Heparin-Induced Thrombocytopenia in Patients Undergoing Autologous Breast Reconstruction With a Deep Inferior Epigastric Perforator Free Flap
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of ePlasty or HMP Global, their employees, and affiliates.
Questions
- What is heparin-induced thrombocytopenia (HIT)?
- How does HIT affect free flaps?
- How is HIT diagnosed in postoperative patients?
- What is the management of HIT in reconstructive surgery patients?
Case Description
A 46-year-old nonsmoking woman with a medical history of hypertension, hyperlipidemia, type I diabetes, and duodenal cancer with a BRCA gene mutation underwent bilateral skin-sparing mastectomies and immediate deep inferior epigastric perforator (DIEP) flap reconstruction. She received preoperative subcutaneous heparin and intraoperative heparinized saline flushes. On postoperative day 10, she presented with chest pressure, nausea, and vomiting. Computed tomography imaging revealed a right renal infarct (Figure 1). Exam findings included blanching erythema and large blisters over the DIEP flaps (Figure 2). Surgical reexploration confirmed patent DIEP pedicles with presumed bilateral intraflap microthrombosis compromising skin paddle perfusion. Following administration of intravenous (IV) heparin, the patient developed severe vasodilatory shock and cardiac arrest requiring cardiopulmonary resuscitation. There was a return of spontaneous circulation after administration of epinephrine, suggesting an anaphylactic reaction. She was diagnosed with heparin-induced thrombocytopenia (HIT) based on a positive platelet factor 4 (PF4) antibody and serotonin release assay. She was transitioned to argatroban and later discharged on apixaban. The patient ultimately experienced partial flap loss but recovered well. The flaps successfully integrated, and no further issues were encountered in the long term.
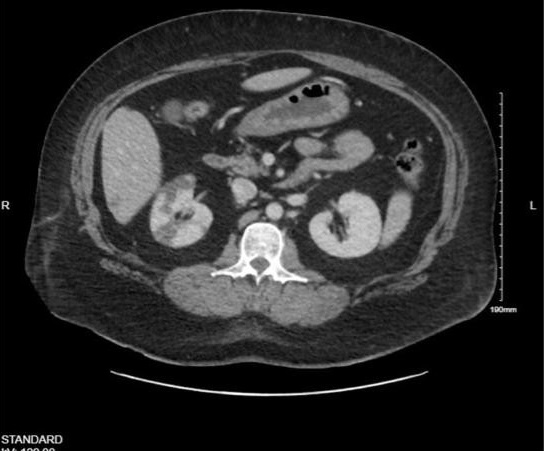
Figure 1. Computed tomography scan of the abdomen depicting the patient’s right renal infarct on readmission (postoperative day 10).
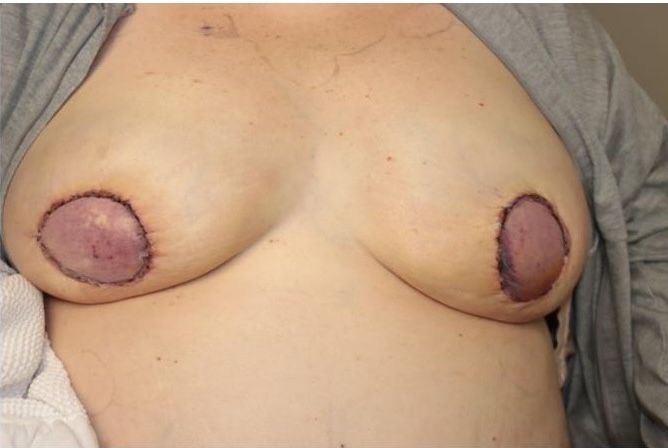
Figure 2. Deep inferior epigastric perforator flaps demonstrating blanching erythema on exam and without doppler signals on postoperative day 10.
Q1. What is HIT?
HIT is an immune-mediated adverse reaction to heparin characterized by thrombocytopenia and a paradoxical prothrombotic state due to platelet activation.1,2 HIT occurs when antibodies form against PF4-heparin complexes, leading to platelet activation, increased thrombin generation, and an elevated risk of both arterial and venous thrombosis.2
HIT is classified, by an older nomenclature, into 2 types: Type I HIT is a transient, nonimmune reaction occurring within the first 2 days of heparin exposure and does not lead to thrombosis. In contrast, Type II HIT, commonly referred to as HIT, is an immune-mediated disorder occurring 5 to 10 days after heparin initiation, with rapid-onset cases occurring sooner in those patients previously sensitized.1,2 The estimated incidence of HIT varies between 0.1% and 5%, with a 25% to 50% risk of associated thrombosis, particularly deep vein thrombosis (DVT) and pulmonary embolism (PE).1,2 Arterial events, including stroke and myocardial infarction (MI), are less frequent but can occur.2 HIT can lead to life-threatening complications, with thrombosis occurring in up to 89% of untreated cases.1 Venous thrombosis, including DVT and PE, is more common than arterial thrombosis, though ischemic limb necrosis, MI, and stroke can occur.1,2 Some patients may also develop skin necrosis at heparin injection sites or disseminated intravascular coagulation.2 Other potential complications associated with HIT are anaphylactoid reactions to unfractionated heparin, which can be fatal, or nonallergic anaphylaxis due to undesirable heparin contaminants.3,4 Anaphylactoid reactions resemble anaphylaxis; however, they are mediated by a different reaction.3 Both can occur in patients who recently received subcutaneous heparin and are then exposed to IV infusion of heparin. Both types of reactions can occur in association with HIT.3,4
Q2: How does HIT affect free flaps?
