Should We Implement a Controlled Pause for CPR During Rescue PCI for Unprotected Left Main Lesion With Sudden Occlusion?
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Cath Lab Digest or HMP Global, their employees, and affiliates.
Morton Kern with contributions from Mir Basir, Detroit, Michigan; James Blankenship, Albuquerque, New Mexico; Rhian Davies, York, Pennsylvania; Ajay Kirtane, New York, New York; Lloyd Klein, Sonoma, California; Mitchell Krucoff, Raleigh, North Carolina; Jeff Schussler, Dallas, Texas; Arnold Seto, Long Beach, California
Morton J. Kern, MD, MSCAI, FACC, FAHA
Clinical Editor; Interventional Cardiologist, Long Beach VA Medical Center, Long Beach, California; Professor of Medicine, University of California, Irvine Medical Center, Orange, California
Disclosures: Dr. Morton Kern reports he is a consultant for Abiomed, Abbott Vascular, Philips, ACIST Medical, and Opsens Inc.
Dr. Kern can be contacted at mortonkern2007@gmail.com
The practice of percutaneous coronary intervention (PCI) of unprotected left main (UPLM) stenosis has a complicated history of success and stilI carries the potential for an ominous outcome for all operators despite the years of experience they may hold. LM lesions can respond in an unpredictable fashion with dissection or thrombosis during PCI. Moreover, not all left main stenoses are the same due to the differences in location (ostial, mid, distal bifurcation, degree of calcification, clinical presentation (i.e. unstable/acute), and cause of the narrowing (e.g., radiation exposure from prior treatment of breast cancer or lymphoma). Protected left main PCI, i.e., those in the setting of a coronary artery bypass graft, are thought to be of lower risk than those lesions undergoing unprotected (UPLM) PCI.
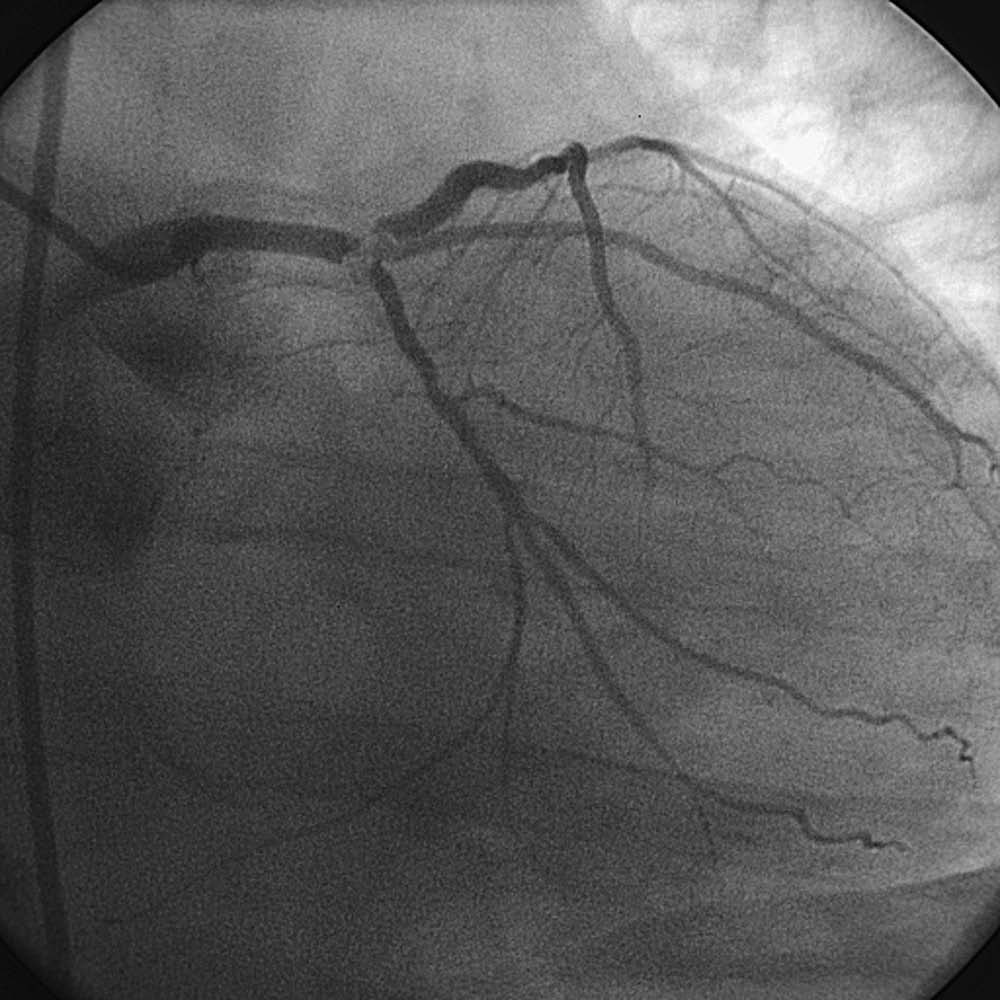
This discussion was prompted by a story I received about a patient who had a cardiac arrest on the cath table during treatment of UPLM non-ST elevation myocardial infarction. Immediately after guidewire passage into both the left anterior descending (LAD) and circumflex (CFx) vessels, there was no flow into either the LAD or CFX; the patient experienced ventricular fibrillation leading to cardiac arrest, and cardiopulmonary resuscitation (CPR) was initiated immediately in accordance with advanced cardiac life support (ACLS) guidance. However, chest compressions resulted in loss of guide catheter engagement and wire position. Once access was lost, stent delivery became extremely difficult. Despite attempts to restore flow, the patient did not survive.
