest clinically as progressive ischemia, tissue loss, or acute limb-threatening events. Contemporary Society for Vascular Surgery clinical practice guidelines highlight distal embolization as a common and clinically significant complication of popliteal artery aneurysms.9
Although the downstream effects of chronic embolization originating from peripheral endovascular interventions have been less extensively studied, the phenomenon has been described in clinical practice and observational reports.10,11 Logically, in patients with pre-existing outflow impairment—and particularly in those undergoing repeated interventions for limb salvage—preservation of distal runoff and microvascular integrity may play an important role in both early and long-term procedural outcomes.
Beyond the macroscopic runoff vessels visible on angiography, limb perfusion is governed by an extensive distal microvascular network that is highly susceptible to obstruction by embolic debris (Figure 1). Experimental and clinical studies across vascular territories have demonstrated that microembolization can impair tissue perfusion disproportionately to angiographic findings, producing persistent microvascular obstruction despite restoration of proximal arterial patency.12-14 This phenomenon, often described as “no reflow,” reflects disruption of distal arterioles and capillary beds that are not detectable by conventional angiography.
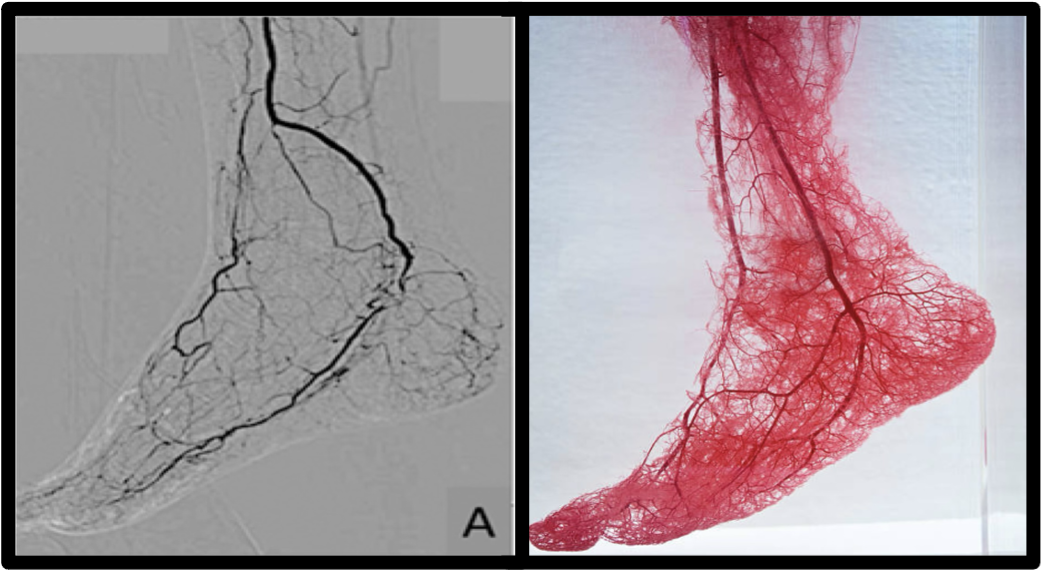
In the lower extremity, where microvascular reserve is frequently compromised by diabetes, diffuse atherosclerosis, and prior intervention, even modest embolic burden may meaningfully affect downstream perfusion. Particulate debris liberated during plaque-modifying procedures commonly falls within a size range capable of obstructing distal arterioles, suggesting that embolization may influence tissue-level perfusion even when angiographic results appear satisfactory.2,3,12 Evidence from studies of chronic limb-threatening ischemia (CLTI) further demonstrates that restoration of large-vessel patency does not necessarily normalize microcirculatory perfusion, reinforcing the importance of preserving distal microvascular integrity during revascularization.15
Temporary occlusion embolectomy (TOE) was developed as a pragmatic strategy to operationalize distal embolic containment in PVI, particularly within office-based lab (OBL) environments where procedural efficiency and economic feasibility are essential.
Temporary Occlusion Embolectomy Technique
1. Occlude: A self-regulating tourniquet is positioned distal to the lesion and inflated (~20 mm Hg above systolic blood pressure) before plaque modification.
2. Treat: Angioplasty and/or atherectomy is performed under temporary distal occlusion.
3. Remove: Mechanical embolectomy is performed prior to restoring distal flow.
This manuscript presents a retrospective case series of 104 PVIs in which TOE was performed in 103 cases. Embolic material was photographically documented in 101 of 103 TOE-assisted procedures (98.1%). The analysis evaluates embolic occurrence, technical feasibility, and workflow integration of TOE in contemporary office-based PAD intervention.