HIT can have severe consequences for free flaps, primarily because of its paradoxical hypercoagulable state, which increases the risk of microvascular thrombosis and venous congestion, leading to flap failure.5,6 Free flaps rely on adequate perfusion; any impairment in venous drainage or arterial inflow can cause ischemia, tissue necrosis, and, ultimately, total or partial flap loss. HIT-related thrombosis in free flaps may be progressive and multifocal, leading to repeated vascular compromise despite initial successful anastomosis.5 Development of micro-thrombosis within the flap can lead to partial or total flap loss. This is particularly problematic in microsurgical reconstruction, where venous thrombosis is already a leading cause of flap failure. Venous outflow obstruction results in congestion, edema, and tissue ischemia, which may require reexploration surgery or alternative salvage techniques such as leech therapy to maintain circulation while new venous pathways form.5
The onset of HIT can be unpredictable, with platelet counts declining 5 to 14 days after heparin exposure. Given this delay, microvascular complications can occur well into the postoperative period. Diagnosis of HIT requires great vigilance, especially in patients with unexplained thrombosis or recurrent flap compromise.6 In some cases, progressive thrombotic events can involve not only the flap vasculature but also larger vessels, as seen in a case where HIT-related thrombosis extended to the posterior tibial artery, ultimately necessitating a second flap procedure.6 Given its devastating impact, HIT should be suspected in any patient with unexplained free flap thrombosis and a history of heparin exposure. Early recognition and intervention are crucial to improving outcomes in microsurgical reconstruction.5,6
Q3: How is HIT diagnosed in postoperative patients?
HIT is diagnosed through a combination of clinical assessment and laboratory testing. The 4Ts score is a widely used clinical tool that stratifies the probability of HIT based on thrombocytopenia severity, timing of platelet decline relative to heparin exposure, presence of new thrombosis, and alternative explanations for thrombocytopenia.7 A low 4Ts score (<4) has a high negative predictive value, effectively ruling out HIT, while a high score (≥6) indicates a strong likelihood of HIT.8 Laboratory confirmation involves 2 main categories of tests: immunoassays and functional platelet activation assays.9 The most common immunoassay is the PF4-heparin enzyme-linked immunosorbent assay. Although this test has high sensitivity, it can also yield false positives because it detects both pathogenic and nonpathogenic antibodies.8,9 A positive PF4 therefore necessitates confirmatory functional testing, such as the serotonin release assay (SRA) or the heparin-induced platelet activation (HIPA) test, which detects platelet-activating antibodies and has higher specificity.8,9 A positive SRA is the gold standard for definitive HIT diagnosis.7
In postoperative patients, HIT can present as unexplained thrombocytopenia or thrombosis, often occurring 5 to 14 days after heparin exposure, though it may develop sooner in those with prior sensitization.7 The presence of thrombosis, particularly in free flap surgery, should prompt immediate HIT evaluation given that delayed diagnosis increases morbidity.8 With the potential for overdiagnosis, testing should be reserved for patients with intermediate-to-high pretest probability to avoid unnecessary treatment.9
Intraoperative evaluation of flap perfusion is helpful in assessing flap viability. Technologies such as laser angiography using the SPY system (Stryker) or near-infrared spectroscopy are reliable at assessing perfusion.10,11 In our case, we did not utilize any similar technology; however, the use of similar technology is highly recommended.10,11
Q4: What is the management of HIT in reconstructive surgery patients?
The management of HIT in reconstructive surgery patients requires immediate discontinuation of all heparin products to prevent further platelet activation and thrombosis.12,13 Because of HIT’s strong prothrombotic state, transitioning to a non-heparin anticoagulant is essential. Direct thrombin inhibitors, such as argatroban or bivalirudin, and indirect factor Xa inhibitors, such as fondaparinux or danaparoid, are commonly used alternatives.13
In the context of free flap surgery, early identification and treatment of HIT are crucial to flap salvage. Delays in treatment increase the risk of thrombosis within the microvascular anastomoses, leading to a total or partial flap loss.12 Studies have shown that transitioning to fondaparinux in HIT patients undergoing reconstructive surgery can successfully maintain flap viability while preventing further thrombotic events.12 Additionally, hematology consultation is recommended to guide anticoagulant selection based on individual risk factors, renal function, and surgical considerations.12 Long-term anticoagulation is required in patients with HIT-associated thrombosis, with oral anticoagulation (eg, warfarin or direct oral anticoagulants like apixaban) initiated only after platelet recovery to prevent warfarin-induced microvascular thrombosis.13
In the case of HIT-associated anaphylactoid or nonallergic anaphylactic reaction, it is important to avoid all heparins.4 Once the anaphylactic reaction is recognized, the initial step should be to discontinue heparin. If anticoagulation is still needed, alternative anticoagulants, such as hirudins or direct thrombin inhibitors, should be used.4 If the anaphylactic reaction is severe and a shock ensues, then management will follow regular shock management with IV fluids, epinephrine, and airway support.14 Collecting history from patients prior to administering heparin is essential; if there is any history of skin reaction at the heparin injection site, then avoidance of heparin should be considered.3,4 Currently, we are advocating for changing our institutional protocols to better respond to future cases of HIT.
