Trickle Charging Flaps: Maintaining Deep Inferior Epigastric Perforator Flap Perfusion After Pedicle Division
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of ePlasty or HMP Global, their employees, and affiliates.
Abstract
Background. Trickle charging is a process by which an external power supply is used to maintain charge in a battery. When applied to flaps, trickle charging allows temporary perfusion of the flap (battery) by using a perforator that is discontinuous from the main pedicle (external power supply), allowing main pedicle ligation. Two procedures that benefit from this technique are the conventional open bilateral deep inferior epigastric perforator (DIEP) flap and the robotic bilateral DIEP flap.
Methods. In the conventional open bilateral DIEP flap, trickle charging is used to improve perioperative efficiency. In the event that the recipient site is not ready for flap transfer (ie, mastectomy and/or chest vessel preparation are not completed), trickle charging enables the main pedicle to be harvested from the intraabdominal cavity. This facilitates early rectus fascia and partial abdominal closure. In a bilateral robotic DIEP flap, trickle charging allows the second hemi-abdominal flap to maintain perfusion after the main pedicles in the intra-abdominal cavity are robotically harvested. Both scenarios demonstrate the possibility of temporary perfusion of the flap, despite the main pedicle being ligated.
Results. Sixteen total patients had hemi-abdominal flaps that were trickle charged (5 conventional, 11 robotic). All the flaps were tested for proper perfusion intraoperatively using multiple different modalities. None of the patients experienced total flap loss, partial flap loss, or fat necrosis.
Conclusions: The authors review the safety of trickle charging in consecutive robotic and open bilateral DIEP flap breast reconstructions. This technique can be easily replicated for various other flaps with the help of preoperative computed tomography or magnetic resonance angiography to identify main pedicle vessels and an alternative temporary perforator.
Introduction
Conventionally, trickle charging is the process by which an external power supply is used to charge a battery at a rate that matches the battery self-discharge rate to keep the battery from depleting. This concept can be transferred to flaps, where trickle charging a flap maintains perfusion of the flap on a temporary perforator that is external to the main pedicle. Trickle charging increases operative efficiency by enabling main pedicle division and, therefore, flap harvest while retaining continuous perfusion. Two procedures that benefit from trickle charging are the conventional open bilateral deep inferior epigastric perforator (DIEP) flap surgery and the robotic bilateral DIEP flap reconstruction.
A total of 16 consecutive patients had hemi-abdominal flaps that were trickle charged (5 conventional, 11 robotic). All hemi-abdominals flap that underwent trickle charging had clinical evidence of perfusion, including audible Doppler signal, good color, and appropriate capillary refill. De-epithelialized areas had bright red bleeding. Indocyanine green (ICG) angiography demonstrated blood flow in the temporary perforator and perfusion within the flap itself. Trickle charging with main pedicle transection in all 16 patients allowed for early fascial closure prior to harvesting and transferring the flap for microsurgery and inset. Postoperatively, the patients had no total flap loss, partial flap loss, or fat necrosis (Supplemental Figure 1).
Case Presentations
Case 1: trickle charging in bilateral conventional DIEP flap surgery
The patient was a 46-year-old woman with left breast cancer who opted for bilateral nipple-sparing mastectomy with immediate bilateral breast reconstruction using DIEP flaps. The right mastectomy was completed first, thereby allowing the contralateral left hemi-abdominal flap to be harvested and anastomosed without complication. The second (right) flap was dissected prior to completion of the left mastectomy and recipient vessel exposure. This right hemi-abdominal flap was based on medial row perforators of the deep inferior epigastric artery and vein (Figure 1). Pedicle ligation of the right hemi-abdominal flap was performed prior to the flap being ready for transfer. This flap maintained perfusion (trickle charging) with a single lateral row perforator that was no longer in continuity with the main pedicle (Figure 2). With main pedicle ligation, the preserved lateral row perforator most probably obtained its blood supply from an intercostal branch that had yet to be divided, or from its continuation with the superior epigastric system. It is also important to note that flaps based on lateral row perforators can also be perfused or trickle charged with a single medial row perforator.