Methods
Study design and population
This retrospective observational case series evaluated embolic debris retrieval and procedural feasibility of TOE during complex PVI. A total of 104 procedures were performed across 2 OBLs by 2 treating physicians between April 17, 2023 and August 22, 2024. TOE was performed in 103 of 104 cases when clinically appropriate. The primary objectives were to document the frequency of embolic debris retrieval and assess integration of TOE into routine procedural workflows.
Procedural technique
TOE consists of temporary distal flow arrest during plaque modification followed by embolic removal prior to reperfusion. A self-regulating tourniquet was positioned distal to the target lesion and inflated to approximately 20 mm Hg above systolic blood pressure before angioplasty, atherectomy, and/or stenting. In heavily calcified vessels, inflation pressure was increased by 5 to 10 mm Hg to maintain effective flow arrest. Occlusion was maintained during plaque modification to prevent antegrade embolic propagation and permit accumulation of liberated particulate debris proximal to the tourniquet. Mechanical embolectomy was then performed before tourniquet deflation to remove embolic debris, after which distal flow was restored and final angiographic assessment completed.
RoVo mechanical thrombectomy system
Mechanical embolectomy was performed using the RoVo Mechanical Thrombectomy System (Verge Medical), which includes an aspiration syringe, rotational actuation handle, and curved-tip braided catheter. Negative pressure was generated through a valve-integrated aspiration syringe to create controlled suction within the vessel. Manual handle compressions produced catheter rotations at a 1:1 ratio. The rotating curved-tip design generates localized flow disturbance within the occluded segment, creating a vortex effect that agitates accumulated particulate debris proximal to the tourniquet. This agitation helps mobilize trapped material into suspension, facilitating more effective aspiration prior to restoration of distal flow.
Without active mechanical agitation, embolic debris may remain adherent to the vessel wall or accumulate within the catheter lumen, potentially limiting effective removal. The combined rotational and aspiration mechanism was therefore integral to the TOE workflow to promote consistent debris extraction.
Data collection and analysis
Procedural data were recorded at the time of each intervention using standardized case evaluation forms. These data were subsequently entered into a structured electronic database for aggregation and analysis. Blank fields were treated as missing and excluded from variable-specific analyses; denominators are specified in the result tables where applicable. No statistical imputation was performed. A limited number of clerical inconsistencies were corrected only when internal form logic rendered the appropriate value unequivocal. All analyses were descriptive in nature. Continuous variables are reported as mean ± standard deviation, and categorical variables are presented as counts and percentages. This retrospective analysis utilized de-identified procedural data collected during routine clinical care. All procedures were performed according to standard practice, and no additional interventions were conducted for research purposes.
Results
Summary
A total of 104 PVIs were evaluated, representing a cohort characteristic of complex PAD treatment. Patients were predominantly older adults with a male predominance; baseline demographics are summarized in Table 1.
Table 1. Patient characteristics
| Characteristic | Value | Percentage |
| Total procedures | 104 | - |
| Age, years (mean ± SD) (n = 77) | 7.10 ± 9.4 | - |
| Sex (n = 104) | ||
| Male | 77 | 74% |
| Female | 27 | 26% |
Table 1 key takeaways: The cohort reflects an elderly peripheral arterial disease population with male predominance (mean age ~71 years; ~74% male), consistent with typical patients undergoing peripheral vascular intervention; the sample represents a real-world clinical population rather than a selective screened cohort, supporting generalizability of the procedural observations. Abbreviation: SD, standard deviation.
Lesions demonstrated substantial complexity. Treated segments were frequently long (mean length 304 mm) and severe, with chronic total occlusions present in 76.5% of cases and in-stent restenosis in 30.4%. Moderate-to-severe calcification occurred in 54.8% of target vessels, creating a procedural environment associated with elevated embolic potential. Lesion characteristics are detailed in Table 2.
