Cardioneuroablation and the VAFIT Protocol: Interview With José Carlos Pachón-Mateos, MD, PhD, FHRS, FEHRA
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of EP Lab Digest or HMP Global, their employees, and affiliates.
EP LAB DIGEST. 2026;26(3).
Interview by Jodie Elrod
José Carlos Pachón-Mateos, MD, PhD, FHRS, FEHRA, discusses his recent data presented at CRT 2026.
What is new or novel about the data you presented at CRT 2026 on the impact of cardioneuroablation (CNA) in atrial fibrillation (AF) ablation? How does your work advance our understanding of the autonomic nervous system’s role in AF?
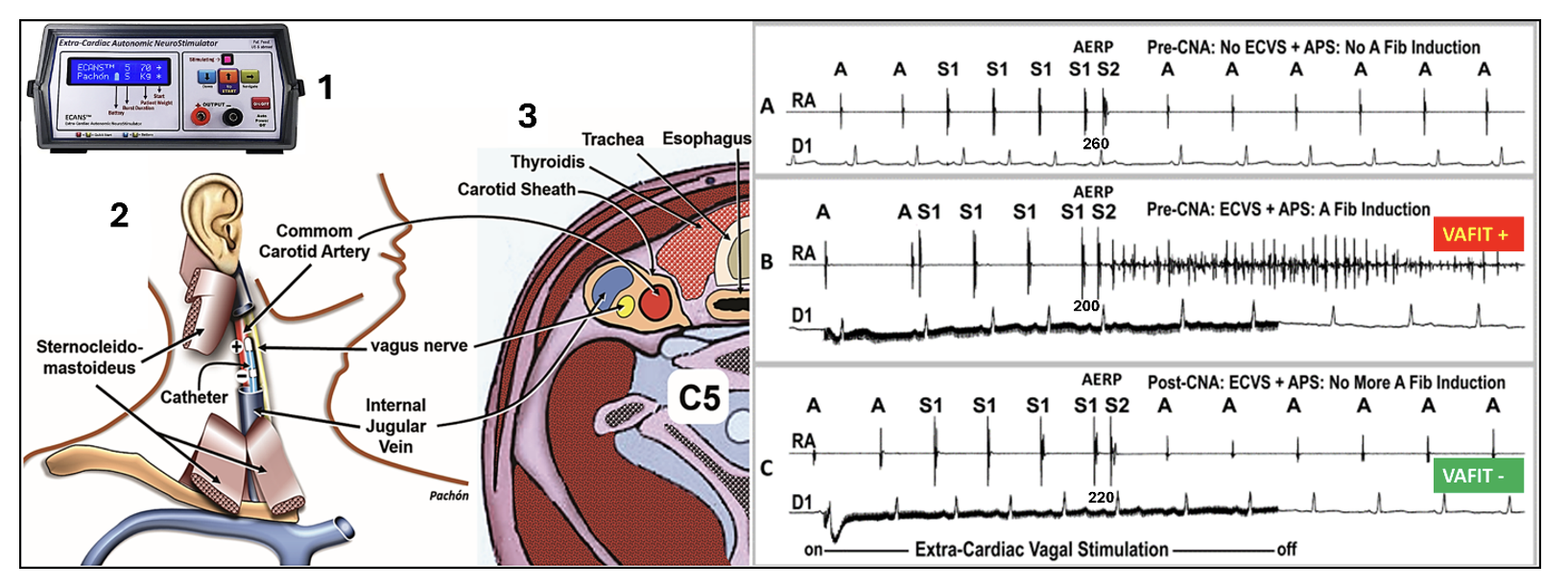
The central novelty of this study is the introduction of the Vagal AF Induction Test (VAFIT)1 as the first functional, reproducible endpoint for assessing completeness of vagal denervation during AF ablation (Figure 1). Despite decades of progress in catheter ablation, we still lack a reliable procedural endpoint beyond pulmonary vein isolation (PVI). Conventional inducibility testing with burst pacing and isoproterenol has repeatedly shown no prognostic value. VAFIT may fill this critical gap.