Daniel Aboyeji, RN, BSc, from MMUH, in Birmingham, UK, wanted to know our thoughts on implementing a controlled or delayed CPR in such a setting. He writes, “In highly selected cases of cardiac arrest clearly caused by abrupt left main occlusion during PCI, I wondered whether a very brief, structured window to prioritise immediate stent deployment before initiating chest compressions might help preserve guide stability and allow rapid restoration of coronary flow. This would not represent a fixed delay, but a clinically judged, minimal interval — likely on the order of seconds and not typically exceeding 1–2 minutes, aimed at facilitating definitive reperfusion in a controlled setting.”
I polled my colleagues for their thoughts.
 Mort Kern, Long Beach, California: Cardiac arrest and death in the cath due to a complication of UPLM PCI is rare. Even so, some labs even without surgical backup might undertake a high-risk PCI, like UPLM. Because our center does not do cardiothoracic (CT) surgery, our tolerance for very high-risk PCI is low and we would transfer this patient to a full-service hospital with CT surgery and mechanical circulatory support (MCS) devices. Whether the operators there would perform MCS PCI depends on a variety of factors, but the option exists.
Mort Kern, Long Beach, California: Cardiac arrest and death in the cath due to a complication of UPLM PCI is rare. Even so, some labs even without surgical backup might undertake a high-risk PCI, like UPLM. Because our center does not do cardiothoracic (CT) surgery, our tolerance for very high-risk PCI is low and we would transfer this patient to a full-service hospital with CT surgery and mechanical circulatory support (MCS) devices. Whether the operators there would perform MCS PCI depends on a variety of factors, but the option exists.
The question about pausing CPR to permit guide catheter stabilization and guidewires passed into the vessels to rescue an UPLM complication is worth considering. A brief pause may be needed but only for a very short time (<20-30 sec). One can ask the team to hold for 30 seconds to reseat the guide and see if the operator can get wires back down the vessel. CPR would continue again with intermittent breaks to permit wires/balloons/stents to be delivered. We would insert an Impella (Abiomed), while hoping for return of spontaneous cardiac rhythm (ROSC). Survival depends on reestablishing flow despite MCS.
This case also suggests that for UPLM PCI, MCS has some real value, despite conflicting studies. The literature/guidelines for requiring MCS for high-risk PCI is still being argued as to MCS benefit. CABG surgery rarely helps the situation, because of the logistics of getting to the OR in a timely manner.
As for a choice of MCS, personally, I don't think an intra-aortic balloon pump (IABP) provides strong enough cardiac output to support a patient with cardiac arrest during PCI/stenting salvage. In many UPLM PCI, especially with reduced left ventricular (LV) ejection fraction, Impella placement seems the right approach before LM wiring to help manage the scenario of an arrest during the procedure. Colleagues, what are your thoughts?
Jeff Schussler, Dallas, Texas: Unfortunate, but we've been there. We do a fair amount of UPLM stenting and understand that there are theoretical risks (just like there are theoretical risks to putting in support). I don't think there's a one-size-fits-all plan for this.
With regards to holding off on CPR, there are times when we do pause CPR for stent placement (not just LM, but other arteries as well) due to the inability to accurately place the stent during compressions. Depending on when this happens (e.g., are the stents in the body already and about to be deployed?) you could make a cogent case for delaying CPR while stents are being placed.
I do also think that if you're doing CPR due to LM occlusion and you've lost your wire position, the only chance the patient is going to have is mechanical support (Impella/extracorporeal membrane oxygenation [ECMO]).
 Mir Basir, Detroit, Michigan: Sad story, Mort. I would start by asking the following questions: was anticoagulation appropriate? Were the patient’s hemodynamics stable to start the case? Was the lesion so tight that wiring resulted in occlusion and arrest, or did a complication occur during wiring like dissection or disruption of plaque?