Table 2. Target vessel details
| Characteristic | Value | Percentage |
| Totals lesions treated (n = 164) | ||
| Superficial femoral | 89 | 54.3% |
| Popliteal | 43 | 26.2% |
| Common femoral | 6 | 3.7% |
| Tibioperoneal trunk | 6 | 3.7% |
| Posterior tibial | 6 | 3.7% |
| Iliac | 5 | 3.0% |
| Peroneal | 5 | 3.0% |
| Anterior tibial | 4 | 2.4% |
| Lesion length, mm (mean ± SD) (n = 96) | 304.1 ± 137.4 | - |
| <100 mm | 11 | 11.5% |
| 100-300 mm | 65 | 67.7% |
| >300 mm | 20 | 20.8% |
| Target vessel stenosis pre-intervention, % (mean ± SD) (n = 102) | 96.8% ± 6.4% | - |
| Chronic total occlusions | 78 | 76.5% |
| 81% to 98% occluded | 17 | 16.7% |
| <81% occluded | 7 | 6.9% |
| In-stent restenosis cases | 31 | 30.4% |
| Iliac tortuosity (n = 104) | ||
| Mild | 37 | 35.6% |
| Moderate | 44 | 42.3% |
| Severe | 7 | 6.7% |
| N/A | 16 | 15.4% |
| Calcification (n = 104) | ||
| Severe | 35 | 33.7% |
| Moderate | 22 | 21.2% |
| None/mild | 31 | 29.8% |
| N/A | 16 | 15.4% |
Table 2 Key takeaways: Treated lesions were most concentrated in the superficial femoral artery (54.3%) and popliteal artery (26.2%), with 6 additional vessels comprising the remaining ~20% of interventions, reflecting broad anatomic representation across peripheral territories; lesions demonstrated significant complexity, including a mean lesion length of 304 mm, in-stent restenosis in 30.4% of cases, and chronic total occlusions in 76.5%, consistent with advanced disease burden and elevated embolic risk; morphologic complexity was further evidenced by moderate-to-severe calcification in 54.8% of target vessels. Abbreviations: SD, standard deviation; N/A, not available.
Procedural context further reflected embolic risk. Atherectomy was performed in 103 of 104 cases, and interventions achieved effective luminal restoration based on post-intervention stenosis (Table 3).
Table 3. Procedural information
| Characteristic | Value | Percentage |
| Access approach (n = 104) | ||
| Contralateral | 86 | 82.7% |
| Antegrade | 16 | 15.4% |
| Retrograde | 2 | 1.9% |
| Access location (n = 104) | ||
| Groin | 79 | 76.0% |
| Groin + popliteal/tibial | 20 | 19.2% |
| Popliteal or tibial | 4 | 3.8% |
| Brachial | 1 | 1.0% |
| Sheath size (n = 104) | ||
| 5F | 1 | 1.0% |
| 6F | 48 | 46.2% |
| 7F | 48 | 46.2% |
| 8F | 4 | 3.8% |
| Multiple | 3 | 2.9% |
| Atherectomy performed (n = 104) | ||
| Yes | 103 | 99.0% |
| No | 1 | 1.0% |
| Atherectomy device and manufacturer (n = 103) | ||
| Abbott/Cardiovascular: Diamondback 360 | 33 | 32.0% |
| Avinger: Pantheris | 26 | 25.2% |
| BD: Rotarex | 24 | 23.3% |
| Cardio Flow: FreedomFlow | 7 | 6.8% |
| Boston Scientific: Jetstream | 6 | 5.8% |
| Medtronic: TurboHawk | 2 | 1.9% |
| Multiple devices | 5 | 4.9% |
| Other techniques performed with atherectomy PTA used pre? (n = 101) | ||
| Yes | 17 | 16.5% |
| No | 86 | 83.5% |
| PTA/DCB used post? (n = 101) | ||
| Yes | 100 | 99.0% |
| No | 1 | 1,0% |
| Stented post? (n = 84) | ||
| Yes | 37 | 44.0% |
| No | 47 | 56.0% |
| Target vessel stenosis post-intervention, % (mean ± SD) (n = 101) | 26.8% ± 21.4% | - |
| <10% | 38 | 37.6% |
| 11% to 50% | 60 | 59.4% |
| >50% | 3 | 3.0% |
Table 3 key takeaways: Procedures were performed using standard access strategies and sheath sizes, supporting real-world applicability; atherectomy was performed in 103/104 cases, creating a high embolic-risk procedural environment relevant to evaluation of protection strategies; the average stenosis post-intervention was 26.8%, down from an average 96.8% stenosis pre-intervention. Abbreviations: PTA, percutaneous transluminal angioplasty; DCB, drug-coated balloon; SD, standard deviation.
TOE integration was consistent across interventions. The technique was performed in 103 cases, with 100.0% successful embolectomy device delivery. Embolic debris was captured in 101 of 103 procedures (98.1%), a frequency consistent with near-universal embolic liberation during plaque modification. The TOE technique added a mean of 6 minutes to procedure duration, and physician usability ratings classified 97.1% of cases as “Excellent.” TOE-specific outcomes are summarized in Table 4.