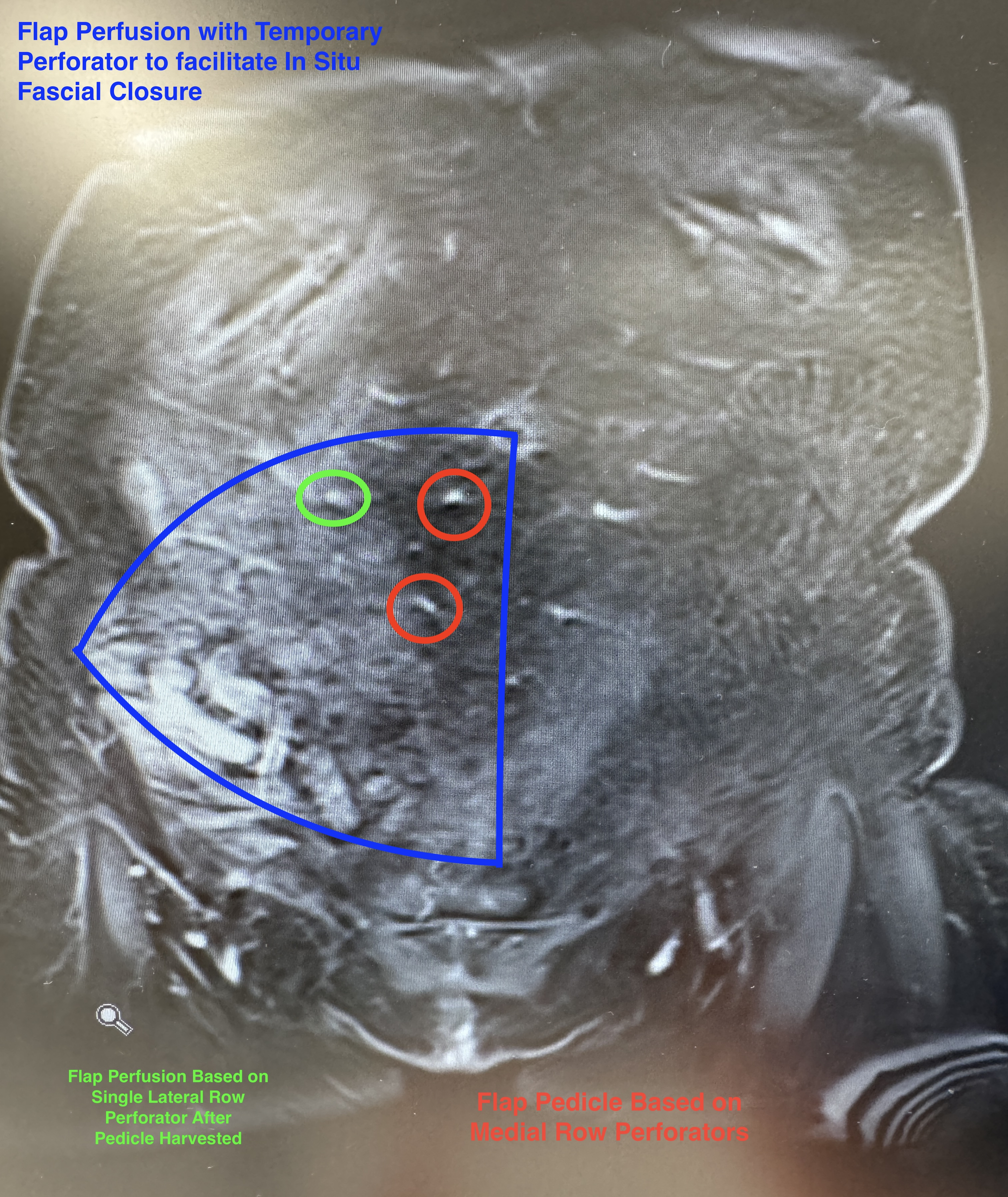
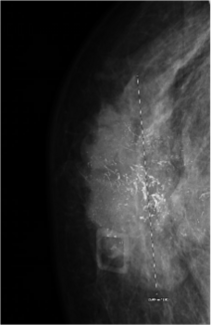
Figure 1: Coronal view of magnetic resonance angiography demonstrated the medial row perforators (red) on which the right hemi-abdominal flap was based. The single lateral row perforator (green) was used to temporarily perfuse the flap (trickle charging).
