Carfentanil and the EMS Response: Pharmacology, Clinical Challenges, and Emerging Treatments
At a recent conference presented by Biomedical Advanced Research and Development Authority (BARDA), presentations on the potential weaponization of opiates and the confounding and confusing landscape relevant to substance use were presented. (As a note of disclosure, I made a presentation at this conference on the EMS response to opioids in the United States).1
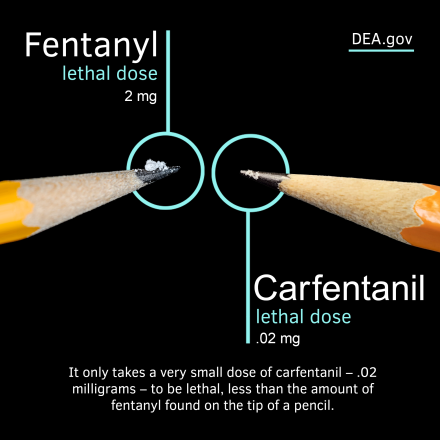
In the mid-1980s black tar heroin threatened to overturn the landscape for substance use disorders and EMS. It was quickly overshadowed by crack cocaine. In the late 1990s and early 2000s it was prescription opiates, with oxycodone dominating the discussion in public health departments and the nightly news.1 Now fentanyl has brought tragedy to every community in the U.S.1-2 In recent years, the emergence of ultra-potent synthetic opioids lurks around the corner, ready to reshape the landscape of EMS. Among the most alarming of these substances is carfentanil,2-3 a synthetic opioid estimated to be approximately 10,000 times more potent than morphine and 100 times more potent than fentanyl.2 We have seen a sevenfold increase in deaths related to carfentanil from January 2021 to June 2024.2
Originally developed as a veterinary anesthetic for large animals such as elk and elephants,1,2,4 carfentanil found its way into the illicit drug supply around a decade ago, often mixed with heroin, fentanyl, or other drugs. It has now reemerged,2 creating a high-risk environment not only for users but also for emergency responders tasked with managing the aftermath of overdose events.
Carfentanil has the potential to be weaponized for use in a terror incident and was used in Russia by federal forces to incapacitate terrorists in a movie theater.1,5 For EMS professionals, understanding the pharmacology of carfentanil, its differences from fentanyl, and the limitations of existing treatment protocols is essential. Equally important is staying informed about emerging therapeutic strategies that may soon transform how we approach opioid overdose in the field.
Pharmacology of Carfentanil
Carfentanil belongs to the fentanyl family of synthetic opioids, which act primarily on the mu-opioid receptor to produce analgesia, sedation, and euphoria. However, the potency of carfentanil exceeds that of its analogs by several orders of magnitude. Its lipid solubility allows it to cross the blood-brain barrier rapidly, leading to profound and often sudden respiratory depression. Unlike morphine or fentanyl, carfentanil exhibits an exceptionally high receptor binding affinity and a prolonged duration of action, both of which complicate field management.1-3
What differentiates carfentanil pharmacodynamically from other opioids is the rate at which it transfers from plasma to the receptor sites.1-3 According to pharmacokinetic-pharmacodynamic (PK-PD) modeling, this rate of transfer is a key determinant in predicting the onset and severity of respiratory depression.1-3 For instance, while fentanyl has a plasma-to-receptor site half-life of approximately 4.7 to 6.6 minutes, morphine's half-life is measured in hours.1-3 Carfentanil, due to its ultra-potency and high receptor affinity, initiates respiratory failure with alarming speed and intensity, often outpacing the ability of bystanders to react, and for EMS clinicians to respond before the patient proceeds into respiratory arrest.
Differences Between Carfentanil and Fentanyl
Although both substances are classified as synthetic opioids, the clinical presentation of carfentanil overdose differs in important ways from fentanyl overdose. First, the dose-response relationship is significantly narrower with carfentanil, meaning the margin between a psychoactive dose and a lethal dose is exceedingly small.1-2 This makes accidental overdose not only more likely but also more difficult to reverse.