Table 4. RoVo Mechanical Thrombectomy Information
| Characteristic | Value | Percentage |
| RoVo handheld used? (n = 104) | ||
| Yes | 104 | 100.0% |
| No | 0 | 0.0% |
| RoVo catheter used? (n = 103) | ||
| Yes | 71 | 68.9% |
| No | 32 | 31.1% |
| Other catheter used? (n = 103) | ||
| Yes | 32 | 31.1% |
| No | 71 | 68.9% |
| Successfully delivered to point of treatment? | ||
| Yes | 103 | 100.0% |
| No | 0 | 0.0% |
| Number of aspirations performed (n = 103) | ||
| 1 | 67 | 65.0% |
| 2 | 26 | 25.2% |
| 3 | 6 | 5.8% |
| 4+ | 4 | 3.9% |
| Visible emboli captured? (n = 103) | ||
| Yes | 101 | 98.1% |
| No | 2 | 1.9% |
| RoVo time prep to removal, minutes (mean ± SD) (n = 103) | 5.4 ± 4.1 | - |
| Total case time, minutes (mean ± SD) (n = 92) | 107.3 ± 42.5 | - |
| Physician satisfaction with RoVo (n = 103) | ||
| Excellent | 100 | 97.1% |
| Good | 2 | 1.9% |
| Acceptable | 1 | 1.0% |
| Poor | 0 | 0.0% |
Table 4 Key takeaways: Successful device delivery occurred in 100.0% of deployments, confirming procedural feasibility across anatomically complex lesion environments; embolic debris was visually captured in 101 of 103 cases (98.1%), underscoring the frequent occurrence of embolization during contemporary peripheral vascular intervention; use of the embolectomy system added an average of 6 minutes to procedure duration, indicating minimal workflow disruption; physician-reported usability ratings classified 97.1% of cases as “Excellent”, supporting favorable integration into established procedural practice. Abbreviation: SD, standard deviation.
Result photos
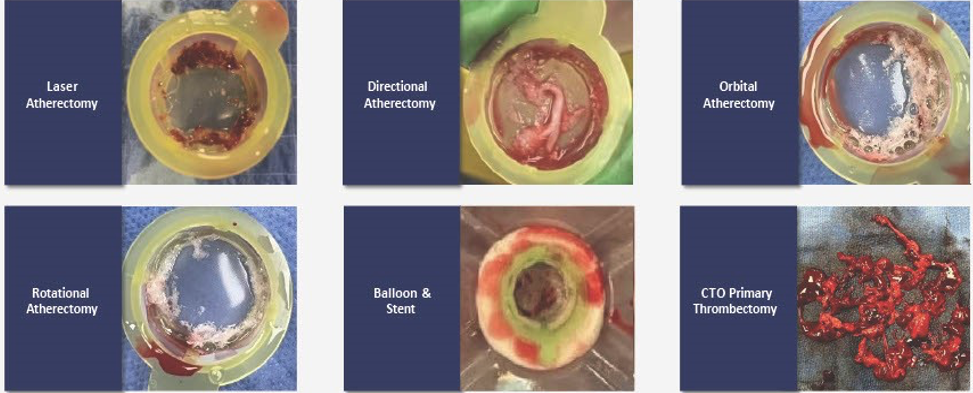
Representative images from 6 cases (Figure 2) show embolic material retrieved using the TOE technique across multiple intervention types, illustrating routine particulate liberation and the potential for distal embolic propagation without containment.
Discussion
Embolization is underrecognized
Distal embolization is a mechanistically predictable consequence of PVI. Continuous translesional Doppler monitoring during superficial femoral artery procedures has demonstrated embolic signals in 100.0% of treated vessels, occurring during lesion crossing, balloon inflation, and atherectomy alike.1 These findings confirm that embolization is procedure-intrinsic rather than device-specific.
When embolic burden is assessed directly, capture rates are consistently high. Systematic filter-based analyses during lower extremity atherectomy have demonstrated distal particulate debris in 90% to 100% of cases.2-4,16 In multiple series, visible embolic fragments measuring 1 mm or greater were identified, and sub-millimeter debris was nearly universal.2,3 In contrast, registry-reported rates of clinically recognized distal embolization requiring treatment range from approximately 1% to 5%.5,6 The marked disparity between debris capture rates approaching 100% and angiographically recognized embolization rates below 5% underscores the limited sensitivity of angiography for detecting microembolization.