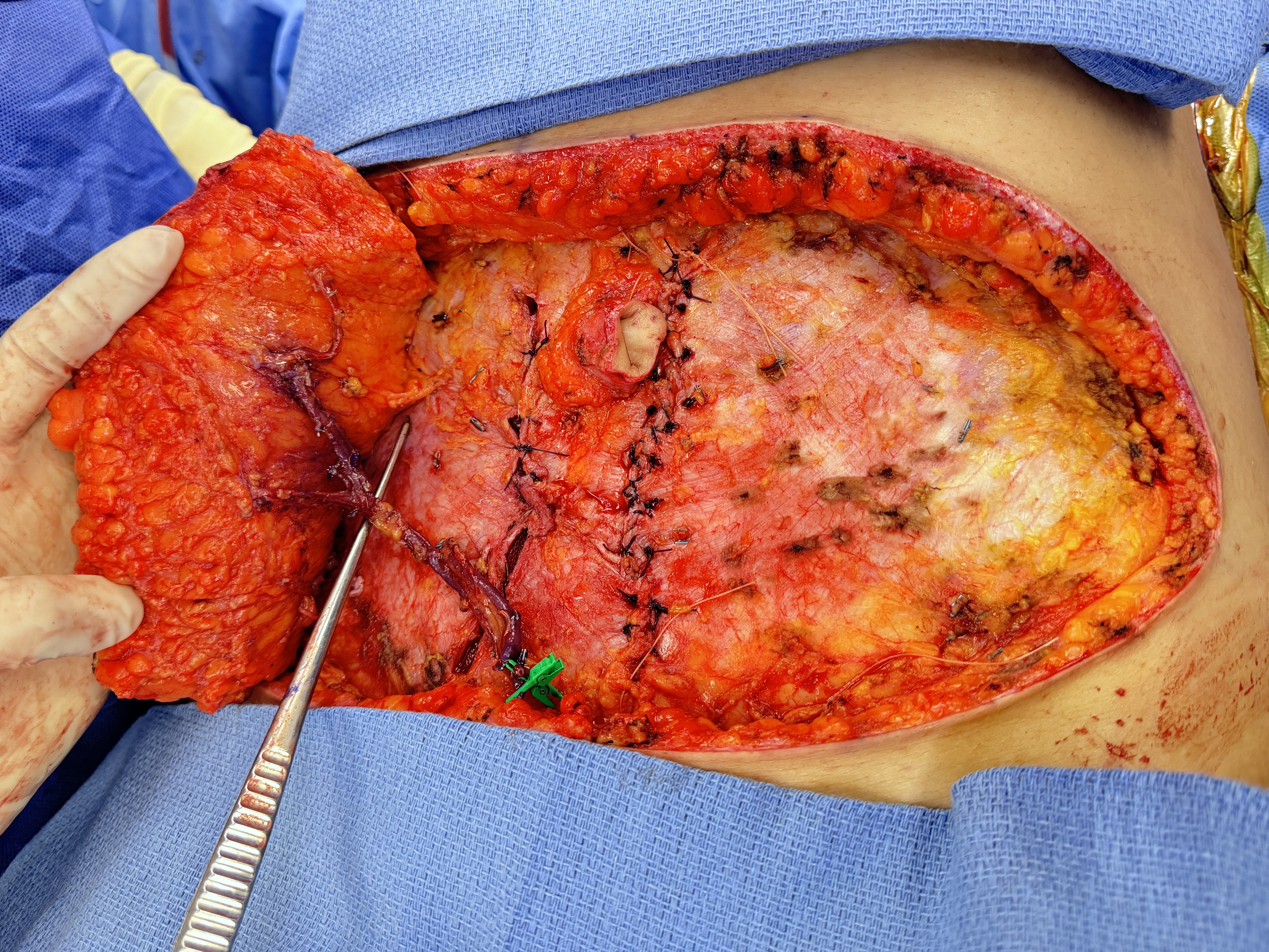
Figure 2: Single lateral row perforator provided temporary perfusion to the right hemi-abdominal deep inferior epigastric perforator flap after the main pedicle with medial row perforators was divided.