Second, carfentanil is more resistant to reversal by naloxone, the standard opioid antagonist used in EMS.1,6 In fentanyl overdoses, a typical naloxone dose of 2 mg to 4 mg may suffice; however, carfentanil overdoses often require multiple, repeated doses or a continuous infusion. Additionally, due to its long half-life and high receptor affinity, patients may experience renarcotization, re-sedation, and respiratory depression after initial reversal, necessitating prolonged monitoring and potentially advanced airway management.
Third, it’s telling that carfentanil is used for sedation and analgesia for elk and elephants.4 The relationship between a person’s or animal’s weight and the dosage or suitability of certain medications is primarily rooted in pharmacokinetics—how the body absorbs, distributes, metabolizes, and excretes a drug. Larger animals, such as elephants, typically require higher doses or more potent drugs due to their massive body mass and slower metabolic rates. In comparison, a small dose of carfentanil for a human would be lethal. Conversely if one were to use fentanyl on an elephant, it would require prohibitively high volumes to sedate them because of their size and metabolism.
Traditional EMS Treatment Approaches and Their Limitations
The standard EMS response to opioid overdose involves airway management, ventilation with supplemental oxygen, and administration of naloxone.7-8 However, as noted, naloxone has limitations when used against ultra-potent opioids.1,3,6 The drug's short half-life (30 to 90 minutes) is often inadequate to match the prolonged effects of carfentanil, leading to a mismatch in therapeutic coverage. Repeated intramuscular (IM) or intranasal doses may be needed, and in severe cases—because renarcotization can occur—the use of IV administration and continuous infusions is a necessity, resources typically used by advanced-level EMS clinicians.
Nalmefene may be a better choice for the initial treatment and resuscitation.1-6 Nalmefene is an opioid receptor antagonist similar to naloxone but with a significantly longer half-life, making it a better choice for treating overdoses involving ultra-potent synthetic opioids such as carfentanil. Nalmefene binds strongly to the same opioid receptors but remains active in the body for eight hours or more, reducing the likelihood of rebound respiratory depression and the need for repeated dosing.1,6 Its extended duration and high receptor affinity make it a potentially more effective and safer option in managing overdoses from high-potency opioids in resource-limited settings.
Nalmefene may be administered IV, IM, or intranasal (IN).1,6 This is important because if carfentanil has the same impact on the American landscape as fentanyl, we may need to deploy IN nalmefene instead of naloxone for public access/leave behind medication programs.
Airway and ventilation support become critical when naloxone proves insufficient; they are always the first two EMS interventions for any and all overdoses in which the patient has a decreased level of consciousness, an impaired airway, and insufficient ventilations.7-8 EMS personnel must be prepared to escalate care quickly, including using bag-valve mask ventilation or even transitioning to endotracheal intubation if reversal agents are unsuccessful.
Emerging Treatments: Monoclonal Antibodies and Respiratory Stimulants
One thing I find surprising is the number of EMS agencies that don’t carry reversal agents specific to the medications they use. While everyone has naloxone for opiates, rarely do we see agencies carry flumazenil for benzodiazepine overdose or sugammadex for rocuronium, even though these are sedatives or paralytics we may carry in our med boxes.
Recognizing the limitations of naloxone, researchers have begun to explore new therapeutic avenues. Among the most promising are monoclonal antibodies (mAbs) engineered to target synthetic opioids such as fentanyl and carfentanil.9-10 These antibodies work by binding to opioid molecules in the bloodstream, preventing them from crossing the blood-brain barrier and reaching their receptor targets. Because they don’t cross into the central nervous system, they do not interfere with endogenous opioid signaling, reducing the risk of withdrawal and potentially allowing for longer-term protection.9-10
One such mAb, CSX-1004, has demonstrated efficacy in preclinical studies by reducing the potency and duration of fentanyl-induced respiratory depression.10 While not yet widely available for clinical use, monoclonal antibodies represent a viable future intervention for both pre- and post-exposure scenarios. A single dose of mAb could prevent a patient from overdosing for weeks.