In the present series, embolic debris was retrieved in 101 of 103 TOE-assisted interventions (98.1%), closely mirroring debris-based capture rates reported in the literature and substantially exceeding clinically recognized event rates. These findings reinforce that embolic liberation during complex PAD intervention is expected when systematically assessed.
Microvascular vulnerability
The physiologic significance of embolization must be considered within the context of distal microvascular vulnerability. In PAD, the microcirculation represents the functional end-organ of limb perfusion. This relationship is particularly relevant in CLTI, where impaired tissue perfusion is associated with ulceration, limb loss, and substantial mortality. One-year mortality following major lower extremity amputation in patients with PAD approaches 40% to 50%.⁸
Particle characterization studies demonstrate that embolic material liberated during PVI frequently includes debris measuring 500 µm or greater, with a meaningful proportion exceeding 1 mm.2,3,17 Particles within this size range are capable of occluding distal arterioles and small-caliber vessels. Experimental and clinical investigations across multiple vascular territories have demonstrated that microembolization can produce persistent microvascular obstruction despite restoration of proximal arterial patency, a phenomenon commonly referred to as “no reflow.”12-14 In this setting, tissue perfusion may remain impaired even when angiographic results appear satisfactory.
The lower extremity circulation may be particularly susceptible to these effects. Patients undergoing intervention for PAD frequently exhibit compromised microvascular reserve due to diabetes, diffuse atherosclerosis, and prior revascularization. Under such conditions, even modest embolic burden may influence distal perfusion by obstructing already limited microvascular pathways. Studies evaluating perfusion in CLTI further demonstrate that restoration of large-vessel patency does not necessarily normalize downstream microcirculatory flow, reinforcing the importance of preserving distal microvascular integrity during revascularization.15
Lesion complexity further amplifies embolic risk. Lesion length greater than 100 mm, chronic total occlusion, in-stent restenosis, moderate-to-severe calcification, and atherectomy-based plaque modification have all been associated with increased embolic generation.2-4,6 In the present cohort, mean lesion length was 304 mm, chronic total occlusions were present in 76.5% of cases, moderate-to-severe calcification in 54.8%, and atherectomy was performed in 103 of 104 procedures. This constellation of high-risk features provides biologic context for the observed 98.1% debris retrieval rate.
Underuse of embolic protection
Despite consistent evidence that embolic material is generated during PVI, embolic protection in lower extremity procedures has historically reflected selective, operator-driven utilization rather than routine implementation across peripheral arterial practice.18 In carotid artery stenting, embolization risk prompted integration of mandatory protection strategies into national guidelines following randomized data demonstrating stroke reduction with protected stenting.7,19 Protection devices are now considered standard of care during carotid stenting when technically feasible.
A similar evolution occurred in coronary intervention, where distal embolization was recognized as a contributor to no-reflow and periprocedural myocardial infarction.20 The randomized SAFER trial demonstrated significant reduction in major adverse cardiac events with distal protection, establishing embolic containment as routine practice in saphenous vein graft intervention.21 Subsequent coronary percutaneous coronary intervention guidelines formally recommended distal protection in this setting.22
In contrast, contemporary PAD guidelines do not recommend routine distal embolic protection during lower extremity intervention,23 despite debris-based evidence demonstrating embolic liberation in up to 90% to 100% of atherectomy cases.2-4,16 Large observational analyses have not demonstrated consistent separation in long-term endpoints such as reintervention or limb loss with embolic protection use;24 however, these analyses rely predominantly on angiographically recognized embolization or overt clinical events. When debris capture approaches 100% yet clinically recognized embolization remains below 5%,5,6 conventional endpoints may lack the sensitivity required to detect microvascular consequences.
This divergence (guideline-mandated protection in carotid and coronary territories vs selective adoption in PAD despite near-universal embolic liberation) highlights a fundamental inconsistency in vascular practice and underscores the need for re-evaluation of embolic containment strategies in complex lower extremity intervention.
Adoption barriers
The selective adoption of embolic protection in PAD appears to be driven less by biologic skepticism than by procedural and economic realities. Reviews of peripheral embolization literature cite additional device inventory, increased fluoroscopy time, and workflow complexity as barriers to routine distal filter utilization.3,4 In high-volume outpatient environments, even modest increases in procedural steps or equipment burden may influence adoption patterns.
PVI has increasingly migrated to OBLs and ambulatory settings, where procedural efficiency and economic predictability are central to sustainability.25 Analyses of outpatient PAD intervention identify device utilization and procedure duration as major contributors to cost variability.26 Conventional distal filter systems require lesion crossing, distal deployment beyond the treatment segment, and subsequent retrieval, introducing incremental device expense and additional procedural steps. In many outpatient settings, embolic protection filters may not have consistent separate reimbursement, creating financial disincentive despite biologic rationale.