Perfusion was confirmed clinically and with intravenous angiography. The skin paddle of the flap was noted to have an audible Doppler signal, good color, and appropriate capillary refill. De-epithelialized areas had bright red bleeding (Video 1). Based on the lateral row perforator, retrograde flow was noted from the main pedicle (Video 2). ICG angiography demonstrated blood flow in the perforator and perfusion within the flap itself (Videos 1 and 3). With the pedicle ligated and removed from the intra-abdominal cavity, the hemi-abdominal flap was displaced and secured laterally. This facilitated anterior rectus fascia closure with the flap in-situ at the donor site (Figures 3 and 4). In addition, the patient was placed in a semi-reflexed position, and the abdomen was partially closed (contralateral side at the first hemi-abdominal flap donor site). Following completion of the mastectomy and recipient site vessel exposure, the flap was transferred to the chest for anastomosis, and the vessels were anastomosed with no intra- or postoperative complications, including total flap loss, partial flap loss, or fat necrosis
Video 1: The main pedicle of the right hemi-abdominal flap was divided and removed from the abdominal cavity. The flap was temporarily perfused on the lateral row perforator. Intravenous angiography confirmed perfusion by demonstrating indocyanine green fluorescence in the flap.
Video 2: The main pedicle of the right hemi-abdominal flap was divided and removed from the abdominal cavity. The flap was temporarily perfused on the lateral row perforator. Retrograde blood flow was seen through the main pedicle.
Video 3: Intravenous angiography confirmed perfusion by demonstrating indocyanine green fluorescence in the temporary lateral row perforator.
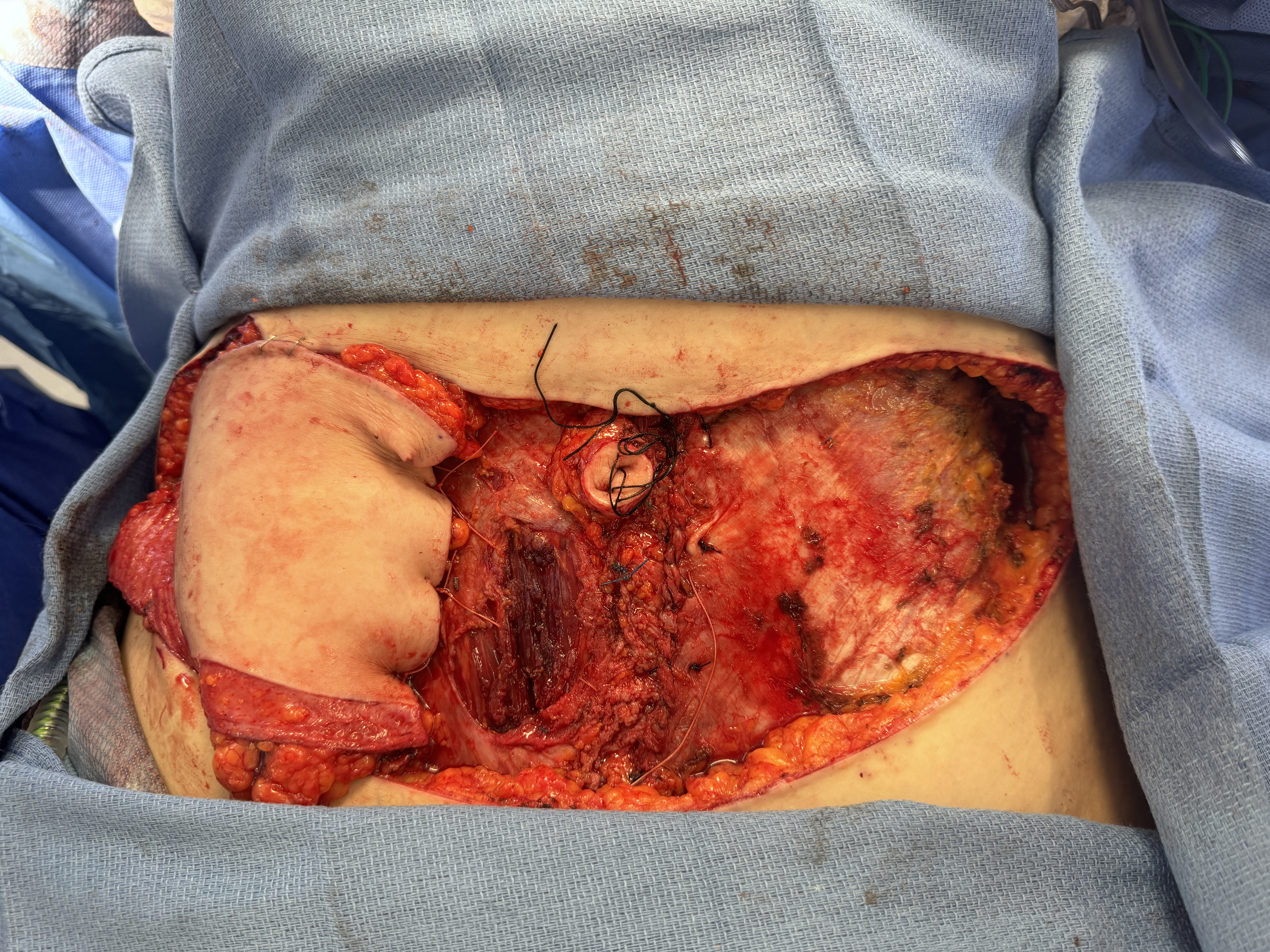
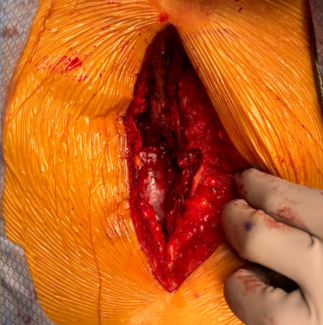
Figure 3: The right hemi-abdominal flap was laterally displaced after the pedicle was divided and removed from the intra-abdominal cavity. The flap was temporarily perfused by a lateral row perforator.
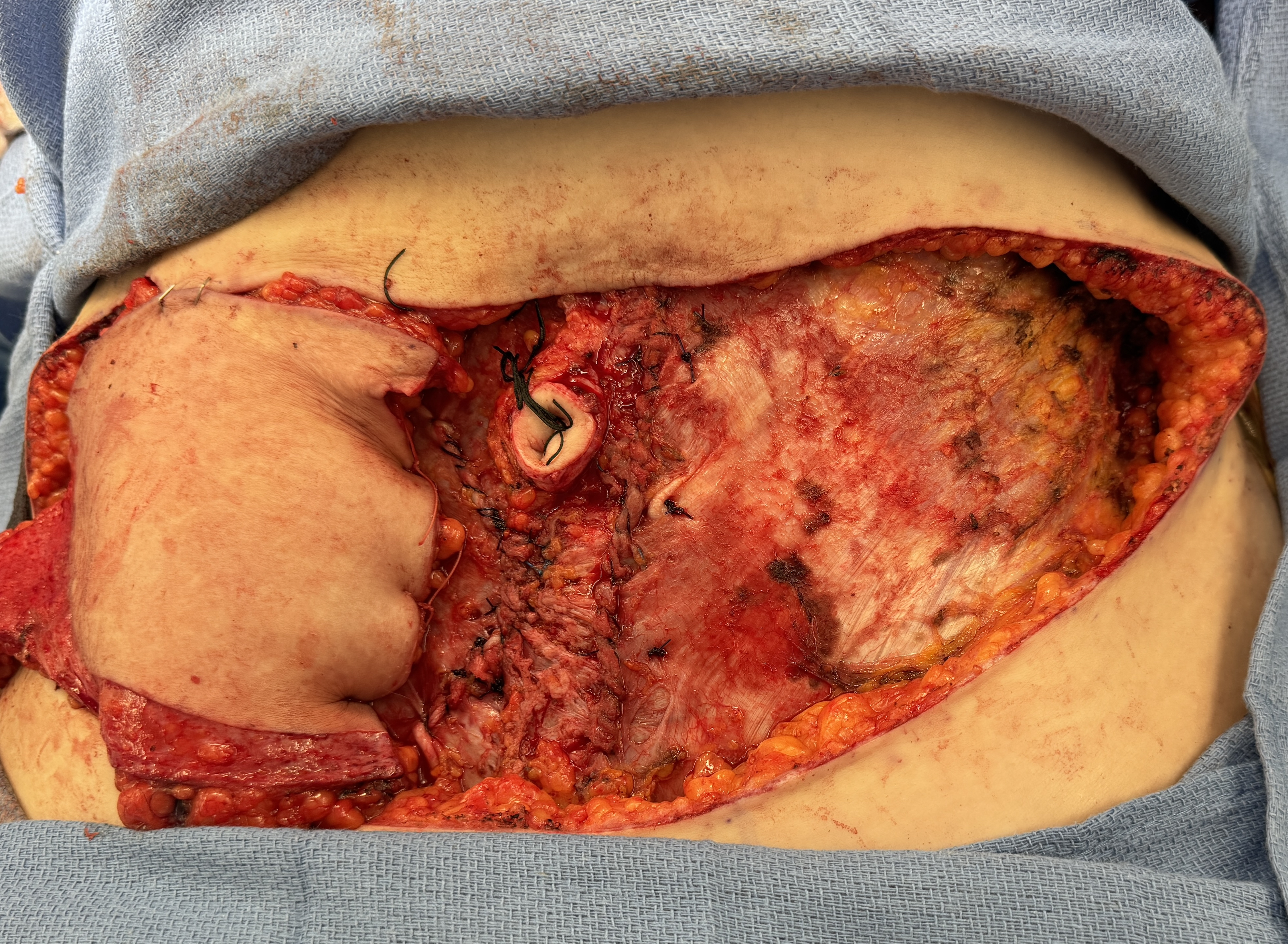
Figure 4: The rectus fascia was closed with deep inferior epigastric perforator flap in-situ at the abdominal donor site while the flap awaited transfer to the recipient site.