For EMS, these therapies could someday be incorporated into hospital-based treatment protocols or even in advanced EMS systems with comprehensive systems of care for treating and assisting patients with substance use disorder at the scene.
In addition to mAbs, novel respiratory stimulants such as Sudaxine are being developed to counteract opioid-induced respiratory depression without interfering with analgesia.11 Sudaxine increases ventilatory drive through nitric oxide-thiolate signaling pathways, offering an adjunct or alternative to opioid antagonists. Early animal studies have shown that it may reverse respiratory depression caused by xylazine-fentanyl combinations, scenarios increasingly seen in the field offering a potential solution to solo xylazine overdose as well.11
These new therapies aren’t ready for deployment yet, but they hold significant promise if they successfully complete their clinical trials.
Clinical Implications for EMS Providers
For EMS professionals, the emergence of carfentanil as a street-level drug necessitates an evolution in both training and clinical protocols. Field providers must be aware that traditional indicators such as respiratory rate and mental status may not fully reflect the severity of carfentanil toxicity. Tools like pulse oximetry, while useful, have limitations, particularly in patients with darker skin tones or peripheral vasoconstriction.12-15 The delay in pulse oximetry readings, often 17 seconds to several minutes, means providers must make rapid decisions based on clinical presentation rather than waiting for monitor confirmation.
Assessment tools should include evaluation of accessory muscle use, patient positioning, phonation ability, and mental status over time.7-8 Realistically we don’t want to fully reverse patients. They are anoxic and will wake up scared, going from a dreamlike state to a room full of strangers in uniforms. We compound this situation if we fully reverse their overdose; they may then proceed into withdrawal, with nausea and vomiting. They are more apt to refuse further treatment and transport at this point, and if offered suboxone, they may refuse that as well.
Instead we should go with smaller doses of naloxone: 0.4 mg to 0.8 mg initially. We want the patient rousable, having the ability to maintain their own airway and breath, but not conscious enough to refuse further treatment and transport. Those patients who are then transported to hospitals can then be brought out in a more controlled environment, and most importantly, trained staff can better manage the patient transition to medication-assisted therapy and detoxification.
A patient who is unable to speak or who displays a silent chest despite obvious distress may be in the late stages of respiratory collapse. These signs should prompt immediate airway intervention, even if pulse oximetry readings appear stable.
Frequent reassessment is critical. Initial improvement with naloxone may be temporary, and without continuous monitoring, patients may rapidly decline. EMS systems must adopt protocols that emphasize the need for prolonged observation, possibly in conjunction with emergency department coordination for patients who have received high-dose or repeated naloxone administration. When carfentanil supplants fentanyl in the community, the days of reversing overdoses and allowing the patient to refuse transport may become a thing of the past. Nalmefene may replace naloxone on every EMS unit, and it will most likely become the treatment of choice in our leave-behind medication programs.
Conclusion
Carfentanil represents a new frontier in the opioid epidemic, one marked by extreme potency, rapid onset of life-threatening symptoms, a transition to nalmefene to improve resuscitation, and resistance to standard treatments. EMS professionals must be equipped not only with pharmacological knowledge but also with the clinical acumen to recognize subtle and severe presentations of overdose. As traditional tools like naloxone face limitations, emerging therapies such as monoclonal antibodies and novel respiratory stimulants offer hope for more effective, sustained treatment.
The future of EMS care in opioid overdose will likely involve a multi-modal approach, integrating airway management, pharmacological reversal, and longer-term solutions tailored to synthetic opioids. Education, training, and system-level adaptations will be key to ensuring that EMS remains at the forefront of response in this evolving public health crisis.
References
- National Institutes of Health, Assistant Secretary of Preparedness and Response, Biomedical Advanced Research and Development Authority (BARDA), Advances in Developing Medical Countermeasures Against Opioids and Drug-Induced Respiratory Depression December 17, 2024 – December 18, 2024
- Tanz, L. J., Stewart, A., Gladden, R. M., Ko, J. Y., Owens, L., & O’Donnell, J. (2024). Detection of illegally manufactured fentanyls and carfentanil in drug overdose deaths , United States, 2021–2024. Morbidity and Mortality Weekly Report, 73(48), 1099–1105. https://www.cdc.gov/mmwr/volumes/73/wr/mm7348a2.htm
- Jalal H, Burke DS. Carfentanil and the rise and fall of overdose deaths in the United States. Addiction. 2021 Jun;116(6):1593-1599. doi: 10.1111/add.15260. Epub 2020 Sep 25. PMID: 32935381; PMCID: PMC8019064.