TOE was developed to address these structural barriers. While TOE requires transient wire removal to accommodate rotational embolectomy and subsequent wire reintroduction, it avoids distal filter deployment and retrieval and does not leave a capture device across the treated segment. In this series, the TOE technique added a mean of approximately 6 minutes to total procedure time, suggesting limited incremental time burden.
Importantly, when performed for clinically appropriate indications, mechanical embolectomy services may align with established procedural coding pathways, whereas passive filter deployment often does not generate independent reimbursement in outpatient settings. By integrating embolic containment with active extraction, TOE may provide a workflow model that better aligns biologic protection with economic sustainability in contemporary PAD practice.
Economic considerations
The migration of PVI to OBLs and ambulatory settings has intensified focus on procedural efficiency and reimbursement alignment. In many outpatient environments, distal filter-based embolic protection devices are not associated with consistent separate reimbursement, requiring operators to absorb incremental device cost within bundled payment structures.
Mechanical embolectomy, when clinically indicated, aligns with established peripheral thrombectomy coding pathways (eg, CPT 37184–37187), whereas passive embolic protection devices typically do not generate independent procedural reimbursement in outpatient settings. Containment-and-extraction strategies that integrate embolic removal within existing procedural workflows may therefore offer a model that better aligns biologic protection with economic sustainability, particularly in high-volume PAD settings.
Temporary occlusion embolectomy as a containment-and-extraction framework
TOE represents a shift from passive embolic capture to active containment and extraction. Rather than relying on distal filtration alone, transient flow arrest during plaque modification limits antegrade embolic dispersion, and embolectomy performed prior to reperfusion enables deliberate removal of accumulated particulate burden.
This containment-before-reperfusion model reframes embolization as a predictable physiologic consequence requiring systematic management rather than selective mitigation. In complex PAD intervention, where long lesion segments, calcification, and atherectomy are common, embolization should be anticipated rather than regarded as an infrequent complication.
By integrating embolic removal within standard plaque-modification workflows and demonstrating minimal incremental procedural time in this series, TOE offers a practical mechanism for aligning embolic management with contemporary outpatient intervention. The broader implication is not device-specific, but conceptual: procedural success in PAD may need to incorporate preservation of distal microvascular integrity alongside luminal gain.
Limitations
This retrospective observational series lacks a contemporaneous comparator arm and was not designed to establish causal relationships between embolic containment and downstream clinical outcomes. Debris retrieval was visually documented but not quantitatively characterized by particle size distribution or volume. Objective measures of distal perfusion, microvascular function, or limb salvage were not systematically assessed. Additionally, findings reflect the experience of 2 operators across 2 OBLs and may not fully capture variability across practice environments. Importantly, the intent of this study was to characterize embolic occurrence and procedural feasibility within a real-world PAD population rather than to evaluate long-term outcome superiority. The consistency of debris retrieval and successful device integration observed herein establishes feasibility and biologic plausibility, but prospective validation is required to determine clinical impact.
Future Directions
Future investigation should incorporate quantitative particulate analysis, objective distal perfusion assessment, and standardized microvascular endpoints to better define the physiologic consequences of embolic containment. Comparative studies evaluating TOE against contemporary standard practice will be necessary to determine whether systematic containment-and-extraction strategies translate into improvements in limb-based outcomes. Particular attention should be directed toward patients with CLTI and poor distal runoff, in whom microvascular integrity may be most clinically consequential. As endovascular therapy continues to advance, refinement of procedural endpoints beyond luminal gain toward preservation of distal perfusion may represent an important evolution in PAD intervention strategy.
Conclusion
Distal embolization is a predictable consequence of contemporary PVI, particularly in the treatment of long, calcified, and atherectomy-modified lesions. The findings presented here reinforce the near-universal liberation of particulate debris during complex PAD intervention and underscore the disconnect between embolic generation and routine containment in current practice.
TOE demonstrated consistent feasibility, minimal incremental workflow burden, and reproducible debris retrieval across diverse lesion morphologies and treatment modalities. By combining transient distal containment with active mechanical extraction prior to reperfusion, TOE operationalizes a containment-and-extraction framework aligned with the biologic inevitability of embolization.