Case 2: trickle charging in bilateral robotic DIEP flap surgery
A 43-year-old female underwent delayed bilateral breast reconstruction with robotic DIEP flaps. Both left and right hemi-abdominal flaps were initially elevated, similar to a conventional open approach. Bilateral tissue expander removal and recipient vessel exposure were completed prior to docking the robot. Similarly, the order of flap transfer and anastomosis was identified prior to robotic dissection. In the hemi-abdominal flap that would be transferred second, a separate perforator (discontinuous from the main pedicle) was identified and preserved. This perforator enabled temporary perfusion (trickle charging) of the flap after the pedicle was harvested intra-abdominally with robotic assistance. For both hemi-abdominal flaps, when the main pedicle was identified in the submuscular plane, dissection was then converted to a robotic-assisted intra-abdominal pedicle harvest.
Both hemi-abdominal pedicles were ligated and retrieved from the abdominal cavity. The fascia was closed bilaterally while the first hemi-abdominal flap was transferred to the recipient site for microsurgical anastomosis. The second hemi-abdominal flap awaited transfer while maintaining perfusion on the temporary perforator (trickle charging). This temporary perforator of the second hemi-abdominal flap was then sacrificed once the second flap was ready for transposition and microsurgical anastomosis. The remainder of the operation progressed similar to a conventional DIEP flap. No flap-related complications were noted.
Discussion
Bilateral DIEP flap reconstruction is a highly technical and time-intensive procedure.1-3 Optimizing surgeon workflow to reduce the time when surgeons are not actively operating on the body is key to improving efficiency and reducing operative time.4 Innovations such as the use of a co-surgeon, preoperative imaging, parallel work, and loupe magnification have drastically improved operative efficiency for DIEP flap reconstruction.3 Additionally, quality improvement implementations like Lean and Six Sigma have resulted in workflow and process efficiency advances such as optimal positioning of personnel, microscope, mayo stand, and back tables while process maps provide temporal cues to keep surgeons synchronized and on pace.4
Trickle charging is yet another novel method of improving operative efficiency. For the conventional open bilateral DIEP flap surgery, trickle charging allows early fascial closure with potential early abdominal closure, which reduces wasted time spent waiting for the next step in the procedure. In a robotic bilateral DIEP flap reconstruction, trickle charging allows the second hemi-abdominal flap to maintain perfusion after the main pedicle is robotically harvested.
Conclusions
These 2 simple cases demonstrate the applicability and utility of trickle charging DIEP flaps. However, trickle charging is not a technique limited to the DIEP flap, as similar temporary perfusion vessels exist for various other flaps. With assistance from preoperative computed tomography or magnetic resonance angiography to identify main pedicle vessels and a temporary perforator, this technique is easily replicable.
Acknowledgments
Authors: Eric Bao, BS; Isabelle T. Smith, BA; Hannah B. Hoffman, MD; Gabrielle P. Odoom, BA; Mark L. Smith, MD; Neil Tanna, MD, MBA
Affiliation: Division of Plastic and Reconstructive Surgery, Northwell Health, Great Neck, New York
Correspondence: Neil Tanna, MD, Division of Plastic and Reconstructive Surgery, Northwell Health, 600 Northern Blvd, Suite 310, Great Neck, NY 11021, USA. Email: ntanna@gmail.com
Disclosures: The authors disclose no financial or other conflicts of interest.