Acknowledgments
Authors: Mohammed Yamin, BS1; Micaela Tobin, BA1; Tricia Raquepo, BA1; John B. Park, PharmD1; Helen Xun, MD1; Eric Zhu, PharmD2; Maria J. Escobar-Domingo, MD1; James E. Fanning, BS1; Amy Chen, BS1; Haobo Ma, MD3; Bernard T. Lee, MD, MBA, MPH1; Ryan P. Cauley, MD, MPH1
Affiliations: 1Division of Plastic and Reconstructive Surgery, Department of Surgery, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, Massachusetts; 2Department of Pharmacy, Beth Israel Deaconess Medical Center, Boston, Massachusetts; 3Department of Anesthesia, Critical Care, and Pain Medicine, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, Massachusetts
Correspondence: Ryan P. Cauley, MD, MPH; rcauley@bidmc.harvard.edu
Disclosures: The authors disclose no relevant financial or nonfinancial interests.
References
-
Salter BS, Weiner MM, Trinh MA, et al. Heparin-induced thrombocytopenia: a comprehensive clinical review. J Am Coll Cardiol. 2016;67(21):2519-2532. doi:10.1016/j.jacc.2016.02.073.
-
Warkentin TE. Clinical picture of heparin-induced thrombocytopenia. In: Warkentin TE, Greinacher A, eds. Heparin-Induced Thrombocytopenia. 5th ed. CRC Press; 2013:24-76.
-
Singla A, Amini MR, Alpert MA, Gornik HL. Fatal anaphylactoid reaction associated with heparin-induced thrombocytopenia. Vasc Med. 2013;18(3):136-138. doi:10.1177/1358863X13483865.
-
Anders D, Trautmann A. Allergic anaphylaxis due to subcutaneously injected heparin. Allergy Asthma Clin Immunol. 2013;9(1):1. doi:10.1186/1710-1492-9-1.
-
Nikolis A, Christopoulos A, Saint-Cyr M, Cordoba C, Guertin L, Harris PG. Recurrent venous thrombosis following free flap surgery: the role of heparin-induced thrombocytopenia. Can J Plast Surg. 2003;11(1):37-40. doi:10.1177/229255030301100107.
-
Zaman SR, Rawlins JM. Heparin induced thrombocytopaenia (HIT) as a cause of free flap failure in lower limb trauma. J Plast Reconstr Aesthet Surg. 2014;67(6):884-886. doi:10.1016/j.bjps.2013.12.027.
-
LaMuraglia GM, Houbballah R, Laposata M. The identification and management of heparin-induced thrombocytopenia in the vascular patient. J Vasc Surg. 2012;55(2):562-570. doi:10.1016/j.jvs.2011.10.082.
-
Warkentin TE. How I diagnose and manage HIT. Hematology Am Soc Hematol Educ Program. 2011;2011(1):143-149. doi:10.1182/asheducation-2011.1.143.
-
Warkentin TE, Greinacher A. Laboratory testing for heparin-induced thrombocytopenia. In: Warkentin TE, Greinacher A, eds. Heparin-Induced Thrombocytopenia. 5th ed. CRC Press; 2013:272-314.
-
Gurtner GC, Jones GE, Neligan PC, et al. Intraoperative laser angiography using the SPY system: review of the literature and recommendations for use. Ann Surg Innov Res. 2013;7(1):1. doi:10.1186/1750-1164-7-1.
-
Hill WF, Webb C, Monument M, McKinnon G, Hayward V, Temple-Oberle C. Intraoperative near-infrared spectroscopy correlates with skin flap necrosis: a prospective cohort study. Plast Reconstr Surg Glob Open. 2020;8(4):e2742. doi:10.1097/GOX.0000000000002742.
-
Mehdizade T, Kelahmetoglu O, Gurkan V, Cetin G, Guneren E. Early suspicion of heparin-induced thrombocytopenia for successful free flap salvage: reports of two cases. J Hand Microsurg. 2021;13(3):178-180. doi:10.1055/s-0040-1713692.
-
Warkentin TE, Greinacher A. Treatment of heparin-induced thrombocytopenia: an overview. In: Warkentin TE, Greinacher A, eds. Heparin-Induced Thrombocytopenia. 5th ed. CRC Press; 2013:315-355.
-
Pflipsen MC, Vega Colon KM. Anaphylaxis: recognition and management. Am Fam Physician. 2020;102(6):355-362.