As PVI continues to evolve, particularly in outpatient settings, procedural success must extend beyond luminal restoration to include preservation of distal microvascular integrity. Future investigation should determine whether systematic embolic containment translates into measurable improvements in perfusion, limb preservation, and long-term outcomes. Pragmatic strategies that align biologic protection with procedural efficiency and economic sustainability may represent an important step toward more comprehensive PAD revascularization. n
To read Dr Craig Walker's commentary on this article, click here. To watch a video of the commentary, click here.
Affiliations and Disclosures
Tom Davis, MD, is from Henry Ford St. John Hospital, Detroit, Michigan; Peter Dovgan, MD, is from Space Coast Vascular, Melbourne, Florida; and Eric Dippel, MD, is from the Vascular Institute of the Midwest, Davenport, Iowa.
The authors have disclosed potential conflicts of interest regarding the content herein. Tom Davis, MD, is co-founder of 2MG; Peter Dovgan, MD, is an investor in 2MG; and Eric Dippel, MD, is a member of the Medical Advisory Board for 2MG. 2MG developed the RoVo Mechanical Thrombectomy System discussed in this manuscript. The RoVo technology is now owned by Verge Medical.
Manuscript accepted March 10, 2026.
Address for Correspondence: Tom Davis, MD, Heart & Vascular Institute, 22720 Michigan Ave #200, Dearborn, MI 48124.Email: tpdavis60@aol.com
References
1. Lam RC, Shah S, Faries PL, McKinsey JF, Ken KC, Morrissey NJ. Incidence and clinical significance of distal embolization during percutaneous interventions involving the superficial femoral artery. J Vasc Surg. 2007;46(6):1155-1159. doi:10.1016/j.jvs.2007.07.058
2. Wasty N, Khakwani MZ, Kotev S, et al. Ubiquitous nature of distal athero/thromboembolic events during lower extremity atherectomy procedures involving the superficial femoral artery. Int J Angiol. 2016;25(4):252-257. doi:10.1055/s-0036-1587694
3. Roberts D, Niazi K, Miller W, et al; DEFINITIVE Ca++ Investigators. Effective endovascular treatment of calcified femoropopliteal disease with directional atherectomy and distal embolic protection: final results of the DEFINITIVE Ca++ trial. Catheter Cardiovasc Interv. 2014;84(2):236-244. doi:10.1002/ccd.25384
4. Katsanos K, Spiliopoulos S, Reppas L, Karnabatidis D. Debulking atherectomy in the peripheral arteries: is there a role and what is the evidence? Cardiovasc Intervent Radiol. 2017;40(7):964-977. doi:10.1007/s00270-017-1649-6
5. Shammas NW, Shammas GA, Dippel EJ, Jerin M, Shammas WJ. Predictors of distal embolization in peripheral percutaneous interventions: a report from a large peripheral vascular registry. J Invasive Cardiol. 2009;21(12):628-631.
6. McKinsey JF, Zeller T, Rocha-Singh KJ, Jaff MR, Garcia LA; DEFINITIVE LE Investigators. Lower extremity revascularization using directional atherectomy: 12-month prospective results of the DEFINITIVE LE study. JACC Cardiovasc Interv. 2014;7(8):923-933. doi:10.1016/j.jcin.2014.05.006
7. Brott TG, Halperin JL, Abbara S, et al; American College of Cardiology; American Stroke Association; American Association of Neurological Surgeons; American College of Radiology; American College of Radiology; Society of NeuroInterventional Surgery; Society for Vascular Medicine; Society for Vascular Surgery. 2011 ASA/ACCF/AHA/AANN/AANS/ACR/ASNR/CNS/SAIP/SCAI/SIR/SNIS/SVM/SVS guideline on the management of patients with extracranial carotid and vertebral artery disease. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American Stroke Association, American Association of Neuroscience Nurses, American Association of Neurological Surgeons, American College of Radiology, American Society of Neuroradiology, Congress of Neurological Surgeons, Society of Atherosclerosis Imaging and Prevention, Society for Cardiovascular Angiography and Interventions, Society of Interventional Radiology, Society of NeuroInterventional Surgery, Society for Vascular Medicine, and Society for Vascular Surgery. Circulation. 2011;124(4):e54-e130. doi:10.1161/CIR.0b013e31820d8c98
8. Jones WS, Patel MR, Dai D, et al. High mortality risks after major lower extremity amputation in Medicare patients with peripheral artery disease. Am Heart J. 2013;165(5):809-815.e1. doi:10.1016/j.ahj.2012.12.002
9. Farber A, Angle N, Avgerinos E, et al. The Society for Vascular Surgery clinical practice guidelines on popliteal artery aneurysms. J Vasc Surg. 2022;75(1 S):109S-120S. doi:10.1016/j.jvs.2021.04.040
10. Kong LS, Kasirajan K, Milner R. Popliteal artery aneurysms. Endovasc Today. November 2003.
11. Rundback JH, Schneider PA, Fulton RE. Embolization during popliteal and tibial intervention for CLTI. Endovasc Today. 2022;21(5):57-61.