The concept is simple yet powerful: under normal conditions, atrial refractory period (ARP) determination (typically 200–300 ms) causes no arrhythmia (Figure 1A). However, by performing programmed atrial stimulation during extracardiac vagal stimulation (ECVS), AF can be induced with a single extrastimulus at coupling intervals far below the baseline ARP, sometimes as short as 80 ms in atria with a basal effective ARP of 260 ms (Figure 1B). This occurs because vagal activation produces a dramatic, heterogeneous reduction in atrial refractoriness through acetylcholine-mediated hyperpolarization at AF-nest sites, creating massive refractory dispersion. When CNA achieves sufficient denervation, this vagal shortening is abolished, dispersion disappears, and AF can no longer be induced, rendering the VAFIT negative (Figure 1C).
What makes this work particularly novel is the integration of VAFIT into a 3-domain vagal innervation assessment framework: sinus node domain (abolition of sinus pause), atrioventricular (AV) node domain (elimination of AV block), and, most critically, the atrial wall domain (VAFIT negativity, abolition of AF inducibility during ECVS). We now demonstrate that atrial wall denervation, measured by VAFIT, is the most clinically impactful parameter for predicting long-term freedom from AF1 (Figure 2).
This work study provides direct mechanistic evidence that vagal activity is intrinsically proarrhythmic in the atria but completely innocuous in the ventricles.2 In a substudy of 16 patients, ECVS caused dramatic ARP shortening (from ~231 ms to ~129 ms) and induced AF in every case, while ventricular refractoriness remained unchanged (280.5 vs 279.7 ms; p=0.91). This differential effect is explained by the near-absence of Kir3.1 potassium channels in ventricular myocardium, the effectors of muscarinic receptor-mediated hyperpolarization, providing molecular confirmation that CNA carries no risk of ventricular refractoriness.
Can you summarize the key findings from your research and the main take-home messages for clinicians?
In a prospective cohort of 228 patients with paroxysmal AF who underwent PVI combined with CNA, the key findings are compelling1:
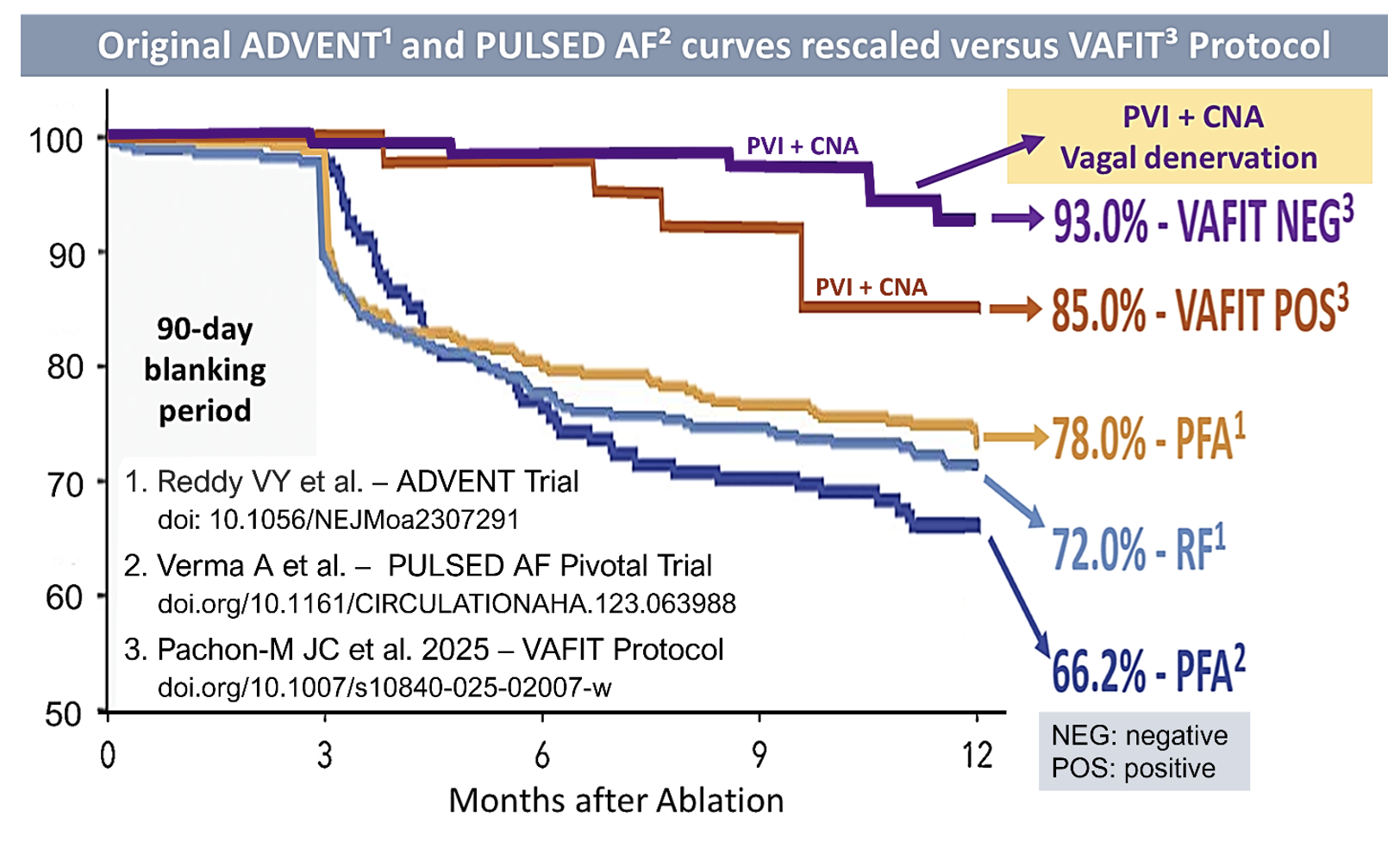
