Chronic Hand Eczema: From Pathogenic Subtypes to Targeted JAK and IL-4/13 Therapy
At the 2026 Masterclasses in Dermatology Annual Meeting, Emma Guttman, MD, PhD, delivered a mechanistic and practice-changing update on chronic hand eczema (CHE), reframing it as a heterogeneous inflammatory disease requiring targeted therapy.
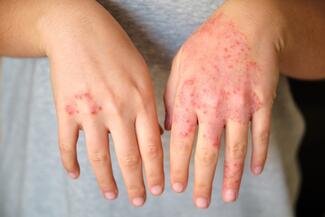
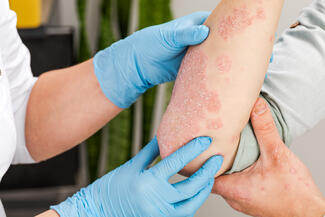
CHE affects up to 10% of the population and is the most common occupational skin disease. It is frequently chronic and recurrent, with hybrid phenotypes: approximately 50% irritant contact dermatitis (ICD), 15% allergic contact dermatitis (ACD), and 50% to 80% with atopy. More than one-third of patients have moderate-to-severe disease, and about one-third have a history of atopic dermatitis (AD).
Dr Guttman emphasized that CHE has variable pathogenesis depending on subtype. ICD demonstrates a Th1/Th17 profile; ACD can be Th1/Th17 (metals) or Th2/Th22 (rubber, fragrance); AD-associated CHE is primarily Th2/Th22. “Ideally, treatments should be directed toward primary abnormalities.”
Biomarker profiling using tape strips and RNA sequencing confirmed these endotypes. Patients with CHE and AD showed stronger Th2/Th22 skewing, whereas non-AD CHE demonstrated stronger Th1/Th17 signatures. Terminal differentiation abnormalities, such as filaggrin and loricrin, were observed regardless of AD status. Several biomarkers, including CCL17 and PDE4-related pathways, correlated with severity scores.
The therapeutic landscape is evolving. Delgocitinib 2% cream, a topical pan-JAK inhibitor, is the first US Food and Drug Administration-approved treatment for CHE. In the phase 3 DELTA trials, significantly more patients achieved Investigator Global Assessment (IGA)-CHE 0/1 and Hand Eczema Severity Index 75 at week 16 vs vehicle, with meaningful improvements in itch, pain, and Dermatology Life Quality Index scores.
Ruxolitinib cream also demonstrated significant IGA-CHE treatment success and ≥4-point Itch Numeric Rating Scale improvement in phase 2 studies. “The safety profile was also very clean,” Dr Guttman noted.
Systemic agents are under investigation for moderate-to-severe CHE. Gusacitinib (oral JAK/SYK inhibitor) improved Physician Global Assessment responses in phase 2 trials, with asymptomatic creatine phosphokinase elevations noted but no opportunistic infections, thromboembolic events, or major adverse cardiovascular events reported. However, with varied side effects with gusacitinib, Dr Guttman noted, “I’m not sure just for CHE that patients will be willing to accept these side effects.”
In AD-associated CHE, dupilumab demonstrated IGA 0/1 responses comparable to overall AD efficacy in a phase 3 hand and foot AD trial, leading to a January 2024 label expansion for patients aged 12 years and older. Tralokinumab also showed significant improvements in IGA-CHE and itch and pain scores in AD-HAND data.
Dr Guttman concluded with future directions: “We need to incorporate non-invasive biomarkers such as tape strips in the study of chronic hand eczema phenotypes (ICD/ACD/AD) and their different response to treatments in clinical trials.”
For more meeting coverage, visit the Masterclasses in Dermatology newsroom.
Reference
Guttman E. CHE and therapeutic implications. Presented at: Masterclasses in Dermatology; February 19–22, 2026; Sarasota, FL.