Mir Basir, Detroit, Michigan: Sad story, Mort. I would start by asking the following questions: was anticoagulation appropriate? Were the patient’s hemodynamics stable to start the case? Was the lesion so tight that wiring resulted in occlusion and arrest, or did a complication occur during wiring like dissection or disruption of plaque?
You can call me new school, but I don't like the idea of quickly stenting. I think the stent complicates matters further. If there is thrombus in the left main, for example, you could embolize it into the LCx, and make things more difficult. If it's an acute lesion on chronic disease, the stent may be under expanded. In the shock initiative,1 it took only minutes to implant Impella, commonly allowing operators to stop CPR by restoring ROSC. Maybe in this case, the operators should have just shocked the patient out of VF and given meds to see if that was all that is needed to restore perfusion before starting CPR. When we are doing extracorporeal cardiopulmonary resuscitation (ECPR), we sometimes need to hold CPR for a few seconds while getting access, but I find it hard to hold off for more than a few seconds — 2 minutes seems too long.
 Ajay Kirtane, New York, New York: I agree with the other comments. Every situation is different, and the root cause must be identified and treated. For many cases, the LM is treated with stenting but for others it might be aspiration or something else. Certainly, when the commotion of CPR, etc., ensues, it becomes even more imperative to pay attention to and maintain wire position (but with very aggressive compressions and in the throes of a code, this becomes more treacherous). Shocking for ventricular tachycardia/VF and giving pressors is less likely to displace a wire than uncontrolled CPR.
Ajay Kirtane, New York, New York: I agree with the other comments. Every situation is different, and the root cause must be identified and treated. For many cases, the LM is treated with stenting but for others it might be aspiration or something else. Certainly, when the commotion of CPR, etc., ensues, it becomes even more imperative to pay attention to and maintain wire position (but with very aggressive compressions and in the throes of a code, this becomes more treacherous). Shocking for ventricular tachycardia/VF and giving pressors is less likely to displace a wire than uncontrolled CPR.
Finally, there are many shock cases for which the pump should ideally be inserted prior to reperfusion. When there is no flow beyond a left main occlusion, we often put in hemodynamic support and wire the vessel to restore flow at the same time.
 Rhian Davies, York, Pennsylvania: Sad case. I agree with prior comments. 2 mins would feel like an eternity. The most critical part is running through the possible causes and treatment in an expedited way — reengage, rewire, image, aspirate, deliver meds, balloon, etc. Fortunately, here (Pennsylvania) in most cath labs we can quickly get support in, but unfortunately this is not the case universally.
Rhian Davies, York, Pennsylvania: Sad case. I agree with prior comments. 2 mins would feel like an eternity. The most critical part is running through the possible causes and treatment in an expedited way — reengage, rewire, image, aspirate, deliver meds, balloon, etc. Fortunately, here (Pennsylvania) in most cath labs we can quickly get support in, but unfortunately this is not the case universally.
 James Blankenship, Albuquerque, New Mexico: We had a case of LMCA/LAD PCI with low ejection fraction (EF). Impella was placed prospectively. LAD stenting caused no-reflow and VF arrest was refractory to multiple defibrillations. Effective CPR was provided (systolic 100 mm) while ECMO was rapidly implemented. Defibrillation was successful on ECMO. LMCA was stented with ECMO support. The patient was decannulated the next day and went home several days later in good condition. In this case, Impella support was not enough, but ECMO implemented rapidly while delivering effective CPR proved a winning strategy. If ECMO had not been available, it would have been difficult to have a good outcome. LMCA stent placement requires some finesse, and is even harder to do under CPR. A 2-minute pause might be enough, but often salvage PCI cannot be done that fast.
James Blankenship, Albuquerque, New Mexico: We had a case of LMCA/LAD PCI with low ejection fraction (EF). Impella was placed prospectively. LAD stenting caused no-reflow and VF arrest was refractory to multiple defibrillations. Effective CPR was provided (systolic 100 mm) while ECMO was rapidly implemented. Defibrillation was successful on ECMO. LMCA was stented with ECMO support. The patient was decannulated the next day and went home several days later in good condition. In this case, Impella support was not enough, but ECMO implemented rapidly while delivering effective CPR proved a winning strategy. If ECMO had not been available, it would have been difficult to have a good outcome. LMCA stent placement requires some finesse, and is even harder to do under CPR. A 2-minute pause might be enough, but often salvage PCI cannot be done that fast.
 Mitchell Krucoff, Raleigh, North Carolina: Many insightful comments, and no matter what you do, or how much, we accept that some bad outcomes, including death, are inevitably a part of our profession. It is always soul-rattling to kill someone with your own hands…as I tell our fellows, if you don’t feel that way, you are in the wrong profession.
Mitchell Krucoff, Raleigh, North Carolina: Many insightful comments, and no matter what you do, or how much, we accept that some bad outcomes, including death, are inevitably a part of our profession. It is always soul-rattling to kill someone with your own hands…as I tell our fellows, if you don’t feel that way, you are in the wrong profession.