VAFIT-negative status was achieved in 79.8% of patients. Those who achieved it had a dramatically lower AF recurrence rate of 3.8%, compared to 19.6% in the VAFIT-positive group (OR 6.08, p=0.001). On multivariate analysis, a positive VAFIT was independently associated with AF recurrence (adjusted HR 3.80, p=0.011). Postprocedure heart rate increased by 20.3% (p<0.0001), confirming effective vagal denervation (Figure 2).
Perhaps the most striking finding is the 12-month success rate of 93% in the VAFIT-negative group. Acknowledging the limitations of cross-trial comparisons, this contrasts with the ADVENT trial (78% with pulsed field ablation [PFA], 72% with radiofrequency [RF]) and PULSED AF (66.2%), all evaluating PVI alone. Our data suggest that when VAFIT remains positive after PVI, the addition of CNA to render it negative provides substantial benefit beyond PVI (Figure 2).
Based on your evolving experience and the data you presented at CRT 2026, how do you see CNA reshaping standard AF ablation protocols, and what unanswered questions remain for future trials?
I believe we are approaching a paradigm shift in how we conceptualize AF ablation. For 2 decades, the field has focused almost exclusively on electrical isolation, disconnecting PVs from the atrial substrate. But our data show that AF ablation rests on 2 complementary pillars: PVI and neuroatrial isolation (NAI). PVI addresses the trigger; CNA addresses refractoriness dispersion and substratum (AF-nests5) and both (PVI + NAI) addresses resonance and microreentry.
In practical terms, I envision CNA becoming an adjunctive step in AF ablation, compatible with all existing technologies. Whether the operator uses RF, cryoballoon, or PF energy for PVI, CNA can be performed afterward, targeting ganglionated plexi, the P area of the interatrial septum, and AF-nests using conventional RF or dual-energy, when VAFIT remains positive. Notably, while PFA spares extracardiac structures such as the esophagus and phrenic nerve, it may also spare vagal fibers within atrial fat pads, potentially producing less denervation than RF. This could paradoxically leave a higher proportion of patients VAFIT-positive after PFA alone, making the CNA step even more critical in the PFA era. Ideally, future PFA energy parameters would be tuned to also eliminate vagal innervation.
