UCLA Researchers Develop Smart Stent to Track Blood Flow in Real Time
Implantable, self-powered device could lead to early detection of post-angioplasty artery renarrowing
Implantable, self-powered device could lead to early detection of post-angioplasty artery renarrowing
A multidisciplinary UCLA research team has developed a battery-free, magnetoelastic smart stent designed to track blood flow continuously after angioplasty, potentially enabling earlier detection of complications.
The researchers outlined their findings in a study published in Nature Cardiovascular Research and featured on the journal’s February cover.
Early restenosis often produces no symptoms and can go undetected without follow-up imaging, potentially leading to additional procedures and more serious health complications.
Current predominant methods for restenosis detection, including computed tomography angiography scans, ultrasounds and other imaging techniques, require proactive clinic visits and may present challenges for patients unable to access such care. Delays in early detection may lead to serious complications, such as the recurrent buildup of arterial plaque at the stent site or a heart attack.
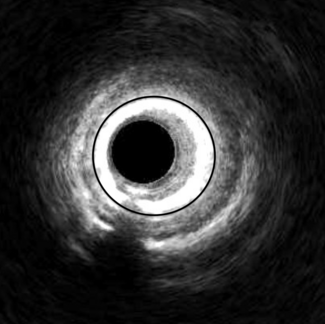
To address this challenge, a UCLA team of bioengineers, computer scientists, and physicians designed a stent to be implanted during an angioplasty and powered by the natural pulsing of blood through the artery. With each heartbeat, the artery expands and contracts, causing the stent’s magnetoelastic mesh to subtly change shape and generate magnetic signals. A small, flexible coil affixed to the skin above the artery with an adhesive picks up these magnetic changes, converting them into electrical signals that can then be analyzed remotely by physicians.
While a range of wearable and implantable technologies have been developed for post-angioplasty monitoring, this system is uniquely self-powered and leverages the magnetoelastic effect to convert vascular motion into continuous electrical signals.
“We believe this approach could enhance current stent technologies and enable earlier diagnosis of restenosis,” said study principal investigator Jun Chen, an associate professor of bioengineering at the UCLA Samueli School of Engineering. “The magnetoelastic mesh adds diagnostic capability without compromising the biocompatibility of the procedure or how the stent supports the artery.”
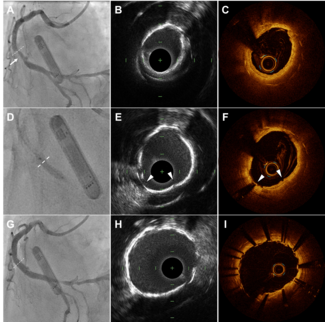
To test their prototype stent, the researchers implanted it in pig arteries using standard clinical catheters and real-time imaging guidance. A sensor coil was attached to the skin and electrical signals were recorded by a current preamplifier. The device successfully captured abnormal blood flow patterns with manually induced artery narrowing, and AI-assisted signal analysis confirmed the changes in real time.
Following initial successful demonstrations in lab settings, the team plans to refine the technique in preparation for future human clinical studies. Chen said the ultimate goal is to build an affordable commercial device that could be paired with a smartphone app to automatically alert a doctor’s office if the stent detects signals suggesting the onset of artery narrowing.
“This is an important first step toward true continuous, remote monitoring of stents by building on tools and procedures already in use by endovascular specialists,” said study co-corresponding author Dr. Geoffrey Colby, a cerebrovascular and endovascular neurosurgeon at UCLA Health. “If ultimately proven clinically reliable, the approach could offer a practical way to monitor stents long term, reducing diagnostic delays and improving long-term patient outcomes.”
UCLA Samueli bioengineering doctoral student Guorui Chen, a member of Chen’s Bioelectronics Research Group, is the first author on the paper. Senior authors include UCLA Health Dr. Anthony Wang, a cerebrovascular neurosurgeon, and Dr. Olujimi Ajijola, a cardiac electrophysiologist. Other UCLA senior authors are Paul Weiss, a distinguished professor of chemistry and biochemistry, bioengineering and materials science and engineering; Wei Wang, a professor of computer science; and Song Li, a professor of bioengineering.
Other authors, all affiliated with UCLA at the time of the study, include Dr. Wi Jin Kim, a former UCLA neurosurgery resident; current and former postdoctoral scholars Yihao Zhou, Zeyang Liu, Tzuchun Chung and Yan-Ruide Li; current and former bioengineering graduate students Youcheng Yang, Jing Tian, Xun Zhao, Kamryn Scott, Jing Xu and Jarod Carol; computer science doctoral student Junkai Zhang advised by Wang; and UCLA staff research associate Lily DeFelice, who is a member of Ajijola’s research lab.
The research was funded by the National Institutes of Health, the National Science Foundation, the U.S. Office of Naval Research, the American Heart Association, the California Institute for Regenerative Medicine, the VIVA Foundation, Amazon Web Services and the UCLA Department of Neurosurgery.
Find More:
Renal Denervation Topic Center
Cardiovascular Ambulatory Surgery Centers (ASCs) Topic Center
Grand Rounds With Morton Kern, MD
Peripheral Artery Disease Topic Center