This discussion reminds us that the key is to think and connect with others, to consider what can be learned to at least reduce the likelihood of similar outcomes if you plan to make a living doing high-risk cases. Here are some of my basic principles to keep on our radar:
1. Assess the risk of the coronary anatomy in the context of LV function. If the patient has a low EF, ischemia in a more modest territory of functional myocardium may have more catastrophic effects. Hypertrophic hearts (e.g., aortic stenosis) may be much less forgiving (important especially in the era of transcatheter aortic replacement). If the left coronary is strongly dominant, a UPLM PCI complication, even with a normal LV, may not be tolerated.
2. If the little hairs on the back of your neck stand up contemplating the procedural risk, be preemptive. MCS works better preventing catastrophe than reversing catastrophe. Many of us old folks can testify that a good old IABP placed before you engage the coronary is much better at providing tolerance for PCI than for reversing shock, with fewer complications than an Impella.
3. I was trained to double-dilute the contrast used to prep balloons, as it lowers contrast viscosity and deflates faster than routine “half ‘n half” we use in elective settings, and to have a prepped 2.5 mm balloon on the table before you wire a potentially hazardous lesion. This puts you into a position to very quickly dilate the culprit — not as a happy ending, but as the quickest step to restorative flow.
4. If there is no MCS in place and the patient crashes, we try to get two teams going: one to insert the MCS, the other to advance the PCI and get the coronary open. CPR alone will not save this life.
5. I absolutely would NOT consider prolonged CPR interruption. We have learned that even defibrillation and ventilation should NOT delay CPR. So, we continue CPR until we are ready to take a step with the MCS or with the revascularization. We will interrupt CPR to re-intubate the LM with the guide, then CPR, then interrupt to re-advance the wire, then CPR, then interrupt to get balloon across, inflated for 10 seconds and back in the guide, then CPR…etc.
6. To get a new access and bring an MCS device into play, access during CPR and even bring the wire up around the arch, get the device into the iliac, then interrupt CPR to position properly, quickly, and resume CPR while you purge, flush, fill.
It’s no fun to relive all of this, but one of the reasons I love these exchanges, after 42 years, is that I am still learning from all of you…
 Lloyd Klein, Sonoma, California: My experience has been that there is no chance of survival without opening the artery. Stop the resuscitation and get it done as fast as possible. It’s not a chicken-and-egg dilemma, because the acute occlusion caused the arrest, and it’s unlikely to stop until coronary flow is re-established.
Lloyd Klein, Sonoma, California: My experience has been that there is no chance of survival without opening the artery. Stop the resuscitation and get it done as fast as possible. It’s not a chicken-and-egg dilemma, because the acute occlusion caused the arrest, and it’s unlikely to stop until coronary flow is re-established.
 Arnold Seto, Long Beach, California: The risk of UPLM intervention depends on whether the right coronary artery is occluded, small, or nondominant, whether the lesion is at the distal bifurcation or not, the presence of calcification, and the patient's baseline LVEF. Like other cases of "CHIP" or high-risk intervention, the multitude of factors make it hard to define a single, preferred technique. In addition, the risk tolerance depends on the malpractice environment, with highly litigious states or countries demanding more precautions. Some LM cases in the U.S. are performed with 8 French guides. In contrast, Impella support would be performed with 6 French radial access without any support in the UK.
Arnold Seto, Long Beach, California: The risk of UPLM intervention depends on whether the right coronary artery is occluded, small, or nondominant, whether the lesion is at the distal bifurcation or not, the presence of calcification, and the patient's baseline LVEF. Like other cases of "CHIP" or high-risk intervention, the multitude of factors make it hard to define a single, preferred technique. In addition, the risk tolerance depends on the malpractice environment, with highly litigious states or countries demanding more precautions. Some LM cases in the U.S. are performed with 8 French guides. In contrast, Impella support would be performed with 6 French radial access without any support in the UK.
The Bottom Line
Complications of UPLM PCI should be anticipated and preemptive measures put in place to avoid a fatal outcome. A very brief pause in CPR is acceptable, but a prolonged delay was not favored by the experts here. Lloyd Klein in Sonoma, California, sums it up well: “you have no chance of survival without opening the artery. Stop the resuscitation and get it done”. Pause, work to open the vessel, pump, and repeat.
Reference
1. Naidu SS, Nathan S, Basir MB, Baran DA, Marbach JA, Grines CL. SCAI Door to Lactate Clearance (SCAI DLC) Cardiogenic Shock Initiative: Definition, Hypothesis, and Call to Action. J Soc Cardiovasc Angiogr Interv. 2025 Sep 18;4(11):103996. doi: 10.1016/j.jscai.2025.103996