The VAFIT protocol itself could reshape procedural workflow. Currently, the decision to perform additional substrate modification after PVI is often empirical. With VAFIT, the operator has an objective tool: perform VAFIT after PVI; if positive, proceed with CNA rendering VAFIT negative. This introduces a rational, reproducible decision-making framework.
VAFIT in Redo Procedures
The VAFIT is particularly valuable in redo ablations. Often, all PVs are found to remain isolated, raising the question of what additional intervention to pursue. Our workflow begins with ECVS to assess residual vagal innervation, followed by VAFIT-guided CNA to render it negative, while confirming PVI integrity. Additionally, in redo procedures AF induction frequently yields transitory AF that organizes into atypical flutter or a background tachycardia, which should be specifically mapped and ablated for optimal results.
The most pressing need is a randomized controlled trial comparing PVI alone versus PVI+CNA with VAFIT guidance. While our prospective data are encouraging, a randomized design is essential to confirm that the observed benefit is attributable to CNA-mediated denervation rather than selection bias or additional lesion burden.
Take-home messages for clinicians
- PVI alone, regardless of energy source, leaves a significant autonomic substrate untreated. The vagal nervous system drives AF through its effect on atrial refractoriness dispersion and ignoring it may explain a considerable proportion of recurrences.
- CNA can be added to any PVI technique, RF or PFA, and the VAFIT provides an objective, reproducible endpoint to guide denervation. This transforms autonomic modulation from an empirical add-on into a measurable, goal-directed therapy.
- CNA is safe. There is no ventricular proarrhythmic risk, no significant complications beyond standard ablation, and the learning curve for experienced operators is short.
Achieving VAFIT-negative status should be a procedural goal. When VAFIT remains positive after PVI, additional CNA targeting AF-nests and ganglionated plexi can be pursued to improve long-term outcomes.
In summary, cardioneuroablation guided by VAFIT represents an evolution from “PVI-only” to “PVI-plus-denervation” in AF ablation. The data we presented at CRT 2026 provides the strongest evidence yet that this approach can meaningfully improve outcomes and that the autonomic nervous system deserves a central role in our ablation strategies.
References
1. Pachon-M JC, Pachon-M EI, Santillana-P TG, et al. Vagal AF induction test (VAFIT): a new endpoint for optimizing atrial fibrillation ablation through cardioneuroablation. J Interv Card Electrophysiol. 2025;68(2):293-306. doi:10.1007/s10840-025-02007-w
2. Pachon-M JC, Pachon-M EI, Pachon CT, et al. Impact of vagal effect on atrial and ventricular refractoriness and atrial fibrillation: a comparative analysis before and after cardioneuroablation. Heart Rhythm. 2025 Oct 13:S1547-5271(25)02951-0. doi:10.1016/j.hrthm.2025.10.011
3. Reddy VY, Gerstenfeld EP, Natale A, et al. Pulsed field or conventional thermal ablation for paroxysmal atrial fibrillation. N Engl J Med. 2023;389(18):1660-1671. doi:10.1056/NEJMoa2307291
4. Pachon M JC, Pachon M EI, Pachon M JC, et al. A new treatment for atrial fibrillation based on spectral analysis to guide the catheter RF-ablation. Europace. 2004;6(6):590-601. doi:10.1016/j.eupc.2004.08.005
5. Verma A, Haines DE, Boersma LV, et al. Pulsed field ablation for the treatment of atrial fibrillation: PULSED AF pivotal trial. Circulation. 2023 May 9;147(19):1422-32. doi:10.1161/CIRCULATIONAHA.123.063988










