Icotrokinra Demonstrates Biologic-Level Efficacy With Oral Administration in Psoriasis
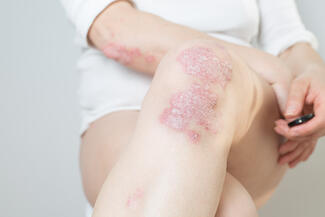
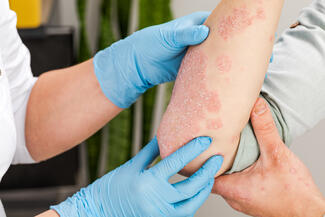
Icotrokinra, an oral interleukin-23 receptor (IL-23R) antagonist, may offer efficacy comparable to biologic therapies in moderate-to-severe plaque psoriasis, according to a comprehensive review of phase 2 and 3 clinical trials. The findings highlight a potential shift in treatment options for patients seeking effective oral therapies.
Current oral agents for psoriasis are often limited by modest efficacy or tolerability concerns, while biologics targeting IL-23 and IL-17 have established high efficacy but require parenteral administration. Icotrokinra, a first-in-class macrocyclic peptide, was designed to selectively block IL-23R, a key driver of psoriatic inflammation.
In the phase 2b FRONTIER-1 trial, icotrokinra demonstrated a dose-response relationship, with sustained efficacy observed through 52 weeks in the extension study. In the phase 3 ICONIC-ADVANCE-1 and ICONIC-ADVANCE-2 trials, icotrokinra was superior to placebo in achieving both Investigator’s Global Assessment (IGA) 0/1 and Psoriasis Area and Severity Index 90 endpoints at week 16. The therapy also demonstrated greater efficacy than deucravacitinib at week 24.
Long-term data from the ICONIC-LEAD study confirmed durable efficacy through 1 year, while ICONIC-TOTAL showed consistent benefit in difficult-to-treat areas, including the scalp, genitals, and palms/soles.
The authors noted that icotrokinra is “the first oral IL-23R antagonist to achieve high and durable levels of skin clearance with a favorable safety profile,” and emphasized its potential to overcome limitations of existing oral therapies. Adverse events were generally mild and comparable to placebo, with no new safety concerns identified.
Reference
Ferreira C, Torres T. Icotrokinra: an oral interleukin-23 receptor antagonist peptide for the treatment of psoriasis. Am J Clin Dermatol. Published online March 12, 2026. doi:10.1007/s40257-026-01019-0