- Miller, M. W., Wild, M. A., & Lance, W. R. (1996). Efficacy and safety of naltrexone hydrochloride for antagonizing carfentanil citrate immobilization in captive Rocky Mountain elk (Cervus elaphus nelsoni). Journal of wildlife diseases, 32(2), 234-239.
- Tin D, Kallenborn Z, Hart A, Hertelendy AJ, Ciottone GR. Opioid Attack and the Implications for Counter-Terrorism Medicine. Prehospital and Disaster Medicine. 2021;36(6):661-663. doi:10.1017/S1049023X21001059
- Laffont, C. M., Bhaskar, S., & Mann, A. (2024). Comparison of intranasal naloxone and intranasal nalmefene in a translational model assessing the impact of synthetic opioid overdose on respiratory depression and cardiac arrest. Frontiers in Psychiatry, 15, 1399803. https://doi.org/10.3389/fpsyt.2024.1399803
- AMLS: Advanced Medical Life Support: Advanced Medical Life Support 3rd Edition by National Association of Emergency Medical Technicians (NAEMT), Jones and Bartlett, 2021
- “The Paramedic,” McGraw-Hill, NY, NY April 2011
- Hicks, D., Baehr, C., Silva-Ortiz, P., Khaimraj, A., Luengas, D., Hamid, F. A., & Pravetoni, M. (2022). Advancing humanized monoclonal antibody for counteracting fentanyl toxicity towards clinical development. Human Vaccines & Immunotherapeutics, 18(6). https://doi.org/10.1080/21645515.2022.2122507
- Bremer, P. T., Burke, E. L., Barrett, A. C., et al. (2023). Investigation of monoclonal antibody CSX-1004 for fentanyl overdose. Nature Communications, 14, 7700. https://doi.org/10.1038/s41467-023-43126-0
- Altawallbeh G, Smith L, Lewis SJ, et al. Pharmacokinetic study of Sudaxine in dog plasma using novel LC–MS/MS method. Drug Test Anal. 2019; 11: 403–410. https://doi.org/10.1002/dta.2507
- Bickler PE, Feiner JR, Severinghaus JW. Effects of skin pigmentation on pulse oximeter accuracy at low saturation. Anesthesiology. 2005 Apr;102(4):715-9. doi: 10.1097/00000542-200504000-00004. PMID: 15791098.
- Shi C, et al. “The accuracy of pulse oximetry in measuring oxygen saturation by levels of skin pigmentation: a systematic review and meta-analysis.” BMC Med. 2022 Aug 16;20(1):267. doi: 10.1186/s12916-022-02452-8. PMID: 35971142; PMCID: PMC9377806.
- Gudelunas MK, Lipnick M, Hendrickson C, Vanderburg S, Okunlola B, Auchus I, Feiner JR, Bickler PE. Low Perfusion and Missed Diagnosis of Hypoxemia by Pulse Oximetry in Darkly Pigmented Skin: A Prospective Study. Anesth Analg. 2024 Mar 1;138(3):552-561. doi: 10.1213/ANE.0000000000006755. Epub 2023 Dec 18. PMID: 38109495.
- Sylvia E K Sudat, Paul Wesson, Kim F Rhoads, Stephanie Brown, Noha Aboelata, Alice R Pressman, Aravind Mani, Kristen M J Azar, Racial Disparities in Pulse Oximeter Device Inaccuracy and Estimated Clinical Impact on COVID-19 Treatment Course. American Journal of Epidemiology, Volume 192, Issue 5, May 2023, Pages 703–713, https://doi.org/10.1093/aje/kwac164