Supplemental Material
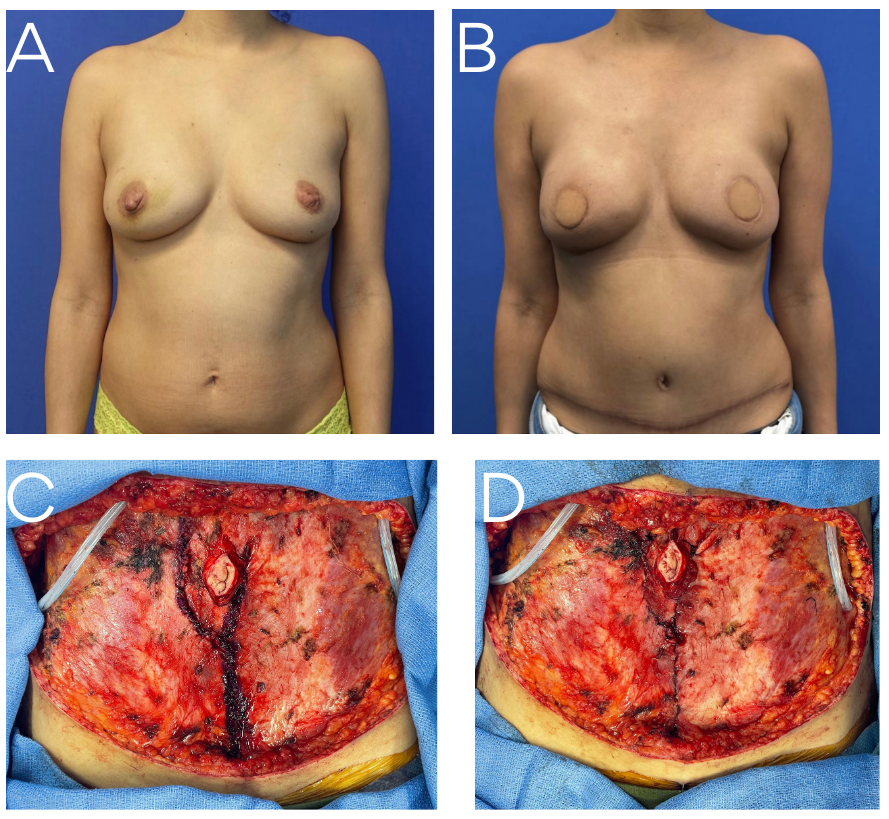
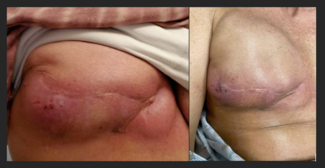
Supplemental Figure: The patient was a 41-year-old female who presented with right breast cancer. She elected to undergo bilateral skin-sparing mastectomy with bilateral deep inferior epigastric perforator flap breast reconstruction. (A) Pre- and (B) postoperative frontal photographs demonstrated flap viability while the patient awaited second-stage nipple-areola reconstruction. (C) Intraoperatively, a fascial-sparing approach to the pedicles with a single midline fascial cut was performed. (D) Trickle charging to the second flap allowed for fascial closure prior to the second hemi-abdominal flap being transferred for microsurgery and inset.
References
- Depypere B, Herregods S, Denolf J, et al. 20 years of DIEAP flap breast reconstruction: a big data analysis. Sci Rep. 2019;9(1):12899. doi:10.1038/s41598-019-49125-w
- Issa CJ, Lu SM, Boudiab EM, et al. Comparing plastic surgeon operative time for DIEP flap breast reconstruction: 2-stage more efficient than 1-stage? Plast Reconstr Surg Glob Open. 2021;9(6):e3608. doi:10.1097/GOX.0000000000003608
- Haddock NT, Teotia SS. Efficient DIEP flap: bilateral breast reconstruction in less than four hours. Plast Reconstr Surg Glob Open. 2021;9(9):e3801. doi:10.1097/GOX.0000000000003801
- Tanna N, Clappier M, Barnett SL, et al. Streamlining and consistency in surgery: Lean Six Sigma to improve operating room efficiency. Plast Reconstr Surg. 2023;152(3):682-690. doi:10.1097/PRS.0000000000010240