12. Kloner RA, Ganote CE, Jennings RB. The “no-reflow” phenomenon after temporary coronary occlusion in the dog. J Clin Invest. 1974;54(6):1496-1508. doi:10.1172/JCI107898
13. Heusch G, Kleinbongard P, Böse D, et al. Coronary microembolization: from bedside to bench and back to bedside. Circulation. 2009;120(18):1822-1836. doi:10.1161/CIRCULATIONAHA.109.888784
14. Kawarada O, Yasuda S, Nishimura K, et al. Effect of single tibial artery revascularization on microcirculation in the setting of critical limb ischemia. Circ Cardiovasc Interv. 2014;7(5):684-691. doi:10.1161/CIRCINTERVENTIONS.113.001311
15. Geskin G, Mulock MD, Tomko NL, Dasta A, Gopalakrishnan S. Effects of lower limb revascularization on the microcirculation of the foot: a retrospective cohort study. Diagnostics (Basel). 2022;12(6):1320. doi:10.3390/diagnostics12061320
16. Shammas NW, Dippel EJ, Coiner D, Shammas GA, Jerin M, Kumar A. Preventing lower extremity distal embolization using embolic filter protection: results of the PROTECT registry. J Endovasc Ther. 2008;15(3):270-276. doi:10.1583/08-2397.1
17. Shammas NW, Pucillo A, Jenkins JS, et al. WIRION embolic protection system in lower extremity arterial interventions: results of the pivotal WISE LE trial. JACC Cardiovasc Interv. 2018;11(19):1995-2003. doi:10.1016/j.jcin.2018.05.025
18. Ochoa Chaar CI, Kim TI, Shebl FM, Dardik A. The use of embolic protection devices during lower extremity peripheral vascular interventions. Ann Vasc Surg. 2020;69:9-16. doi:10.1016/j.avsg.2020.06.014
19. Yadav JS, Wholey MH, Kuntz RE, et al; Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy Investigators. Protected carotid-artery stenting versus endarterectomy in high-risk patients. N Engl J Med. 2004;351(15):1493-1501. doi:10.1056/NEJMoa040127
20. Henriques JPS, Zijlstra F, Ottervanger JP, et al. Incidence and clinical significance of distal embolization during primary angioplasty for acute myocardial infarction. Eur Heart J. 2002;23(14):1112-1117. doi:10.1053/euhj.2001.3035
21. Baim DS, Wahr D, George B, et al; Saphenous vein graft Angioplasty Free of Emboli Randomized (SAFER) Trial Investigators. Randomized trial of a distal embolic protection device during percutaneous intervention of saphenous vein aorto-coronary bypass grafts. Circulation. 2002;105(11):1285-1290.
22. Levine GN, Bates ER, Blankenship JC, et al; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines; Society for Cardiovascular Angiography and Interventions. 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. J Am Coll Cardiol. 2011;58(24):e44-e122. doi:10.1016/j.jacc.2011.08.007
23. Gornik HL, Aronow HD, Goodney PP, et al; Peer Review Committee Members. 2024 ACC/AHA/AACVPR/APMA/ABC/SCAI/SVM/SVN/SVS/SIR/VESS Guideline for the Management of Lower Extremity Peripheral Artery Disease: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149(24):e1313-e1410. doi:10.1161/CIR.0000000000001251
24. Fereydooni A, Bai H, Baril D, Chanda V, Chaar CIO, et al. Embolic protection devices are not associated with improved outcomes of atherectomy for lower extremity revascularization. Ann Vasc Surg. 2022;86:168-176. doi:10.1016/j.avsg.2022.04.045
25. Shishehbor MH, White CJ, Gray BH, et al. Critical limb ischemia: an expert statement. J Am Coll Cardiol. 2016;68(18):2002-2015. doi:10.1016/j.jacc.2016.04.071
26. Jones WS, Mi X, Qualls LG, et al. Trends in settings for peripheral vascular intervention and the effect of changes in the outpatient prospective payment system. J Am Coll Cardiol. 2015;65(9):920-927. doi:10.1016/j.jacc.2014.12.048











